Abstract
Background
This study started from typical replant disease symptoms limited to specific foci within three multi-generation orchards showing homogeneous growth.
Methods
A plant growth assay was conducted using soil from symptomatic and asymptomatic tree root zones along planted rows and from strip-rows. Root colonizing fungal communities were investigated, then the study turned to pathogenicity of Cylindrocarpon-like fungi (Dactylonectria and Ilyonectria spp) and their extracellular exudates.
Results
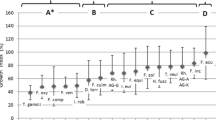
Growth of apple rootstock plantlets in soil from symptomatic trees was significantly lower than in those observed in soil from asymptomatic trees and from strip-rows. Among the main group of endophytic filamentous fungi isolated from roots, Cylindrocapon-like fungi (Dactylonectria torresensis and, to a lesser extent, Ilyonectria robusta), along with binucleate Rhizoctonia spp., prevailed mostly in plantlets grown in soil collected from planted rows. On the other hand, Fusarium spp. prevailed in plantlets grown in soil from the strip-rows. Cylindrocarpon-like fungi was found to be the most negatively correlated with plant growth. As findings of artificial inoculation with main root colonizing fungal species were not in line with what was observed in native soils, a further investigation was performed on secondary metabolites through which Dactylonectria torresensis exerts pathogenicity; this analysis revealed that both phytotoxins (tentoxin, HC toxin and zearalenone) and cytotoxic compounds (rabelomycin and nidulin) may be involved.
Conclusion
Findings suggest that extracellular compounds released by D. torresensis may have contributed to the severe growth reduction associated with replant disease-like symptoms.




Similar content being viewed by others
Abbreviations
- AG:
-
Anastomosis group
- ARD:
-
Apple replant disease
- bnRizoctonia :
-
Binucleate Rhizoctonia sp.
References
Bachelot B, Uriarte M, Zimmerman J et al (2016) Long-lasting effects of land use history on soil fungal communities in second-growth tropical rain forests. Ecol Appl 26:1881–1895. https://doi.org/10.1890/15-1397.1
Barbetti MJ (2005) Cylindrocarpon didymum — a root pathogen of subterranean clover in the lower south-west of Western Australia. Australas Plant Pathol 34:111. https://doi.org/10.1071/AP04088
Braun PG (1991) The combination of Cylindrocarpon lucidum and Pythium irregulare as a possible cause of apple replant disease in Nova Scotia. Can J Plant Pathol 13:291–297. https://doi.org/10.1080/07060669109500914
Braun PG, Fuller KD, McRae K, Fillmore SAE (2010) Response of “Honeycrisp®” apple trees to combinations of pre-plant fumigation, deep ripping, and hog manure compost incorporation in a soil with replant disease. Hortscience 45:1702–1707
Broeckling CD, Broz AK, Bergelson J, Manter DK, Vivanco JM (2008) Root exudates regulate soil fungal community composition and diversity. Appl Environ Microbiol 74:738–744. https://doi.org/10.1128/AEM.02188-07
Browne GT, Connell JH, Schneider SM (2006) Almond replant disease and its management with alternative pre-plant soil fumigation treatments and rootstocks. Plant Dis 90:869–876. https://doi.org/10.1094/PD-90-0869
Burke DJ, López-Gutiérrez JC, Smemo KA, Chan CR (2009) Vegetation and soil environment influence the spatial distribution of root-associated fungi in a mature beech-maple Forest. Appl Environ Microbiol 75:7639–7648. https://doi.org/10.1128/AEM.01648-09
CAB (Commonwealth Agricultural Bureau) (1990) Description of Pathogenic Fungi and Bacteria. N.1–1000 from January 1964 to March 1990. CAB International Mycological Institute. Kluwer Acc Pub
Cabral A, Groenewald JZ, Rego C et al (2011) Cylindrocarpon root rot: multi-gene analysis reveals novel species within the Ilyonectria radicicola species complex. Mycol Prog 11:655–688. https://doi.org/10.1007/s11557-011-0777-7
Cabral A, Rego C, Nascimento T et al (2012) Multi-gene analysis and morphology reveal novel Ilyonectria species associated with black foot disease of grapevines. Fungal Biol 116:62–80. https://doi.org/10.1016/j.funbio.2011.09.010
Caputo F, Nicoletti F, De Luca Picione F, Manici LM (2015) Rhizospheric changes of fungal and bacterial communities in relation to soil health of multi-generation apple orchards. Biol Control:88:8–8817. https://doi.org/10.1016/j.biocontrol.2015.04.019
Chaverri P, Salgado C, Hirooka Y et al (2011) Delimitation of Neonectria and Cylindrocarpon (Nectriaceae, Hypocreales, Ascomycota) and related genera with Cylindrocarpon-like anamorphs. Stud Mycol 68:57–78. https://doi.org/10.3114/sim.2011.68.03
Crous PW, Groenewald JZ, Risède J-M et al (2004) Calonectria species and their Cylindrocladium anamorphs: species with sphaeropedunculate vesicles. KNAW research portal. Stud Mycol 50:415–429
Cubeta M, Echandi E, Gumpertz M (1991) Survival of binucleate Rhizoctonia species, biological control agents, in soil and plant debris under field conditions. Biol Control 1:218–226. https://doi.org/10.1016/1049-9644(91)90070-G
Dikilitas M, Karakas S, Hashem A, et al (2016) Oxidative stress and plant responses to pathogens under drought conditions. In Parvaiz A (ed) Chapter 8 in “water stress and crop plants: a sustainable approach. https://doi.org/10.1002/9781119054450.ch8
Evans G, Cartwright JB, White NH (1967) The production of a phytotoxin, nectrolide, by some root-surface isolates of Cylindrocarpon radicicola, Wr. Plant Soil 26:253–260. https://doi.org/10.1007/BF01880175
Franke-Whittle IH, Manici LM, Insam H, Stres B (2015) Rhizosphere bacteria and fungi associated with plant growth in soils of three replanted apple orchards. Plant Soil 395:317–333. https://doi.org/10.1007/s11104-015-2562-x
Gronberg H, Kaparakis G, Sen R (2006) Binucleate Rhizoctonia (Ceratorhiza spp.) as non-mycorrhizal endophytes alter Pinus Sylvestris L. seedling root architecture and affect growth of rooted cuttings. Scand J For Res 21:450–457. https://doi.org/10.1080/02827580601019662
Halleen F, Fourie PH, Crous PW (2006) A review of black foot disease of grapevine. Phytopathol Mediterr 45:55–67. https://doi.org/10.14601/PHYTOPATHOL_MEDITERR-1845
Halloin JM, De Zoeten GA, Walker JC (1970) The effects of tentoxin on chlorophyll synthesis and plastid structure in cucumber and cabbage. Plant Physiol 45:310–314
Hammer Ø, Harper DAT, Ryan PD (2001) Past: paleontological statistics software package for education and data analysis. Palaeontol Electron 4:1–9 http://palaeo-electronica.org/2001_1/past/past.pdf. Accessed 25 August 2017
Kelderer M, Manici LM, Caputo F, Thalheimer M (2012) Planting in the “inter-row” to overcome replant disease in apple orchards: a study on the effectiveness of the practice based on microbial indicators. Plant Soil 357:381–393. https://doi.org/10.1007/s11104-012-1172-0
Maciá-Vicente JG, Jansson H-B, Abdullah SK, Descals E, Salinas J, Lopez-Llorca LV (2008) Fungal root endophytes from natural vegetation in Mediterranean environments with special reference to Fusarium spp. FEMS Microbiol Ecol 64:90–105
Mai WF, Abawi GS (1981) Controlling replant disease of pomeand stone fruit in northern United States by preplant fumigation. Plant Dis 65:859–864
Manici LM, Bonora P (2007) Molecular genetic variability of Italian binucleate Rhizoctonia spp. isolates from strawberry. Eur J Plant Pathol 118:31–42. https://doi.org/10.1007/s10658-007-9100-5
Manici LM, Ciavatta C, Kelderer M, Erschbaumer G (2003) Replant problems in South Tyrol: role of fungal pathogens and microbial population in conventional and organic apple orchards. Plant Soil 256:315–324. https://doi.org/10.1023/A:1026103001592
Manici LM, Kelderer M, Franke-Whittle IH et al (2013) Relationship between root-endophytic microbial communities and replant disease in specialized apple growing areas in Europe. Appl Soil Ecol 72:207–214. https://doi.org/10.1016/j.apsoil.2013.07.011
Manici LM, Kelderer M, Caputo F, Mazzola M (2015) Auxin-mediated relationships between apple plants and root inhabiting fungi: impact on root pathogens and potentialities of growth-promoting populations. Plant Pathol 64:843–851. https://doi.org/10.1111/ppa.12315
Manici LM, Caputo F, Saccà ML (2017) Secondary metabolites released into the rhizosphere by Fusarium oxysporum and Fusarium spp. as underestimated component of nonspecific replant disease. Plant Soil 415:85–98. https://doi.org/10.1007/s11104-016-3152-2
Marasas WFO, Nelson PE, Toussoun TA (1984) Toxigenic Fusarium Species. Identification and Mycotoxicology. The Pennsylvania State University Press, University Park, London, p 230
Masuhara G, Katsuya K, Yamaguchi K (1993) Potential for symbiosis of Rhizoctonia solani and binucleate Rhizoctonia with seeds of Spiranthes Sinensis Var. amoena in vitro. Mycol Res 97:746–752. https://doi.org/10.1016/S0953-7562(09)80156-1
Mazzola M (1997) Identification and pathogenicity of Rhizoctonia spp. isolated from apple roots and orchard soils. Phytopathology 87:582–587
Mazzola M (1998) Elucidation of the microbial complex having a causal role in the development of apple replant disease in Washington. Phytopathology 88:930–938. https://doi.org/10.1094/PHYTO.1998.88.9.930
Mazzola M, Manici LM (2012) Apple replant disease: role of microbial ecology in cause and control. Annu Rev Phytopathol 50:45–65. https://doi.org/10.1146/annurev-phyto-081211-173005
Nikolskaya AN, Panaccione DG, Walton JD (1995) Identification of peptide synthetase-encoding genes from filamentous fungi producing host-selective phytotoxins or analogs. Gene 165:207–211
OECD (2003) Proposal for updating guideline for testing of chemicals 208. OECD Draft Document September 2003. http://www.oecd.org/chemicalsafety/testing/33653757.pdf. Accessed 12 July 2017
Petit E, Gubler WD (2005) Characterization of Cylindrocarpon species, the cause of black foot disease of grapevine in California. Plant Dis 89:1051–1059. https://doi.org/10.1094/PD-89-1051
Pusztahelyi T, Holb IJ, PÃcsi I (2015) Secondary metabolites in fungus-plant interactions. Front Plant Sci 6:573. https://doi.org/10.3389/fpls.2015.00573
Quesada-Ocampo LM, Al-Haddad J, Scruggs AC et al (2016) Susceptibility of maize to stalk rot caused by Fusarium graminearum deoxynivalenol and zearalenone mutants. Phytopathology 106:920–927. https://doi.org/10.1094/PHYTO-09-15-0199-R
Saikkonen K, Faeth SH, Helander M, Sullivan TJ (1998) Fungal endophytes: a continuum of interactions with host plants. Annu Rev Ecol Syst 29:319–343
Scotto La Massese C, Minot JC, Voisin R, Palmieri M (1988) Value of biological test for estimating the infuence og soil type, previous crops and soil sterilization on the growth of peach and apple. Acta Hortic (233):53–60. https://doi.org/10.17660/ActaHortic.1988.233.9
Southey JF (ed) (1970) Laboratory methods for work with plant and soil Nema-todes. Ministry of Agriculture, fisheries and food. Tech. Bull. 2. Her Majesty’sStationary Office. p. 148
Tewoldemedhin YT, Mazzola M, Botha WJ et al (2011a) Characterization of fungi (Fusarium and Rhizoctonia) and oomycetes (Phytophthora and Pythium) associated with apple orchards in South Africa. Eur J Plant Pathol 130:215–229. https://doi.org/10.1007/s10658-011-9747-9
Tewoldemedhin YT, Mazzola M, Labuschagne I, McLeod A (2011b) A multi-phasic approach reveals that apple replant disease is caused by multiple biological agents, with some agents acting synergistically. Soil Biol Biochem 43:1917–1927. https://doi.org/10.1016/j.soilbio.2011.05.014
Tewoldemedhin YT, Mazzola M, Mostert L, McLeod A (2011c) Cylindrocarpon species associated with apple tree roots in South Africa and their quantification using real-time PCR. Eur J Plant Pathol 129:637–651. https://doi.org/10.1007/s10658-010-9728-4
Tuncer S, Eken C (2013) Anastomosis grouping of Rhizoctonia solani and binucleate Rhizoctonia spp. isolated from pepper in Erzincan, Turkey. Plant Prot Sci 49:127–131
Utkhede RS, Li TSC (1988) Determining the occurrence of replant disease in British Columbia orchard and vineyard soils by pasteurization. Can. Plant Dis Surv 68:149–151
van Schoor L, Denman S, Cook NC (2009) Characterisation of apple replant disease under south African conditions and potential biological management strategies. Sci Hortic 119:153–162. https://doi.org/10.1016/j.scienta.2008.07.032
Weiß S, Bartsch M, Winkelmann T (2017) Transcriptomic analysis of molecular responses in Malus domestica “M26” roots affected by apple replant disease. Plant Mol Biol 94:303–318. https://doi.org/10.1007/s11103-017-0608-6
Whitelaw-Weckert MA, Rahman L, Appleby LM et al (2013) Co-infection by Botryosphaeriaceae and Ilyonectria spp. fungi during propagation causes decline of young grafted grapevines. Plant Pathol 62:1226–1237. https://doi.org/10.1111/ppa.12059
Yao S, Merwin IA, Bird GW et al (2005) Orchard floor management practices that maintain vegetative or biomass groundcover stimulate soil microbial activity and alter soil microbial community composition. Plant Soil 271:377–389. https://doi.org/10.1007/s11104-004-3610-0
Zhu Y, Fazio G, Mazzola M (2014) Elucidating the molecular responses of apple rootstock resistant to ARD pathogens: challenges and opportunities for development of genomics-assisted breeding tools. Hortic Res 1:14043. https://doi.org/10.1038/hortres.2014.43
Acknowledgements
This study was performed with funds of BIO-INCROP project provided by CORE Organic II Funding Bodies, being partners of the FP7 ERA-Net project, CORE Organic II (Coordination of European Transnational Research in Organic Farming, www.coreorganic2.org).
We would like to thank Dr. Rita Resca and her staff of Renolab srl (San Giorgio di Piano, Bologna, Italy) for the analysis of fungal secondary metabolites.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Birgit Mitter.
Rights and permissions
About this article
Cite this article
Manici, L.M., Kelderer, M., Caputo, F. et al. Involvement of Dactylonectria and Ilyonectria spp. in tree decline affecting multi-generation apple orchards. Plant Soil 425, 217–230 (2018). https://doi.org/10.1007/s11104-018-3571-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-018-3571-3