Abstract
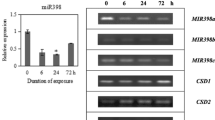
MicroRNAs (miRNAs) are ∼21-nt RNAs that reduce target accumulation through mRNA cleavage or translational repression. Arabidopsis miR398 regulates mRNAs encoding two copper superoxide dismutase (CSD) enzymes and a cytochrome c oxidase subunit. miR398 itself is down-regulated in response to copper and stress. Here we show that miR398 is positively regulated by sucrose, resulting in decreased CSD1 and CSD2 mRNA and protein accumulation. This sucrose regulation is maintained both in the presence and absence of physiologically relevant levels of supplemental copper. Additionally, we show that plants expressing CSD1 and CSD2 mRNAs with altered miR398 complementarity sites display increased mRNA accumulation, whereas CSD1 and CSD2 protein accumulation remain sensitive to miR398 levels, suggesting that miR398 can act as a translational repressor when target site complementarity is reduced. These results reveal a novel miR398 regulatory mechanism and demonstrate that plant miRNA targets can resist miRNA regulation at the mRNA level while maintaining sensitivity at the level of protein accumulation. Our results suggest that even in plants, where miRNAs are thought to act primarily through target mRNA cleavage, monitoring target protein levels along with target mRNA levels is necessary to fully assess the consequences of disrupted miRNA–mRNA pairing. Moreover, the limited complementarity required to maintain robust miR398-directed repression of target protein accumulation suggests that similarly regulated endogenous plant miRNA targets may have eluded detection.











Similar content being viewed by others
Abbreviations
- CSD:
-
Copper superoxide dismutase
- FSD:
-
Iron superoxide dismutase
- miRNA:
-
MicroRNA
- PN:
-
Plant nutrient medium
- PN−m :
-
Plant nutrient medium without micronutrients
- ROS:
-
Reactive oxygen species
- SOD:
-
Superoxide dismutase
References
Abdel-Ghany SE, Muller-Moule P, Niyogi KK, Pilon M, Shikanai T (2005) Two P-type ATPases are required for copper delivery in Arabidopsis thaliana chloroplasts. Plant Cell 17:1233–1251
Alonso JM, Stepanova AN, Leisse TJ, Kim CJ, Chen H, Shinn P, Stevenson DK, Zimmerman J, Barajas P, Cheuk R, Gadrinab C, Heller C, Jeske A, Koesema E, Meyers CC, Parker H, Prednis L, Ansari Y, Choy N, Deen H, Geralt M, Hazari N, Hom E, Karnes M, Mulholland C, Ndubaku R, Schmidt I, Guzman P, Aguilar-Henonin L, Schmid M, Weigel D, Carter DE, Marchand T, Risseeuw E, Brogden D, Zeko A, Crosby WL, Berry CC, Ecker JR (2003) Genome-wide insertional mutagenesis of Arabidopsis thaliana. Science 301:653–657
Aukerman MJ, Sakai H (2003) Regulation of flowering time and floral organ identity by a MicroRNA and its APETALA2-like target genes. Plant Cell 15:2730–2741
Ausubel FM, Brent R, Kingston RE, Moore DD, Seidman JG, Smith JA, Struhl K (eds) (1995) Short protocols in molecular biology. John Wiley & Sons, New York
Axtell MJ, Bartel DP (2005) Antiquity of microRNAs and their targets in land plants. Plant Cell 17:1658–1673
Axtell MJ, Jan C, Rajagopalan R, Bartel DP (2006) A two-hit trigger for siRNA biogenesis in plants. Cell 127:565–577
Axtell MJ, Snyder JA, Bartel DP (2007) Common functions for diverse small RNAs of land plants. Plant Cell 19:1750–1769
Bartel DP (2004) MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 116:281–297
Bartel B, Fink GR (1994) Differential regulation of an auxin-producing nitrilase gene family in Arabidopsis thaliana. Proc Natl Acad Sci USA 91:6649–6653
Bevan M (1984) Binary Agrobacterium vectors for plant transformation. Nucleic Acids Res 12:8711–8721
Bonnet E, Wuyts J, Rouze P, Van de Peer Y (2004) Detection of 91 potential conserved plant microRNAs in Arabidopsis thaliana and Oryza sativa identifies important target genes. Proc Natl Acad Sci USA 101:11511–11516
Celenza JL, Grisafi PL, Fink GR (1995) A pathway for lateral root formation in Arabidopsis thaliana. Genes Dev 9:2131–2142
Chen X (2004) A microRNA as a translational repressor of APETALA2 in Arabidopsis flower development. Science 303:2022–2025
Clough SJ, Bent AF (1998) Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 16:735–743
Couee I, Sulmon C, Gouesbet G, El Amrani A (2006) Involvement of soluble sugars in reactive oxygen species balance and responses to oxidative stress in plants. J Exp Bot 57:449–459
Fattash I, Voss B, Reski R, Hess WR, Frank W (2007) Evidence for the rapid expansion of microRNA-mediated regulation in early land plant evolution. BMC Plant Biol 7:13
Haughn GW, Somerville C (1986) Sulfonylurea-resistant mutants of Arabidopsis thaliana. Mol Gen Genet 204:430–434
Jefferson RA (1989) The GUS reporter gene system. Nature 342:837–838
Jones-Rhoades MW, Bartel DP (2004) Computational identification of plant microRNAs and their targets, including a stress-induced miRNA. Mol Cell 14:787–799
Jones-Rhoades MW, Bartel DP, Bartel B (2006) MicroRNAs and their regulatory roles in plants. Annu Rev Plant Biol 57:19–53
Kliebenstein DJ, Monde RA, Last RL (1998) Superoxide dismutase in Arabidopsis: an eclectic enzyme family with disparate regulation and protein localization. Plant Physiol 118:637–650
Koncz C, Nemeth K, Redei GP, Schell J (1992) T-DNA insertional mutagenesis in Arabidopsis. Plant Mol Biol 20:963–976
LeClere S, Bartel B (2001) A library of Arabidopsis 35S-cDNA lines for identifying novel mutants. Plant Mol Biol 46:695–703
Megraw M, Baev V, Rusinov V, Jensen ST, Kalantidis K, Hatzigeorgiou AG (2006) MicroRNA promoter element discovery in Arabidopsis. RNA 12:1612–1619
Monroe-Augustus M, Zolman BK, Bartel B (2003) IBR5, a dual-specificity phosphatase-like protein modulating auxin and abscisic acid responsiveness in Arabidopsis. Plant Cell 15:2979–2991
Rajagopalan R, Vaucheret H, Trejo J, Bartel DP (2006) A diverse and evolutionarily fluid set of microRNAs in Arabidopsis thaliana. Genes Dev 20:3407–3425
Rizhsky L, Liang H, Mittler R (2003) The water-water cycle is essential for chloroplast protection in the absence of stress. J Biol Chem 278:38921–38925
Roitsch T (1999) Source-sink regulation by sugar and stress. Curr Opin Plant Biol 2:198–206
Schwab R, Palatnik JF, Riester M, Schommer C, Schmid M, Weigel D (2005) Specific effects of microRNAs on the plant transcriptome. Dev Cell 8:517–527
Seki M, Narusaka M, Kamiya A, Ishida J, Satou M, Sakurai T, Nakajima M, Enju A, Akiyama K, Oono Y, Muramatsu M, Hayashizaki Y, Kawai J, Carninci P, Itoh M, Ishii Y, Arakawa T, Shibata K, Shinagawa A, Shinozaki K (2002) Functional annotation of a full-length Arabidopsis cDNA collection. Science 296:141–145
Sunkar R, Zhu JK (2004) Novel and stress-regulated microRNAs and other small RNAs from Arabidopsis. Plant Cell 16:2001–2019
Sunkar R, Kapoor A, Zhu JK (2006) Posttranscriptional induction of two Cu/Zn superoxide dismutase genes in Arabidopsis is mediated by downregulation of miR398 and important for oxidative stress tolerance. Plant Cell 18:2051–2065
Tang G, Reinhart BJ, Bartel DP, Zamore PD (2003) A biochemical framework for RNA silencing in plants. Genes Dev 17:49–63
Vaucheret H (2006) Post-transcriptional small RNA pathways in plants: mechanisms and regulations. Genes Dev 20:759–771
Welchen E, Chan RL, Gonzalez DH (2004) The promoter of the Arabidopsis nuclear gene COX5b-1, encoding subunit 5b of the mitochondrial cytochrome c oxidase, directs tissue-specific expression by a combination of positive and negative regulatory elements. J Exp Bot 55:1997–2004
Xie Z, Allen E, Fahlgren N, Calamar A, Givan SA, Carrington JC (2005) Expression of Arabidopsis MIRNA genes. Plant Physiol 138:2145–2154
Yamasaki H, Abdel-Ghany SE, Cohu CM, Kobayashi Y, Shikanai T, Pilon M (2007) Regulation of copper homeostasis by micro-RNA in Arabidopsis. J Biol Chem 282:16369–16378
Acknowledgements
We thank Daniel Kliebenstein for providing the anti-CSD1, anti-CSD2, and anti-FSD antibodies, the Salk Institute Genomic Analysis Laboratory for generating the sequence-indexed T-DNA insertion mutants, and the Arabidopsis Biological Resource Center at Ohio State University for seeds, cDNA clones, and BAC clones. We thank Matthew Lingard, Lucia Strader, and Andrew Woodward for critical comments on the manuscript, and David Bartel for helpful discussions. This research was supported by the National Institutes of Health (R24-GM069512), the G. Harold and Leila Y. Mathers Charitable Foundation, and the Robert A. Welch Foundation (C-1309). D.V.D. was supported in part by a training grant from the National Institutes of Health (T32-GM08362).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dugas, D.V., Bartel, B. Sucrose induction of Arabidopsis miR398 represses two Cu/Zn superoxide dismutases. Plant Mol Biol 67, 403–417 (2008). https://doi.org/10.1007/s11103-008-9329-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11103-008-9329-1