Abstract
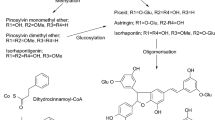
Hydroxycinnamates are found associated with suberin in several forms: covalently linked to the aliphatic suberin; in the residue after suberin-removal; and in the non-polar extractives of suberized tissues in the form of alkyl ferulates. Suberin-associated hydroxycinnamates have been found mainly as ferulic acid-derivatives, sometimes as feruloylamides and in a lesser extent as caffeates. Ferulic acid esters of long-chain ω-hydroxyacids are prevalent in the partial depolymerisation products of suberin. Also, enzymes able to catalyze the feruloylation of ω-hydroxyacids were found timely-associated with the suberization process. It is proposed that ferulic acid, and its dimers, through esterification to ω-hydroxyacids, covalently link the suberin aliphatic polyester to suberin-associated polyaromatics. In this case, the known role of ferulates, and related hydroxycinnamates, as cross-linkers of structurally different polymers would be enlarged to suberized cell-walls.

Similar content being viewed by others
References
Adamovics JA, Johnson G, Stermitz FR (1977) Ferulates from cork layers of Solanum tuberosum and Pseudotsuga menziesii. Phytochemistry 16:1089–1090
Agullo C, Seoane E (1982) Hydrogenolysis of cork suberin by LIBH4—free hydroxyl groups. An Quim 78C:389–393
Arrieta-Baez D, Stark RE (2006) Modeling suberization with peroxidase-catalyzed polymerization of hydroxycinnamic acids: cross-coupling and dimerization reactions. Phytochemistry 67:743–753. doi:10.1016/j.phytochem.2006.01.026
Bento MF, Pereira H, Cunha MA, Moutinho AMC, van den Berg KJ, Boon JJ, van den Brink O, Heeren RMA (2001a) Fragmentation of suberin and composition of aliphatic monomers released by methanolysis of cork from Quercus suber L., analysed by GC-MS, SEC and MALDI-MS. Holzforschung 55:487–493
Bento MFS, Pereira H, Cunha MA, Moutinho AMC, van den Berg KJ, Boon JJ (2001b) A study of variability of suberin composition in cork from Quercus suber L. using thermally assisted transmethylation GC-MS. J Anal Appl Pyrol 57:45–55
Bernards MA (2002) Demystifying suberin. Can J Bot 80:227–240
Bernards MA, Lewis NG (1992) Alkyl ferulates in wound-healing poato-tubers. Phytochemistry 31:3409–3412
Bernards MA, Lewis NG (1998) The macromolecular aromatic domain in suberized tissue: a changing paradigm. Phytochemistry 47:915
Bernards MA, Lopez ML, Zajicek J, Lewis NG (1995) Hydroxycinnamic acid-derived polymers constitute the polyaromatic domain of suberin. J Biol Chem 270:7382–7386
Bernards MA, Fleming WD, Llewellyn DB, Priefer R, Yang XL, Sabatino A, Plourde GL (1999) Biochemical characterization of the suberization-associated anionic peroxidase of potato. Plant Physiol 121:135–145
Borchert R (1978) Time course and spatial distribution of phenylalanine ammonia lyase and peroxidase activity in wounded potato-tuber tissue. Plant Physiol 62:789–793
BorgOlivier O, Monties B (1989) Characterization of lignins, phenolic acids and tyramine in the suberized tissues of natural and wound induced potato periderm. C R Acad Sci III, Sci Vie 308:141–147
BorgOlivier O, Monties B (1993) LIGNIN, suberin, phenolic-acids and tyramine in the suberized, wound-induced potato periderm. Phytochemistry 32:601–606
Bunzel M, Ralph J, Marita JM, Hatfield RD, Steinhart H (2001) Diferulates as structural components in soluble and insoluble cereal dietary fibre. J Sci Food Agric 81:653–660
Bunzel M, Allerdings E, Ralph J, Steinhart H (2008) Cross-linking of arabinoxylans via 8–8-coupled diferulates as demonstrated by isolation and identification of diarabinosyl 8–8 (cyclic)-dehydrodiferulate from maize bran. J Cereal Sci 47:29–40. doi:10.1016/j.jcs.2006.12.005
Cottle W, Kolattukudy PE (1982) Biosynthesis, deposition, and partial characterization of potato suberin phenolics. Plant Physiol 69:393–399
Espelie KE, Kolattukudy PE (1985) Purification and characterization of an abscisic acid inducible anionic peroxidase associated with suberization in potato (Solanum tuberosum). Arch Biochem Biophys 240:539–545
GarciaVallejo MCG, Conde E, Cadahia E, deSimon BF (1997) Suberin composition of reproduction cork from Quercus suber. Holzforschung 51:219–224
Graça J, Pereira H (1998) Feruloyl esters of omega-hydroxyacids in cork suberin. J Wood Chem Technol 18:207–217
Graça J, Pereira H (1999) Glyceryl–acyl and aryl–acyl dimers in Pseudotsuga menziesii bark suberin. Holzforschung 53:397–402
Graça J, Pereira H (2000) Suberin structure in potato periderm: glycerol, long-chain monomers, and glyceryl and feruloyl dimers. J Agric Food Chem 48:5476–5483
Graça J, Santos S (2007) Suberin: a biopolyester of plants’ skin. Macromol Biosci 7:128–135. doi:10.1002/mabi.200600218
Guillemonat A, Poite, Roussel, Traynard JC, Triaca (1963) Sur la constitution chimique du liège. 4. Mémoire—structure de la phellochryseine. Bull Soc Chim Fr: 142–144
King RR, Calhoun LA (2005) Characterization of cross-linked hydroxycinnamic acid amides isolated from potato common scab lesions. Phytochemistry 66:2468–2473. doi:10.1016/j.phytochem.2005.07.014
Kollatukudy PE (2002) Suberin from plants. In: Doi Y, Steinbuchel A (eds) Biopolymers Polyesters I. Wiley-VCH, Weinheim, pp 41–73
Kollatukudy PE, Espelie KE (1989) Chemistry, biochemistry, and function of suberin and associated waxes. In: Rover JW (ed) Natural products of woody plants, vol I. Springer, Berlin, pp 304–367
Laver ML, Fang HHL (1989) Douglas-Fir bark. 4. Ferulic acid-esters from bark of Pseudotsuga menziesii. J Agric Food Chem 37:114–116
Lotfy S, Negrel J, Javelle F (1994) Formation of omega-feruloyloxypalmitic acid by an enzyme from wound-healing poato-tuber disks. Phytochemistry 35:1419–1424
Lotfy S, Javelle F, Negrel J (1995) Distribution of hydroxycinnamoyl-CoA-omega-hydroxypalmitic acid O-hydroxycinnamoyltransferase in higher plants. Phytochemistry 40:389–391
Marques AV, Pereira H, Meier D, Faix O (1994) Quantitative analysis of cork (Quercus suber L.) and milled cork lignin by FTIR spectroscopy, analytical pyrolysis, and total hydrolysis. Holzforschung 48:43–50
Marques AV, Pereira H, Meier D, Faix O (1999) Structural characterization of cork lignin by thioacidolysis and permanganate oxidation. Holzforschung 53:167–174
Molina I, Bonaventure G, Ohlrogge J, Pollard M (2006) The lipid polyester composition of Arabidopsis thaliana and Brassica napus seeds. Phytochemistry 67:2597–2610. doi:10.1016/j.phytochem.2006.09.011
Negrel J, Javelle F, Paynot M (1993) Wound-induced tyramine hydroxycinnamoyl transferase in Potato (Solanum tuberosum) tuber disks. J Plant Physiol 142:518–524
Negrel J, Pollet B, Lapierre C (1996) Ether-linked ferulic acid amides in natural and wound periderms of potato tuber. Phytochemistry 43:1195–1199
Pearce RB, Holloway PJ (1984) Suberin in the sapwood of oak (Quercus robur L.)—its composition from a compartmentalization barrier and its ocurrence in tylosis in undecayed wood. Physiol Plant Pathol 24:71–81
Razem FA, Bernards MA (2002) Hydrogen peroxide is required for poly(phenolic) domain formation during wound-induced suberization. J Agric Food Chem 50:1009–1015. doi:10.1021/jf0110248
Riley RG, Kolattukudy PE (1975) Evidence for covalently attached para-coumaric acid and ferulic acid in cutins and suberins. Plant Physiol 56:650–654
Santos S, Graça J (2006) Glycerol-omega-hydroxyacid-ferulic acid oligomers in cork suberin structure. Holzforschung 60:171–177. doi:10.1515/hf.2006.028
Schmutz A, Jenny T, Amrhein N, Ryser U (1993) Caffeic acid and glycerol are constituents of the suberin layers in green cotton fibers. Planta 189:453–460
Schmutz A, Jenny T, Ryser U (1994) A caffeoyl fatty-acid glycerol ester from wax associated with green cotton fiber suberin. Phytochemistry 36:1343–1346
Schreiber L, Franke R, Hartmann K (2005) Wax and suberin development of native and wound periderm of potato (Solanum tuberosum L.) and its relation to peridermal transpiration. Planta 220:520–530. doi:10.1007/s00425-004-1364-9
Sitte P (1962) Zum feinbau der suberinschichten im flaschenkork. Protoplasma 54:555–559
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Graça, J. Hydroxycinnamates in suberin formation. Phytochem Rev 9, 85–91 (2010). https://doi.org/10.1007/s11101-009-9138-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11101-009-9138-4