Abstract
Context
Transsphenoidal surgery (TSS) to resect a pituitary adenoma is considered first-line treatment for patients with Cushing’s disease (CD). Early, post-operative remission rates >80% are expected for patients with a microadenoma (≤ 10 mm) visible on magnetic resonance (MR) imaging.
Objective
To report surgical outcomes and predictors of remission in a specialist center for patients with CD.
Patients and methods
Clinical data was obtained from a prospective CD database in addition to review of all electronic medical, laboratory and surgical patient records. Patients who underwent their first TSS by one neurosurgeon between 2004 and 2013, and had a minimum 1 year follow up, were evaluated.
Results
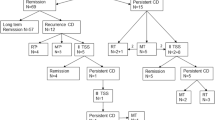
One hundred and one consecutive patients with CD (73F, 28M) underwent TSS. Median (range) age and follow-up were 47 (15–87) and 4.33 (1–9.8) years, respectively. At surgery, 74 (73.2%) patients had a microadenoma, 27 a macroadenoma; six of the latter patients had a planned, subtotal resection to control neurological signs due to mass effect. Initial remission rates were: microadenoma, 89% (66/74); macroadenoma, 63% (17/27); and 81% (17/21) in those macroadenomas where complete surgical removal was anticipated. Initial non-remission occurred in 18 patients, ten macro- and eight microadenoma; six of 18 had residual disease on most recent follow up. Six (2 macro, 4 micro) of the 83 patients with initial remission have had late (>12 months) recurrence of hypercortisolism that required either repeat TSS or adjunctive therapy, three of whom have persistent hypercortisolism. Macroadenoma (p = 0.003) and tumor invasion beyond the pituitary and sella (p < 0.001) were associated with failure to obtain remission with the initial TSS and greater likelihood of late recurrence. Patients in whom no lesion was seen on neuroimaging had rates of initial remission (21/25 or 84%) and a similar late recurrence rate of 4% (1/25) in comparison with those with MR-visible microadenomas (3/49, or 6%).
Conclusions
A team-based approach, in a specialized pituitary center, can lead to initial and durable, long-term remission in patients with CD. The presence of a macroadenoma and tumor extension beyond the pituitary and sella were predictive of initial non-remission as well as risk of late recurrence.

Similar content being viewed by others
Abbreviations
- TSS:
-
Transsphenoidal tumor resection
- ACTH:
-
Adrenocorticotropic hormone
- UFC:
-
Urinary free cortisol
- CD:
-
Cushing’s disease
- CS:
-
Cushing’s syndrome
- IPSS:
-
Inferior petrosal sinus sampling
- ULN:
-
Upper limit of normal
References
Steffensen C, Bak AM, Rubeck KZ, Jørgensen JO (2010) Epidemiology of Cushing’s syndrome. Neuroendocrinology 92(Suppl 1):1–5
Pivonello R, De Leo M, Cozzolino A, Colao A (2015) The treatment of Cushing’s disease. Endocr Rev 36:385–486
Lindholm J, Juul S, Jørgensen JO, Astrup J, Bjerre P, Feldt-Rasmussen U, Hagen C, Jørgensen J, Kosteljanetz M, Kristensen L, Laurberg P, Schmidt K, Weeke J (2001) Incidence and late prognosis of Cushing’s syndrome: a population-based study. J Clin Endocrinol Metab 86:117–123
Swearingen B, Biller BM, Barker FG 2nd, Katznelson L, Grinspoon S, Klibanski A, Zervas NT (1999) Long-term mortality after transsphenoidal surgery for Cushing disease. Ann Intern Med 130:821–824
Clayton RN, Raskauskiene D, Reulen RC, Jones PW (2011) Mortality and morbidity in Cushing’s disease over 50 years in Stoke-on-Trent, UK: audit and meta-analysis of literature. J Clin Endocrinol Metab 96:632–642
Lambert JK, Goldberg L, Fayngold S, Kostadinov J, Post KD, Geer EB (2013) Predictors of mortality and long-term outcomes in treated Cushing’s disease: a study of 346 patients. J Clin Endocrinol Metab 98:1022–1030
Hammer GD, Tyrrell JB, Lamborn KR, Applebury CB, Hannegan ET, Bell S, Rahl R, Lu A, Wilson CB (2004) Transsphenoidal microsurgery for Cushing’s disease: initial outcome and long-term results. J Clin Endocrinol Metab 89:6348–6357
Atkinson AB, Kennedy A, Wiggam MI, McCance DR, Sheridan B (2005) Long-term remission rates after pituitary surgery for Cushing’s disease: the need for long-term surveillance. Clin Endocrinol 63:549–559
Lonser RR, Nieman L, Oldfield EH (2017) Cushing’s disease: pathobiology, diagnosis, and management. J Neurosurg 126:404–417
Hameed N, Yedinak CG, Brzana J, Gultekin SH, Coppa ND, Dogan A, Delashaw JB, Fleseriu M (2013) Remission rate after transsphenoidal surgery in patients with pathologically confirmed Cushing’s disease, the role of cortisol, ACTH assessment and immediate reoperation: a large single center experience. Pituitary 16:452–458
Ciric I, Zhao JC, Du H, Findling JW, Molitch ME, Weiss RE, Refetoff S, Kerr WD, Meyer J (2012) Transsphenoidal surgery for Cushing’s disease: experience with 136 patients. Neurosurgery 70:70–81
Hassan-Smith ZK, Sherlock M, Reulen RC, Arlt W, Ayuk J, Toogood AA, Cooper MS, Johnson AP, Stewart PM (2012) Outcome of Cushing’s disease following transsphenoidal surgery in a single center over 20 years. J Clin Endocrinol Metab 97:1194–1201
Valassi E, Biller BM, Swearingen B, Giraldi FP, Losa M, Mortini P, Hayden D, Cavagnini F, Klibanski A (2010) Delayed remission after transsphenoidal surgery in patients with Cushing’s disease. J Clin Endocrinol Metab 95:601–610
Ram Z, Nieman LK, Cutler GB Jr, Chrousos GP, Doppman JL, Oldfield EH (1994) Early repeat surgery for persistent Cushing’s disease. J Neursurg 80:37–45
Patil CG, Veeravaqu A, Prevedello DM, Katznelson L, Vance ML, Laws ER Jr (2008) Outcomes after repeat transsphenoidal surgery for recurrent Cushing’s disease. Neurosurgery 63:266–270
Lindsay JR, Oldfield EH, Stratakis CA, Nieman LK (2011) The postoperative basal cortisol and CRH tests for prediction of long-term remission from Cushing’s disease after transsphenoidal surgery. J Clin Endocrinol Metab 96:2057–2064
Nieman LK, Biller BMK, Findling JW, Hassan Murad M, Newell-Price J, Savage MO, Tabarin A (2015) Treatment of Cushing’s syndrome: an Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab 100:2807–2831
Zerikly RK, Amiri L, Faiman C, Gupta M, Singh RJ, Nutter B, Kennedy L, Hatipoglu B, Weil RJ, Hamrahian AH (2010) Diagnostic characteristics of late-night salivary cortisol using liquid chromatography-tandem mass spectrometry. J Clin Endocrinol Metab 95:4555–4559
Mulligan GB, Eray E, Faiman C, Gupta M, Pineyro MM, Makdissi A, Suh JH, Masaryk TJ, Prayson R, Weil RJ, Hamrahian AH (2011) Reduction of false-negative results in inferior petrosal sinus sampling with simultaneous prolactin and corticotropin measurement. Endocr Pract 17:33–40
Mulligan GB, Faiman C, Gupta M, Kennedy L, Hatipoglu B, Hui F, Weil RJ, Hamrahian AH (2012) Prolactin measurement during inferior petrosal sinus sampling improves the localization of pituitary adenomas in Cushing’s disease. Clin Endocrinol 77:268–274
Patronas N, Bulakbasi N, Stratakis CA, Lafferty A, Oldfield EH, Doppman J, Nieman LK (2003) Spoiled gradient recalled acquisition in the steady state technique is superior to conventional post contrast spin echo technique for magnetic resonance imagining detection of adrenocortictropin-secreting pituitary tumors. J Clin Endocrinol Metab 88:1565–1569
Yogi-Morren D, Habra MA, Faiman C, Bena J, Hatipoglu B, Kennedy L, Weil RJ, Hamrahian AH (2015) Pituitary MRI findings in patients with pituitary and ectopic ACTH-dependent cushing syndrome: does a 6-mm pituitary tumor size cut-off value exclude ectopic ACTH-syndrome. Endocr Pract 21:1098–1103
Oldfield EH, Vortmeyer AO (2006) Development of a histological pseudocapsule and its use as a surgical capsule in the excision of pituitary tumors. J Neurosurg 104:7–19
Jagannathan J, Smith R, DeVroom HL, Vortmeyer AO, Stratakis CA, Nieman LK, Oldfield EH (2009) Outcome of using the histological pseudocapsule as a surgical capsule in Cushing disease. J Neurosurg 111:531–539
Di Ieva A, Rotondo F, Syro LV, Cusimano MD, Kovacs K (2014) Aggressive pituitary adenomas-diagnosis and emerging treatments. Nat Rev Endocrinol 10:423–435
Rees DA, Hanna FW, Davies JS, Mills RG, Vafidis J, Scanlon MF (2002) Long-term follow-up results of transsphenoidal surgery for Cushing’s disease in a single center using strict criteria for remission. Clin Endocrinol 56:541–551
Newell-Price J, Bertagna X, Grossman AB, Nieman LK (2006) Cushing’s syndrome. Lancet 367:1605–1617
Sharma ST, Nieman LK, Feelders RA (2015) Cushing’s syndrome: epidemiology and developments in disease management. Clin Epidemiol 7:281–293
Woo YS, Isidori AM, Wat WZ, Kaltsas GA, Afshar F, Sabin I, Jenkins PJ, Monson JP, Besser GM, Grossman AB (2005) Clinical and biochemical characteristics of adrenocorticotropin-secreting macroadenomas. J Clin Endocrinol Metab 90:4963–4969
Johnston PC, Hamrahian AH, Weil RJ (2014) Cushing’s disease from an ectopic parasellar adenoma. Postgrad Med J 90:360–361
Johnston PC, Kennedy L, Weil RJ, Hamrahian AH (2014) Ectopic ACTH-secreting pituitary adenomas within the sphenoid sinus. Endocrine 47:717–724
Toms GC, McCarthy MI, Niven MJ, Orteu CH, King TT, Monson JP (1993) Predicting relapse after transsphenoidal surgery for Cushing’s disease. J Clin Endocrinol Metab 76:291–294
Martino J, Acebes JJ, Montanya E, Masuet C, Brell M (2007) Analysis of early prognostic factors for risk of treatment failure in Cushing’s disease treated by trans-sphenoidal pituitary surgery. Med Clin 128:330–332
Chee GH, Mathias DB, James RA, Kendall-Taylor P (2001) Transsphenoidal pituitary surgery in Cushing’s disease can we predict outcome?. Clin Endocrinol 54:617–626
Yap LB, Turner HE, Adams CB, Wass JA (2002) Undetectable postoperative cortisol does not always predict long-term remission in Cushing’s disease: a single center audit. Clin Endocrinol 56:25–31
Sherlock M, Ayuk J, Tomlinson JW, Toogood AA, Aragon-Alonso A, Sheppard MC, Bates AS, Stewart PM (2010) Mortality in patients with pituitary disease. Endocr Rev 31:301–342
Etaxbe J, Vazquez JA (1994) Morbidity and mortality in cushing’s disease: an epidemiological approach. Clin Endocrinol 4:479–484
Van Zaane B, Nur E, Squizzato A, Dekkers OM, Twickler MT, Fliers E, Gerdes VE, Büller HR, Brandjes DP (2009) Hypercoagulable state in Cushing’s syndrome: a systematic review. J Clin Endocrinol Metab 94(8):2743–2750
Stuijver DJ, van Zaane B, Feelders RA, Debeij J, Cannegieter SC, Hermus AR, van den Berg G, Pereira AM, de Herder WW, Wagenmakers MA, Kerstens MN, Zelissen PM, Fliers E, Schaper N, Drent ML, Dekkers OM, Gerdes VE (2011) Incidence of venous thromboembolism in patients with Cushing’s syndrome: a multicenter cohort study. J Clin Endocrinol Metab 96:3525–3532
Van der Pas R, de Bruin C, Leebeek FW, de Maat MP, Rijken DC, Pereira AM, Romijn JA, Netea-Maier RT, Hermus AR, Zelissen PM, de Jong FH, van der Lely AJ, de Herder WW, Lamberts SW, Hofland LJ, Feelders RA (2012) The hypercoagulable state in Cushing’s disease is associated with increased levels of procoagulant factors and impaired fibrinolysis, but is not reversible after short-term biochemical remission induced by medical therapy. J Clin Endocrinol Metab 97:1303–1310
Colao A, Pivonello R, Spiezia S, Faggiano A, Ferone D, Filippella M, Marzullo P, Cerbone G, Siciliani M, Lombardi G (1999) Persistence of increased cardiovascular risk in patients with Cushing’s disease after five years of successful cure. J Clin Endocrinol Metab 84:2664–2672
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors on this manuscript have declared that they have no conflict of interest.
Disclosure
All authors of this manuscript have no conflicts of interest to disclose Acknowledgments: We thank the physician staff at Cleveland Clinic including Dr Leann Olanksy who referred patients for surgery.
Ethical approval
Informed consent was obtained from all individual participants included in the study. This article does not contain any studies with human participants or animals performed by any of the authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Johnston, P.C., Kennedy, L., Hamrahian, A.H. et al. Surgical outcomes in patients with Cushing’s disease: the Cleveland clinic experience. Pituitary 20, 430–440 (2017). https://doi.org/10.1007/s11102-017-0802-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11102-017-0802-1