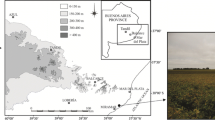
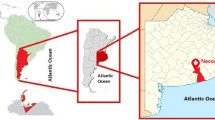
Abstract
Aims
Silicophytoliths contribute to the pool of amorphous silica that is incorporated into the soils acting as intermediary in the biogeochemical cycle of silicon. Most studies focus their attention in the production of aerial parts of plants but not in roots. In order to advance into the knowledge about the relevance of root amorphous silica in soil Si cycle, we studied the silicophytolith content of roots from grasses and crops, and from soil’s roots of different sites from Pampean region, Argentina, one of the most fertile and used for agricultural practices.
Methods
Roots from three crops and seven grasses and from soils under three different uses were studied. Silicophytoliths were extracted by a calcination technique.
Results
All the species produced silicophytoliths derived from vascular and/or endodermal tissues. Silicophytolith content was higher in Pampean grasses (6.34–15.38%) than in crops (0.01–1.58%). Root silicophytolith content in the pasture was 10 times higher than in the other sites.
Conclusions
The potential input of root silica to soils is related to the type and cover of vegetation, the amount of roots in soils and the silicophytoliths accumulated in roots. Results suggested that root silicophytoliths should be consider in future studies of biogeochemical cycle of Si.



Similar content being viewed by others
Abbreviations
- RC:
-
Content of roots present in soil samples
- SiR:
-
The content of silicophytoliths in soil roots
- SiRS:
-
The input of silicophytolith from roots to soils
References
Albert RM, Bamford MK, Cabanes D (2006) Taphonomy of phytoliths and macroplants in different soils from Olduvai Gorge (Tanzania) and the application to Plio-Pleistocene palaeoanthropological samples. Quat Int:78–94. https://doi.org/10.1016/j.quaint.2005.11.026
Alvarez MF, Osterrieth M (2018) Submicroscopy of aggregates of Luvic Phaeozems under different land uses in the southeast of the Buenos Aires Province, Argentina. Eurasian Soil Sci:1487–1496. https://doi.org/10.1134/S106422931813001X
Alvarez MF, Osterrieth M, del Río JL (2012) Changes on aggregates morphology and roughness of induced by different uses of typical Argiudolls, Buenos Aires province, Argentina. Soil Tillage Res 119:38–49. https://doi.org/10.1016/j.still.2011.12.003
Bennett DM (1982) Silicon deposition in the roots of Hordeum sativum Jess, Avena sativa L and Triticum aestivum L, Ann Bot: 239–245. doi: https://doi.org/10.1093/oxfordjournals.aob.a086361
Bertoldi de Pomar H (1975) Los silicofitolitos: sinopsis de su conocimiento. Darwiniana:173–206 http://www.jstor.org/stable/23215540. Accessed 22 March 2018
Borrelli N, Osterrieth M, Marcovecchio J (2008) Interrelations of vegetal cover, silicophytolith content and pedogenesis of typical Argiudolls of the Pampean plain, Argentina. Catena:146–153. https://doi.org/10.1016/j.catena.2008.05.001
Borrelli N, Álvarez MF, Osterrieth M, Marcovecchio JE (2010) Silica content in soil solution and its relation with phytolith weathering and silica biogeochemical cycle in typical Argiudolls of the Pampean plain, Argentina:a Preliminary Study. J Soils Sediments:983–994. https://doi.org/10.1007/s11368-010-0205-7
Burgos JJ, Vidal AL (1951) Los climas de la República Argentina según la nueva clasificación de Thornthwaite. Meteor-Forschung:3–32
Cabrera A (1976) Regiones fitogeográficas de la Argentina. Enciclopedia Argentina de Agricultura y Ganadería, Tomo II, Acme SACI, Buenos Aires
Camargo OA, Alleoni LRF (1997) Compactacan do solo e o dsenvolvimiento das plantas. Escola Superior de Agricultura Luiz de Querioz, Piracicaba, p 132
Cornelis JT, Titeux H, Ranger J, Delvaux B (2011) Identification and distribution of the readily soluble silicon pool in a temperate forest soil below three distinct tree species. Plant Soil:369–378. https://doi.org/10.1007/s11104-010-0702-x
Derry LA, Kurtz AC, Ziegler K, Chadwick OA (2005) Biological control of terrestrial silica cycling and export fluxes to watersheds. Nature:728–731. https://doi.org/10.1038/nature03299
Elger A, Lemoine DG, Fenner M, Hanley ME (2009) Plant ontogeny and chemical defence: older seedlings are better defended. Oikos:767–773. https://doi.org/10.1111/j.1600-0706.2009.17206.x
Epstein E (1994) The anomaly of silicon in plant biology. Proc Natl Acad Sci:11–17. https://doi.org/10.1073/pnas.91.1.11
Epstein E (1999) Silicon. Annu Rev Plant Physiol Plant Mol Biol:641–644. https://doi.org/10.1146/annurev.arplant.50.1.641
Fahn A (1967) Plant Anatomy. New York, Pergamon Press, Oxford
Farmer VC, Delbos E, Miller JD (2005) The role of phytolith formation and dissolution in controlling concentrations of silica in soil solutions and streams. Geoderma:71–79. https://doi.org/10.1016/j.geoderma.2004.11.014
Fernández Honaine M, Benvenuto ML, Borrelli NL, Osterrieth M (2016) Early Silicification of leaves and roots of seedlings of a panicoid grass grown under different conditions: anatomical relations and structural role. Plant Biol:1025–1030. https://doi.org/10.1111/plb.12488
Fernández Honaine M, Benvenuto ML, Osterrieth M (in press) An easy technique for silicophytolith visualization in plants through tissue clearing and immersion oil mounting. Boletín de la Sociedad Argentina de Botánica
Fraysse F, Pokrovsky OS, Schott J, Meunier JD (2006) Surface properties, solubility and dissolution kinetics of bamboo phytoliths. Geochim Cosmochim:1939–1951. https://doi.org/10.1016/j.gca.2005.12.025
Fredlund GG, Tieszen LL (1997) Phytolith and carbon isotope evidence for Late Quaternary vegetation and climate change in the southern Black Hills, South Dakota. Quat Res:206–217. https://doi.org/10.1006/qres.1996.1862
Geis JW (1978) Biogenic opal in three species of Gramineae. Ann Bot:1119–1129. https://doi.org/10.1093/oxfordjournals.aob.a085552
Gocke M, Liang W, Sommer M, Kuzyakov Y (2013) Silicon uptake by wheat: effects of Si pools and pH. J Plant Nutr Soil Sci:1–10. https://doi.org/10.1002/jpln.201200098
Guntzer F, Keller C, Poulton PR, McGrath SP, Meunier JD (2012) Long-term removal of wheat straw decreases soil amorphous silica at Broadbalk, Rothamsted. Plant Soil:173–184. https://doi.org/10.1007/s11104-011-0987-4
Hernandez-Apaolaza L (2014) Can silicon partially alleviate micronutrient deficiency in plants? A review. Planta:447–458. https://doi.org/10.1007/s00425-014-2119-x
Hiweris SO (1987) Nature of resistance to Striga hermonthica (Del.) Benth. parasitism in some Sorghum vulgare (Pers.) Cultivars. Weed Res:305–311. https://doi.org/10.1111/j.1365-3180.1987.tb00767.x
Hodson MJ (2016) The development of phytoliths in plants and its influence on their chemistry and isotopic composition. Implications for palaeoecology and archaeology. J Archaeol Sci:62–69. https://doi.org/10.1016/j.jas.2015.09.002
Hodson MJ, Sangster AG (1989) Subcellular localization of mineral deposits in the roots of wheat (Triticum aestivum L.). Protoplasma:19–32. https://doi.org/10.1007/BF01403298
Hodson MJ, White PJ, Mead A, Broadly MR (2005) Phylogenetic variation in the silicon composition of plants. Ann Bot:1027–1046. https://doi.org/10.1093/aob/mci255
Hussain MI, González L, Reigosa MJ (2011) Allelopathic potential of Acacia melanoxylon on the germination and root growth of native species. Weed Biol Manag:18–28. https://doi.org/10.1111/j.1445-6664.2011.00401.x
Ikegami N, Satake T, Nagayama Y, Kazuyuki I (2014) Changes in silica in litterfall and available silica in the soil of forests invaded by bamboo species (Phyllostachys pubescens and P. bambusoides) in western Japan. Soil Sci Plant Nutr:731–739. https://doi.org/10.1080/00380768.2014.942794
INTA (1989) Mapa de suelos de la provincia de Buenos Aires, E 1: 500000. SAGyP-INTA, Buenos Aires
Jones LHP, Handreck KA (1967) Silica in soils, plants, and animals. In: Norman AG (ed) Adv agron. Academic Press, New York, pp 107–149. https://doi.org/10.1016/S0065-2113(08)60734-8
Labouriau LG (1983) Phytolith work in Brasil, a mini-review. Phytolitharien Newsletter:6–11
Lux A, Luxová M, Morita S, Abe J, Inanaga S (1999) Endodermal silicification in developing seminal roots of lowland and upland cultivars of rice (Oryza sativa L.). Can J Bot:955–960. https://doi.org/10.1139/b99-043
Lux A, Luxová M, Hattori T, Inanaga S, Sugimoto Y (2002) Silicification in sorghum (Sorghum bicolor) cultivars with different drought tolerance. Physiol Plant:87–92. https://doi.org/10.1034/j.1399-3054.2002.1150110.x
Lux A, Luxová M, Abe J, Morita S, Inanaga S (2003a) Silicification of bamboo (Phyllostachys heterocycla Mitf.) root and leaf. In: Abe J (ed) Roots: the dynamic Interface between plants and the earth. Springer, Dordrecht, pp 85–91
Lux A, Luxová M, Abe J, Tanimoto E, Hattori T, Inanaga S (2003b) The dynamics of silicon deposition in the sorghum root endodermis. New Phytol:437–441. https://doi.org/10.1046/j.1469-8137.2003.00764.x
Ma JF (2004) Role of silicon in enhancing the resistance of plants to biotic and abiotic stresses. Soil Sci Plant Nutr:11–18. https://doi.org/10.1080/00380768.2004.10408447
Ma JF, Takahashi E (2002) Soil, fertilizer, and plant silicon research in Japan. Elsevier, Amsterdam
Ma JF, Yamaji N, Mitani-Ueno N (2011) Transport of silicon from roots to panicles in plants. Proc Jpn Acad Ser B:377–385. https://doi.org/10.2183/pjab.87.377
Ma D, Sun D, Wang C, Qin H, Ding H, Li Y, Guo T (2016) Silicon application alleviates drought stress in wheat through transcriptional regulation of multiple antioxidant defense pathways. J Plant Growth Regul:1–10. https://doi.org/10.1007/s00344-015-9500-2
Madella M, Alexandre A, Ball T (2005) International code for Phytolith nomenclature 1.0. Ann Bot:253–260. https://doi.org/10.1093/aob/mci172
Maguire TJ, Templer PH, Battles JJ, Fulweiler RW (2017) Winter climate change and fine root biogenic silica in sugar maple trees (Acer saccharum): implications for silica in the Anthropocene. J Geophys Res Biogeosci:708–715. https://doi.org/10.1002/2016JG003755
Massey FP, Ennos AR, Hartley SE (2007) Herbivore specific induction of silica-based plant defences. Oecologia:677–683. https://doi.org/10.1007/s00442-007-0703-5
Materechera SA, Alston AM, Kirby JM, Dexter AR (1992) Influence of root diameter on the penetration of seminal roots into a compacted subsoil. Plant Soil:297–303. https://doi.org/10.1007/BF00012888
McNaughton SJ, Tarrants JL, McNaughton MM, Davis RH (1985) Silica as a defense against herbivory and a growth promotor in African grasses. Ecology:528–535. https://doi.org/10.2307/1940401
Montti L, Fernández Honaine M, Osterrieth M, Graciano Ribeiro D (2009) Phytolith analysis of Chusquea ramosissima Lindm. (Poaceae: Bambusoideae) and associated soils. Quat Int:80–89. https://doi.org/10.1016/j.quaint.2007.11.024
Motomura H, Fujii T, Suzuki M (2000) Distribution of silicified cells in the leaf blades of Pleioblastus chino (Franchet et Savatier) Makino (Bambusoideae). Ann Bot:751–757. https://doi.org/10.1006/anbo.2000.1124
Osterrieth M, Madella M, Zurro D, Álvarez MF (2009) Taphonomical aspects of silicaphytoliths in the loess sediments of the Argentinean pampas. Quat Int 193:70–79. https://doi.org/10.1016/j.quaint.2007.09.002
Osterrieth M, Borrelli N, Álvarez MF, Fernández Honaine M (2015) Silica biogeochemical cycle in temperate ecosystems of the pampean plain, Argentina. J S Am Earth Sci:172–179. https://doi.org/10.1016/j.jsames.2015.07.011
Parry DW, Kelso M (1977) The ultrastructure and analytical microscopy of silicon deposits in the roots of Saccharum officinarum (L.). Ann Bot:855–862. https://doi.org/10.1093/oxfordjournals.aob.a085361
Piperno DR (1988) Phytolith analysis: an archaeological and geological perspective. Academic Press, San Diego
Reynolds J, Lambin X, Massey FP, Reidinger S, Sherratt JA, Smith MJ, White A, Hartley SE (2012) Delayed induced silica defences in grasses and their potential for destabilising herbivore population dynamics. Oecologia:445–456. https://doi.org/10.1007/s00442-012-2326-8
Sangster AG (1977) Electron-probe microassay studies of silicon deposits in the roots of two species of Andropogon. Can J Bot 55:880–887. https://doi.org/10.1139/b77-107
Sangster AG (1978) Silicon in the roots of higher plants. Am J Bot:929–935. https://doi.org/10.1002/j.1537-2197.1978.tb06157.x
Sangster AG, Parry DW (1976) Endodermal silicon deposits and their linear distribution in developing roots of Sorghum bicolor (L.) Moench. Ann Bot: 361–371. https://doi.org/10.1093/oxfordjournals.aob.a085139
Sangster AG, Parry DW (1981) Ultrastructure of silica in higher plants. In: Simpson TL, Volcani BE (eds) Silicon and siliceous structures in biological systems. Springer, New York, pp 383–407
Scanlan CA, Davies SL (2019) Soil mixing and redistribution by strategic deep tillage in a sandy soil. Soil Tillage Res:139–145. https://doi.org/10.1016/j.still.2018.09.008
Schoelynck J, Muller F, Vandevenne F, Bal K, Barao L, Smis A, Opdekamp W, Meire P, Struyf E (2014) Silicon–vegetation interaction in multiple ecosystems: a review. J Veg Sci:301–313. https://doi.org/10.1111/jvs.12055
Servicio Meteorológico Nacional (2010) Estadísticas climatológicas. Publicaciones del Servicio Meteorológico Nacional, Buenos Aires
Soil Survey Staff (1996) Keys to soil taxonomy, 7th edn. Department of Agriculture, Washington, D.C.
Soininen EM, Brathen KA, Herranz Jusdado JG, Reidinger S, Hartley SE (2013) More than herbivory: levels of silica-based defences in grasses vary with plant species, genotype and location. Oikos:30–41. https://doi.org/10.1111/j.1600-0706.2012.20689.x
Sommer M, Kaczorek D, Kuzyakov Y, Breuer J (2006) Silicon pools and fluxes in soils and landscapes. A Review. J Plant Nutr Soil Sci:310–329. https://doi.org/10.1002/jpln.200521981
Soukup M, Martinka M, Bosnic D, Caplovicova M, Elbaum R, Lux A (2017) Formation of silica aggregates in Sorghum root endodermis is predetermined by cell wall architecture and development. Ann Bot:739–753. https://doi.org/10.1093/aob/mcx060
Tardieu F, Manichon H (1986) Caracterisation en tant que capture d ´eau de l'enracinement du mais en parcelle cultive. II Une methode d'etude de la repartition verticale et horizontale des racine. Agronomie: 415–425. https://hal.archives-ouvertes.fr/hal-00884892/file/hal-00884892.pdf. Accessed 22 March 2018
Tubana BS, Tapasya B, Datnoff LE (2016) A review of silicon in soils and plants and its role in US agriculture: history and future perspectives. Soil Sci:393–411. https://doi.org/10.1097/SS.0000000000000179
Twiss PC (1992) Predicted world distribution of C3 and C4 grass phytoliths. In: Rapp G, Mulholland SC (eds) Phytolith systematics. Advances in archaeological and museum science, vol 1. Springer, Boston
Wieczorek M, Zub K, Szafranska PA, Ksiazek A, Konarzewski M (2015) Plant–herbivore interactions: silicon concentration in tussock sedges and population dynamics of root voles. Functional Ecology 187–194. https://doi.org/10.1111/1365-2435.12327
Zar JH (1984) Biostatistical analysis, 2nd edn. Prentice – Hall, INC, New Jersey
Acknowledgements
Fieldwork was supported by National University of Mar del Plata (UNMdP), Buenos Aires, Argentina (EXA 839/17) and Fund for Scientific and Technology Research (FONCyT - PICT 2495/17). A fellowship from National Council for Scientific and Technical Research (UNMdP-CONICET) to Micaela Paolicchi is acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Jian Feng Ma.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Paolicchi, M., Benvenuto, M.L., Honaine, M.F. et al. Root silicification of grasses and crops from the Pampean region and its relevance to silica and silicophytolith content of soils. Plant Soil 444, 351–363 (2019). https://doi.org/10.1007/s11104-019-04287-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-019-04287-4