Abstract
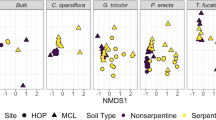
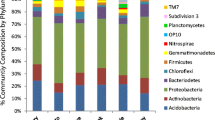
Soil chemistry is a known influence on plant species distribution. Serpentine soils provide a striking example of this due to their discrete nature and long-studied influence on plant communities. Characterized by high levels of heavy metals and low levels of nutrients, they present a challenge for most plant species and allow only a relatively restricted set of species to grow. We do not yet fully understand the suite of adaptations present in serpentine endemics allowing them thrive where other plant species perform poorly or not at all. In this paper we explore the possibility that serpentine plants interact with a unique set of microbial endophytes, which allow them to make a living on this challenging substrate. To examine broad-scale patterns of microbial community composition we used phospholipid fatty acid analysis. To focus more narrowly on arbuscular mycorrhizal fungi community composition we used 18S rDNA markers specific to these fungi. We found only very weak evidence for a relationship with distinct microbial communities using either technique and no evidence to show increased reliance on AMF by serpentine plants. Our results indicate that adaptation of plants to serpentine soil does not involve adaptation to a unique community of soil mutualists.




Similar content being viewed by others
References
Allison VJ, Miller RM, Jastrow JD, Matamala R, Zak DR (2005) Changes in soil microbial community structure in a tallgrass prairie chronosequence. Soil Sci Soc Am J 69:1412–1421
Amir H, Jasper DA, Abbott LK (2008) Tolerance and induction of tolerance to Ni of arbuscular mycorrhizal fungi from new Caledonian ultramafic soils. Mycorrhiza 19:1–6
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Canadian Journal of Biochemistry and Physiology 37:911–917
Brady KU, Kruckeberg AR, Bradshaw HD (2005) Evolutionary ecology of plant adaptation to serpentine soils. Annual Review of Ecology Evolution and Systematics 36:243–266
Brooks RR (1987) Serpentine and its vegetation: a multidisciplinary approach. Dioscorides, Portland, p 454
Brundrett MC (2009) Mycorrhizal associations and other means of nutrition of vascular plants: understanding the global diversity of host plants by resolving conflicting information and developing reliable means of diagnosis. Plant Soil 320:37–77
Casper BB, Castelli JP (2007) Evaluating plant-soil feedback together with competition in a serpentine grassland. Ecol Lett 10:394–400
Castelli JP, Casper BB (2003) Intraspecific AM fungal variation contributes to plant-fungal feedback in a serpentine grassland. Ecology 84:323–336
Clarke KR (1993) Nonparametric multivariate analyses of changes in community structure. Aust J Ecol 18:117–143
Clarke KR, Gorley RN (2001) PRIMER v5: user manual/tutorial. PRIMER-E, Plymouth
Coleman ML, Hedrick DB, Lovley DR, White DC, Pye K (1993) Reduction of Fe(III) in sediments by sulfate-reducing bacteria. Nature 361:436–438
Croll D, Wille L, Gamper HA, Mathimaran N, Lammers PJ, Corradi N, Sanders IR (2008) Genetic diversity and host plant preferences revealed by simple sequence repeat and mitochondrial markers in a population of the arbuscular mycorrhizal fungus Glomus intraradices. New Phytol 178:672–687
Fitzsimons MS, Miller RM, Jastrow JD (2008) Scale-dependent niche axes of arbuscular mycorrhizal fungi. Oecologia 158:117–127
Frostegard A, Baath E, Tunlid A (1993) Shifts in the structure of soil microbial communities in limed forests as revealed by phospholipid fatty-acid analysis. Soil Biol Biochem 25:723–730
Gildon A, Tinker PB (1981) A heavy metal-tolerant strain of a mycorrhizal fungus. Transactions of the British Mycological Society 77:648–649
Global Biodiversity Information Facility (2009) Occurence data for Avenula sulcata. Herbario de la Universidad de Salamanca, Real Jardin Botanico, Universidad de Oviedo, Universidad de Extremadura, Desarrollo Tecnológico e Innovación de la Junta de Extremadura, Institut Botanic de Barcelona, Inventaire national du Patrimoine naturel, Generalitat Valenciana, Universidad de Malaga. http://www.gbif.net. 21 Sept 2009.
Graham JH, Hodge NC, Morton JB (1995) Fatty-acid methyl-ester profiles for characterization of glomalean fungi and their endomycorrhizae. Appl Environ Microbiol 61:58–64
Hanski I (1998) Metapopulation dynamics. Nature 396:41–49
Hardoim PR, van Overbeek LS, van Elsas JD (2008) Properties of bacterial endophytes and their proposed role in plant growth. Trends Microbiol 16:463–471
Harrison S, Safford HD, Grace JB, Viers JH, Davies KF (2006) Regional and local species richness in an insular environment: serpentine plants in California. Ecol Monogr 76:41–56
Helgason T, Daniell TJ, Husband R, Fitter AH, Young JPW (1998) Ploughing up the wood-wide web? Nature 394:431–431
Hopkins NA (1987) Mycorrhizae in a California serpentine grassland community. Canadian Journal of Botany-Revue Canadienne De Botanique 65:484–487
Hurlbert SH (1971) Nonconcept of species diversity- critique and alternative parameters. Ecology 52:577–586
Jenny H (1980) The soil resource. Origin and behaviour. Ecol Stud 37:1–377
Johnson NC, Tilman D, Wedin D (1992) Plant and soil controls on mycorrhizal fungal communities. Ecology 73:2034–2042
Jongman RH, ter Braak CJF, Van Tongeren OFR (1995) Data analysis in community and landscape ecology. Cambridge University, Cambridge; New York, p 299
Koch AM, Kuhn G, Fontanillas P, Fumagalli L, Goudet J, Sanders IR (2004) High genetic variability and low local diversity in a population of arbuscular mycorrhizal fungi. Proc Natl Acad Sci USA 101:2369–2374
Kruckeberg AR (2002) Geology and plant life: the effects of landforms and rock types on plants. University of Washington, Seattle, p 362
Kruskal JB (1964a) Multidimensional scaling by optimizing goodness of fit to a nonmetric hypothesis. Psychometrika 29:1–27
Kruskal JB (1964b) Nonmetrick multidimensional scaling: a numerical method. Psychometrika 29:115–129
Larkin MA, Blackshields G, Brown NP, Chenna R, McGettigan PA, McWilliam H, Valentin F, Wallace IM, Wilm A, Lopez R, Thompson JD, Gibson TJ, Higgins DG (2007) Clustal W and clustal X version 2.0. Bioinformatics 23:2947–2948
Legendre P, Legendre L (1998) Numerical ecology. Elsevier, Amsterdam, p 853
Macarthur RH, Wilson EO (1963) Equilibrium- theory of insular zoogeography. Evolution 17:373–387
Maddison WP, Maddison DR (2007) Mesquite: a modular system for evolutionary analysis.
Menezes de Sequeira E, Pinto da Silva AR (1992) Ecology of serpentinized area of north-east Portugal. In: Roberts BA, Proctor J (eds) The ecology of areas with serpentinized rocks: a world view. Kluwer, Boston
Moser AM, Frank JL, D'Allura JA, Southworth D (2009) Ectomycorrhizal communities of Quercus garryana are similar on serpentine and nonserpentine soils. Plant Soil 315:185–194
Nylander JAA (2004) MrModeltest. Evolutionary Biology Centre, Uppsala
Okanen J, Kindt R and O'Hara RB (2005) Vegan: community ecology package.
Olsson PA, Baath E, Jakobsen I (1997) Phosphorus effects on the mycelium and storage structures of an arbuscular mycorrhizal fungus as studied in the soil and roots by analysis of fatty acid signatures. Appl Environ Microbiol 63:3531–3538
Olsson PA, Thingstrup I, Jakobsen I, Baath E (1999) Estimation of biomass of arbuscular mycorrhizal fungi in a linseed field. Soil Biol Biochem 31:1879–1887
Pal A, Dutta S, Mukherjee PK, Paul AK (2005) Occurrence of heavy metal-resistance in microflora from serpentine soil of Andaman. J Basic Microbiol 45:207–218
Pielou EC (1966) The measurement of diversity in different types of biological collections. J. Theor. Biol. 13:131–144
Posada D, Crandall KA (1998) MODELTEST: testing the model of DNA substitution. Bioinformatics 14:817–818
Proctor J (1999) Toxins, nutrient shortages and droughts: the serpentine challenge. Trends Ecol Evol 14:334–335
R Development Core Team (2006) R: a language and environment for statistical computing. Vienna, Austria.
Rodriguez RJ, White JF, Arnold AE, Redman RS (2009) Fungal endophytes: diversity and functional roles. New Phytol 182:314–330
Ronquist F, Huelsenbeck JP (2003) MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 19:1572–1574
Rosendahl S (2008) Communities, populations and individuals of arbuscular mycorrhizal fungi. New Phytol 178:253–266
Rosenzweig ML (1995) Species diversity in space and time. Cambridge University Press, Cambridge, p 436
Schechter SP, Bruns TD (2008) Serpentine and non-serpentine ecotypes of Collinsia sparsiflora associate with distinct arbuscular mycorrhizal fungal assemblages. Mol Ecol 17:3198–3210
Shannon CE, Weaver W (1963) The mathematical theory of communication. University of Illinois, Urbana, p 125
Simon L, Lalonde M, Bruns TD (1992) Specific Amplification of 18s Fungal Ribosomal Genes from Vesicular-Arbuscular Endomycorrhizal Fungi Colonizing Roots. Appl Environ Microbiol 58:291–295
Smith SE, Read DJ (2008) Mycorrhizal symbiosis. Academic, Boston, p 787
Vestal JR, White DC (1989) Lipid analysis in microbial ecology- Quantitative approaches to the study of microbial communities. Bioscience 39:535–541
Vivas A, Voros A, Biro B, Barea JM, Ruiz-Lozano JM, Azcon R (2003) Beneficial effects of indigenous Cd-tolerant and Cd-sensitive Glomus mosseae associated with a Cd-adapted strain of Brevibacillus sp in improving plant tolerance to Cd contamination. Applied Soil Ecology 24:177–186
Walker RB (1954) The ecology of serpentine soils. 2. Factors affecting plant growth on serpentine soils. Ecology 35:259–266
Wang B, Qiu YL (2006) Phylogenetic distribution and evolution of mycorrhizas in land plants. Mycorrhiza 16:299–363
Weissenhorn I, Leyval C, Berthelin J (1993) Cd-tolerant arbuscular mycorrhizal (AM) fungi from heavy-metal polluted soils. Plant Soil 157:247–256
Zak DR, Ringelberg DB, Pregitzer KS, Randlett DL, White DC, Curtis PS (1996) Soil microbial communities beneath Populus grandidentata crown under elevated atmospheric CO2. Ecol Appl 6:257–262
Zelles L (1999) Fatty acid patterns of phospholipids and lipopolysaccharides in the characterisation of microbial communities in soil: a review. Biol Fertil Soils 29:111–129
Zwickl DJ (2006) Genetic algorithm approaches for the phylogenetic analysis of large biological sequence datasets under the maximum likelihood criterion. University of Texas, Austin
Acknowledgements
We thank Sara Branco for all her help in navigating Portugal and motivating an interest in serpentine soils, Dr. Anabella Martins for providing housing and company in Bragança, and Dr. Carlos Aguiar for his help in identifying plants. We would also like to thank the Escola Superior Agraria de Braganca for allowing use of laboratory space and equipment. We would like to thank the Pritzker lab for Molecular Systematics and Evolution at the Field Museum (Chicago, IL) for space and resources to complete the project. We thank Drs. Peter Avis and Kevin Feldheim for generating the Taq DNA polymerase. We thank Zhanna Yermakov and Cheryl Krol for their helping with PLFA analysis. We thank Tim Wootton for help in manuscript preparation. Finally, we thank the Garden Club of America of America Fellowship in Ecological Restoration and the University of Chicago Hinds Fund for Evolutionary Biology for funding. RMM’s participation was funded by the U.S. Department of Energy, Office of Science, Office of Biological and Environmental Research, Climate Change Research Division under contract DE-AC02-06CH11357.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Angela Hodge.
Appendix
Appendix
Rights and permissions
About this article
Cite this article
Fitzsimons, M.S., Miller, R.M. Serpentine soil has little influence on the root-associated microbial community composition of the serpentine tolerant grass species Avenula sulcata . Plant Soil 330, 393–405 (2010). https://doi.org/10.1007/s11104-009-0213-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-009-0213-9