Abstract
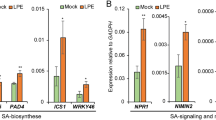
The hypersensitive response (HR) is a powerful resistance system that plants have developed against pathogen attack. There are two major pathways for HR induction; one is through recognition of the pathogen by a specific host protein, and is known as the host HR. The other is through common biochemical changes upon infection—the nonhost HR. We previously demonstrated that hydrogen peroxide derived from polyamine degradation by polyamine oxidase triggers the typical host HR in tobacco plants upon infection with tobacco mosaic virus. However, it remains to be determined whether or not polyamines are involved in the nonhost HR in tobacco, and in the host HR in other plant species. When tobacco plants were infected with Pseudomonas cichorii, a representative nonhost pathogen, transcripts for six genes encoding enzymes for polyamine metabolism were simultaneously induced, and polyamines were accumulated in apoplasts. Hydrogen peroxide was concomitantly produced and hypersensitive cell death occurred at infected sites. Silencing of polyamine oxidase by the virus-induced gene silencing method resulted in suppression of hydrogen peroxide production and in disappearance of visible hypersensitive cell death with an increase in bacterial growth. Our results indicated that polyamines served as the source of hydrogen peroxide during the nonhost HR in tobacco plants. Further analysis revealed that polyamines were accumulated in apoplasts of Arabidopsis thaliana infected with Pseudomonas syringae, and of rice infected with Magnaporthe grisea, both causing the typical host HR. As in tobacco, it is conceivable that the same mechanism operates for nonhost HR in these plants. Our present observations thus suggested that polyamines are commonly utilized as the source of hydrogen peroxide during host- and nonhost HRs in higher plants.






Similar content being viewed by others
Abbreviations
- ADC:
-
Arginine decarboxylase
- DAB:
-
3,3′-diaminobenzidine
- HR:
-
Hypersensitive response
- Nb:
-
Nicotiana benthamiana
- Nt:
-
Nicotiana tabacum
- ODC:
-
Ornithine decarboxylase
- PAO:
-
Polyamine oxidase
- Put:
-
Putrescine
- SAMDC:
-
S-Adenosylmethionine decarboxylase
- Spd:
-
Spermidine
- SPDS:
-
Spermidine synthase
- Spm:
-
Spermine
- SPMS:
-
Spermine synthase
- TMV:
-
Tobacco mosaic virus
- TRV:
-
Tobacco rattle virus
- VIGS:
-
Virus-induced gene silencing
References
Bestwick CS, Brown IR, Mansfield JW (1998) Localized changes in peroxidase activity accompany hydrogen peroxide generation during the development of a nonhost hypersensitive reaction in lettuce. Plant Physiol 118:1067–1078. doi:10.1104/pp.118.3.1067
Cervelli M, Di Caro O, Di Penta A, Angelini R, Federico R, Vitale A, Mariottini P (2004) A novel C-terminal sequence from barley polyamine oxidase is a vacuolar sorting signal. Plant J 40:410–418. doi:10.1111/j.1365-313X.2004.02221.x
Chomczynski P, Sacchi N (1987) Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal Biochem 162:156–159. doi:10.1016/0003-2697(87)90021-2
Cona A, Rea G, Angelini R, Federico R, Tavladorki P (2006) Functions of amine oxidase in plant development and defense. Trends Plant Sci 11:80–88. doi:10.1016/j.tplants.2005.12.009
Cowley T, Walters DR (2002) Polyamine metabolism in barley reacting hypersensitively to the powdery mildew fungus Blumeria graminis f. sp. hordei. Plant Cell Environ 25:461–468. doi:10.1046/j.0016-8025.2001.00819.x
Groppa MD, Benavides MP (2008) Polyamines and abiotic stress: recent advances. Amino Acids 34:35–45. doi:10.1007/s00726-007-0501-8
Hammond-Kosack KE, Jones JD (1997) Plant disease resistance genes. Annu Rev Plant Physiol Plant Mol Biol 48:575–607. doi:10.1146/annurev.arplant.48.1.575
Heath MC (2000) Nonhost resistance and nonspecific plant defenses. Curr Opin Plant Biol 3:315–319. doi:10.1016/S1369-5266(00)00087-X
Huckelhoven R, Dechert C, Kogel KH (2001) Non-host resistance of barley is associated with a hydrogen peroxide burst at sites of attempted penetration by wheat powdery mildew fungus. Mol Plant Pathol 2:199–205. doi:10.1046/j.1464-6722.2001.00067.x
Imanishi S, Hashizume K, Nakakita M, Kojima H, Matsubayashi Y, Hashimoto T, Sakagami Y, Yamada Y, Nakamura K (1998) Differential induction by methyl jasmonate of genes encoding ornithine decarboxylase and other enzymes involved in nicotine biosynthesis in tobacco cell cultures. Plant Mol Biol 38:1101–1111. doi:10.1023/A:1006058700949
Iwai T, Miyasaka A, Seo S, Ohashi Y (2006) Contribution of ethylene biosynthesis for resistance to blast fungus infection in young rice plants. Plant Physiol 142:1202–1215. doi:10.1104/pp.106.085258
Kamada-Nobusada T, Hayashi M, Fukazawa M, Sakakibara H, Nishimura M (2008) A putative peroxisomal polyamine oxidase, AtPAO4, is involved in polyamine catabolism in Arabidopsis thaliana. Plant Cell Physiol 49:1272–1282. doi:10.1093/pcp/pcn114
Keen NT (1990) Gene-for-gene complementarity in plant–pathogen interactions. Annu Rev Genet 24:447–463. doi:10.1146/annurev.ge.24.120190.002311
Knoester M, van Loon LC, van den Heuvel J, Hennig J, Bol JF, Linthorst HJ (1998) Ethylene-insensitive tobacco lacks nonhost resistance against soil-borne fungi. Proc Natl Acad Sci USA 95:1933–1937. doi:10.1073/pnas.95.4.1933
Mellersh DG, Heath MC (2003) An investigation into the involvement of defense signaling pathways in components of the nonhost resistance of Arabidopsis thaliana to rust fungi also reveals a model system for studying rust fungal compatibility. Mol Plant Microbe Interact 16:398–404. doi:10.1094/MPMI.2003.16.5.398
Moschou PN, Sanmartin M, Andriopoulou AH, Rojo E, Sanchez-Serrano JJ, Roubelakis-Angelakis KA (2008) Bridging the Gap between plant and mammalian polyamine catabolism: a novel peroxisomal polyamine oxidase responsible for a full back-conversion pathway in Arabidopsis thaliana. Plant Physiol 147:1845–1857. doi:10.1104/pp.108.123802
Murray MG, Thompson WF (1980) Rapid isolation of high molecular weight plant DNA. Nucleic Acids Res 8:4321–4325. doi:10.1093/nar/8.19.4321
Mysore KS, Ryu CM (2004) Nonhost resistance: how much do we know? Trends Plant Sci 9:97–104. doi:10.1016/j.tplants.2003.12.005
Papadakis AK, Roubelakis-Angelakis KA (2005) Polyamines inhibit NADPH oxidase-mediated superoxide generation and putrescine prevents programmed cell death induced by polyamine oxidase-generated hydrogen peroxide. Planta 220:826–837. doi:10.1007/s00425-004-1400-9
Peart JR, Lu R, Sadanandom A, Malcuit I, Moffett P, Brice DC, Schauser L, Jaggard DA, Xiao S, Coleman MJ, Dow M, Jones JD, Shirasu K, Baulcombe DC (2002) Ubiquitin ligase-associated protein SGT1 is required for host and nonhost disease resistance in plants. Proc Natl Acad Sci USA 99:10865–10869. doi:10.1073/pnas.152330599
Ratcliff F, Martin-Hernandez AM, Baulcombe DC (2001) Technical advance. Tobacco rattle virus as a vector for analysis of gene function by silencing. Plant J 25:237–245. doi:10.1046/j.0960-7412.2000.00942.x
Sharma PC, Ito A, Shimizu T, Terauchi R, Kamoun S, Saitoh H (2003) Virus-induced silencing of WIPK and SIPK genes reduces resistance to a bacterial pathogen, but has no effect on the INF1-induced hypersensitive response (HR) in Nicotiana benthamiana. Mol Genet Genomics 269:583–591. doi:10.1007/s00438-003-0872-9
Soga T, Heiger DN (2000) Amino acid analysis by capillary electrophoresis electrospray ionization mass spectrometry. Anal Chem 72:1236–1241. doi:10.1021/ac990976y
Staskawicz BJ, Ausubel FM, Baker BJ, Ellis JG, Jones JD (1995) Molecular genetics of plant disease resistance. Science 268:661–667. doi:10.1126/science.7732374
Walden R, Cordeiro A, Tiburcio AF (1997) Polyamines: small molecules triggering pathways in plant growth and development. Plant Physiol 113:1009–1013. doi:10.1104/pp.113.4.1009
Walters DR (2000) Polyamines in plant–microbe interactions. Physiol Mol Plant Pathol 57:137–146. doi:10.1006/pmpp.2000.0286
Walters D, Cowley T, Mitchell A (2002) Methyl jasmonate alters polyamine metabolism and induces systemic protection against powdery mildew infection in barley seedlings. J Exp Bot 53:747–756. doi:10.1093/jexbot/53.369.747
Yamakawa H, Kamada H, Satoh M, Ohashi Y (1998) Spermine is a salicylate-independent endogenous inducer for both tobacco acidic pathogenesis-related proteins and resistance against tobacco mosaic virus infection. Plant Physiol 118:1213–1222. doi:10.1104/pp.118.4.1213
Yoda H, Ogawa M, Yamaguchi Y, Koizumi N, Kusano T, Sano H (2002) Identification of early-responsive genes associated with the hypersensitive response to tobacco mosaic virus and characterization of a WRKY-type transcription factor in tobacco plants. Mol Genet Genomics 267:154–161. doi:10.1007/s00438-002-0651-z
Yoda H, Yamaguchi Y, Sano H (2003) Induction of hypersensitive cell death by hydrogen peroxide produced through polyamine degradation in tobacco plants. Plant Physiol 132:1973–1981. doi:10.1104/pp.103.024737
Yoda H, Hiroi Y, Sano H (2006) Polyamine oxidase is one of the key elements for oxidative burst to induce programmed cell death in tobacco cultured cells. Plant Physiol 142:193–206. doi:10.1104/pp.106.080515
Acknowledgements
The authors thank David C. Baulcombe (John Innes Centre) and Hirofumi Yoshioka (Nagoya University) for generous provision of A. tumefaciens GV3101 strain and C58C1 strain containing pBINTRA6, and TRV vector; Jeffery L. Dangl (University of North Carolina) for P. syringae DC3000 (avrRpm1); and Tsutomu Kawasaki (Nara Institute of Science and Technology) for M. grisea (race 031).
Author information
Authors and Affiliations
Corresponding author
Additional information
Hiroshi Yoda and Kazuki Fujimura have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Yoda, H., Fujimura, K., Takahashi, H. et al. Polyamines as a common source of hydrogen peroxide in host- and nonhost hypersensitive response during pathogen infection. Plant Mol Biol 70, 103–112 (2009). https://doi.org/10.1007/s11103-009-9459-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11103-009-9459-0