Abstract
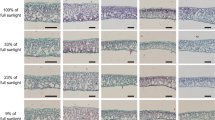
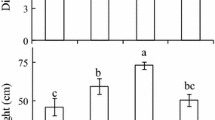
Cistus salvifolius L. is the most widely spread Cistus species around the Mediterranean basin. It colonizes a wide range of habitats growing from sea level to 1,800 m a.s.l., on silicolous and calcicolous soils, in sun areas as well as in the understory of wooded areas. Nevertheless, this species has been mainly investigated in term of its responsiveness to drought. Our aim was to understand which leaf traits allow C. salvifolius to cope with low-light environments. We questioned if biochemical and physiological leaf trait variations in response to a reduced photosynthetic photon flux density were related to leaf morphological plasticity, expressed by variations of specific leaf area (SLA) and its anatomical components (leaf tissue density and thickness). C. salvifolius shrubs growing along the Latium coast (41°43'N,12°18'E, 14 m a.s.l., Italy) in the open and in the understory of a Pinus pinea forest, were selected and the relationships between anatomical, gas exchange, chlorophyll (Chl) fluorescence, and biochemical parameters with SLA and PPFD variations were tested. The obtained results suggested long-term acclimation of the selected shrubs to contrasting light environments. In high-light conditions, leaf nitrogen and Chl contents per leaf area unit, leaf thickness, and Chl a/b ratio increased, thus maximizing net photosynthesis, while in shade photosynthesis, it was downregulated by a significant reduction in the electron transport rate. Nevertheless, the increased pigment-protein complexes and the decreased Chl a/b in shade drove to an increased light-harvesting capacity (i.e. higher actual quantum efficiency of PSII). Moreover, the measured vitality index highlighted the photosynthetic acclimation of C. salvifolius to contrasting light environments. Overall, our results demonstrated the morphological, anatomical, and physiological acclimation of C. salvifolius to a reduced light environment.
Similar content being viewed by others
Abbreviations
- C/N:
-
ratio between soil organic carbon content and total soil nitrogen content
- C a :
-
CO2 concentration in the leaf chamber
- Cab :
-
abaxial cuticle thickness
- Cad :
-
adaxial cuticle thickness
- Car:
-
carotenoids
- Car/Chl:
-
ratio between Car and Chl (a+b)
- C e :
-
apparent carboxylation efficiency
- Chl:
-
chlorophyll
- C i :
-
intercellular CO2 concentration
- DM:
-
dry mass
- E :
-
transpiration rate
- ETab :
-
abaxial epidermis thickness
- ETad :
-
adaxial epidermis thickness
- ETR:
-
electron transport rate
- f ias :
-
fraction of the mesophyll occupied by intercellular air spaces
- FM:
-
fresh mass
- Fm :
-
maximal fluorescence yield of the dark-adapted state
- Fm':
-
maximal fluorescence yield of the light-adapted state
- Fs :
-
steady-state fluorescence yield
- g s :
-
stomatal conductance
- LA:
-
leaf area
- LMA:
-
leaf mass area
- LT:
-
total leaf thickness
- LTD:
-
leaf tissue density
- Narea :
-
nitrogen content per unit leaf area
- Nleaf :
-
leaf nitrogen content
- NORG :
-
organic nitrogen content per unit of leaf area
- NP :
-
nitrogen allocated to the pigment-protein complexes
- NP% :
-
NP as percentage of NORG
- P:
-
palisade parenchyma thickness
- P N :
-
net photosynthetic rate
- R/FR:
-
ratio between irradiance in the red and far red wavelengths
- R D :
-
respiration rate
- R D/P N :
-
ratio between R D and P N
- Rfd :
-
vitality index
- S:
-
spongy parenchyma thickness
- SD:
-
stomatal density
- SLA:
-
specific leaf area
- SPA:
-
stomatal pore area
- SPL:
-
stomatal pore length
- SPW:
-
stomatal pore width
- Tl:
-
leaf temperature
- T m :
-
mean air temperature
- T max :
-
mean maximum air temperature
- T min :
-
mean minimum air temperature
- WUEi :
-
intrinsic water-use efficiency
- a:
-
absorptance
- ΦFPSII:
-
effective quantum yield of PSII photochemistry
- χ:
-
chlorophyll content per unit of leaf area
References
Atkin O.K., Evans J.R., Siebke K.: Relationship between the inhibition of leaf respiration by light and enhancement of leaf dark respiration following light treatment. - Aust. J. Plant. Physiol. 25: 437–443, 1998.
Attaguile G., Russo A., Campisi A. et al.: Antioxidant activity and protective effect on DNA cleavage of extracts from Cistus incanus L. and Cistus monspeliensis L. - Cell. Biol. Toxicol. 16: 83–90, 2000.
Bartolini S., Minnocci A., Vitagliano C.: Influence of temperature on morphoanatomic characteristics of “Trebbiano” gravepine leaves. - Agricolt. Mediterr. 127: 37–43, 1997.
Bosch J.: Floral biology and pollinators of three co-occurring Cistus species (Cistaceae). - Bot. J. Linn. Soc. 109: 39–55, 1992.
Cai Z., Slot M., Fan Z.: Leaf development and photosynthetic properties of three tropical tree species with delayed greening. - Photosynthetica 43: 91–98, 2005.
Chabot B.F., Chabot J.F.: Effects of light and temperature on leaf anatomy and photosynthesis in Fragaria vesca. - Oecologia 26: 363–377, 1977.
Delgado-Sánchez P., Yáñez-Espinosa L., Jiménez-Bremont J.F. et al.: Ecophysiological and anatomical mechanisms behind the nurse effect: Which are more important? A multivariate approach for cactus seedlings. - PLoS ONE 8: e81513, 2013.
DellaPenna D., Pogson B.J.: Vitamin synthesis in plants: tocopherols and carotenoids. - Annu. Rev. Plant. Biol. 57: 711–738, 2006.
Evans J.R.: Photosynthesis and nitrogen relationships in leaves of C3 plants. - Oecologia 78: 9–19, 1989.
Evans J.R.: Leaf anatomy enables more equal access to light and CO2 between chloroplasts. - New Phytol. 143: 93–104, 1999.
Evans J.R., Poorter H.: Photosynthetic acclimation of plants to growth irradiance: the relative importance of specific leaf areamand nitrogen partitioning in maximizing carbon gain. - Plant Cell Environ. 24: 755–767, 2001.
Farley R.A., McNeilly T.: Diversity and divergence in Cistus salvifolius (L.) populations from contrasting habitats. - Hereditas 132: 183–192, 2000.
Fernández-Mazuecos M., Vargas P.: Ecological rather than geographical isolation dominates Quaternary formation of Mediterranean Cistus species. - Mol. Ecol. 19: 1381–1395, 2010.
Flexas J., Gulías J., Jonasson S. et al.: Seasonal patterns and control of gas exchange in local populations of the Mediterranean evergreen shrub Pistacia lentiscus L. - Acta Oecol. 22: 33–43, 2001.
Galmés J., Flexas J., Savé R., Medrano H.: Water relations and stomatal characteristics of Mediterranean plants with different growth forms and leaf habits: responses to water stress and recovery. - Plant Soil 290: 139–155, 2007.
Genty B., Briantais J.M., Baker N.R.: The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. - Biochim. Biophys. Acta 990: 87–92, 1989.
Grant O.M., Tronina Ł., García-Plazaola J.I. et al.: Resilience of a semi-deciduous shrub, Cistus salvifolius, to severe summer drought and heat stress. - Funct. Plant Biol. 42: 219–228, 2014.
Gratani L.: Plant phenotypic plasticity in response to environmental factors. - Adv. Bot. 2014: 208–747, 2014.
Guzmán B., Lledó M.D., Vargas P.: Adaptive radiation in Mediterranean Cistus. - PLoS ONE 4: 1–13, 2009.
Hallik L., Niinemets Ü., Kull O.: Photosynthetic acclimation to light in woody and herbaceous species: a comparison of leaf structure, pigment content and chlorophyll fluorescence characteristics measured in the field. - Plant Biol. 14: 88–99, 2012.
Harley P.C., Tenhunen J.D., Beyschlag W., Lange O.L.: Seasonal changes in net photosynthesis rates and photosynthetic capacity in leaves of Cistus salvifolius, a European Mediterranean semideciduous shrub. - Oecologia 74: 380–388, 1987.
Holm G.: Chlorophyll mutations in barley. - Acta Agr. Scand. BS P. 4: 457–471, 1954.
Kitajima K., Hogan K.P.: Increases of chlorophyll a/b ratios during acclimation of tropical woody seedlings to nitrogen limitation and high light. - Plant Cell Environ. 26: 857–865, 2003.
Krall J.P., Edwards G.E.: Relationship between photosystem IIactivity and CO2 fixation in leaves. - Physiol. Plantarum 86: 180–187, 1992.
Kull O., Niinemets Ü.: Variation in leaf morphometry and nitrogen concentration in Betula pendula Roth., Corylus avellana L. and Lonicera xylosteum L. - Tree Physiol. 12: 311–318, 1993.
Kursar T.A.: Relating tree physiology to past and future changes in tropical rainforest tree communities. - Climatic Change 39: 363–379, 1998.
Lichtenthaler H.K., Rinderle U.: The role of chlorophyll fluorescence in detection of stress conditions in plants. - Crit. Rev. Anal. Chem. 19: S29–S85, 1988.
MacLachlan S., Zalik S.: Plastid structure, chlorophyll concentration, and free aminoacid composition of a chlorophyll mutant of barley. - Can. J. Bot. 41: 1053–1062, 1963.
Matsubara S., Krause G.H., Aranda J. et al.: Sun-shade patterns of leaf carotenoid composition in 86 species of neotropical forest plants. - Funct. Plant. Biol. 36: 20–36, 2009.
Mendes M.M., Gazarini L.C., Rodrigues M.L.: Acclimation of Myrtus communis to contrasting Mediterranean light environments–effects on structure and chemical composition of foliage and plant water relations. - Environ. Exp. Bot. 45: 165–178, 2001.
Middleton L.: Shade-tolerant flowering plants: adaptations and horticultural implications. - Acta Hortic. 552: 95–102, 2001.
Mishra M.K.: Stomatal characteristics at different ploidy levels in Coffea L. - Ann. Bot.-London 80: 689–692, 1997.
Nar H., Saglam A., Terzi R. et al.: Leaf rolling and photosystem IIefficiency in Ctenanthe setosa exposed to drought stress. - Photosynthetica 47: 429–436, 2009.
Niinemets Ü.: Components of leaf dry mass per area - thickness and density - alter leaf photosynthetic capacity in reverse directions in woody plants. - New Phytol. 144: 35–47, 1999.
Niinemets Ü.: The controversy over traits conferring shadetolerance in trees: ontogenetic changes revisited. - J. Ecol. 94: 464–470, 2006.
Niinemets Ü.: Photosynthesis and resource distribution through plant canopies. - Plant Cell Environ. 30: 1052–1071, 2007.
Niinemets Ü.: A review of light interception in plant stands from leaf to canopy in different plant functional types and in species with varying shade tolerance. - Ecol. Res. 25: 693–714, 2010.
Niinemets Ü., Kull O.: Stoichiometry of foliar carbon constituents varies along light gradients in temperate woody canopies: implications for foliage morphological plasticity. - Tree Physiol. 18: 467–479, 1998.
Niinemets Ü., Kull O., Tenhunen J.D.: Within canopy variation in the rate of development of photosynthetic capacity is proportional to integrated quantum flux density in temperate deciduous trees. - Plant Cell Environ. 27: 293–313, 2004.
Núñez-Olivera E., Martínez-Abaigar J., Escudero J.C.: Adaptability of leaves of Cistus ladanifer to widely varying environmental conditions. - Funct. Ecol. 10: 636–646, 1996.
Papaefthimiou D., Papanikolaou A., Falara V. et al.: Genus Cistus: a model for exploring labdane- type-diterpenes’ biosynthesis and a natural source of high value products with biological, aromatic, and pharmacological properties. - Front. Chem. 2: 1–19, 2014.
Poorter H., Niinemets Ü., Poorter L. et al.: Causes and consequences of variation in leaf mass per area (LMA): a metaanalysis. - New Phytol. 182: 565–588, 2009.
Price T.D., Qvarnström A., Irwin D.E.: The role of phenotypic plasticity in driving genetic evolution. - P. Roy. Soc. Lond. B Bio. 270: 1433–1440, 2003.
Puglielli G., Crescente M.F., Frattaroli A.R., Gratani L.: Morphological, anatomical and physiological leaf trait plasticity of Sesleria nitida in open vs shaded conditions. - Pol. J. Ecol. 63: 10–22, 2015.
Reich P.B., Walters M.B., Kloeppel B.D., Ellsworth D.S., Walters M.B.: Different photosynthesis nitrogen relations in deciduous hardwood and evergreen coniferous tree species. - Oecologia 104: 24–30, 1995.
Sack L., Cowan P.D., Jaikumar N., Holbrook N.M.: The „hydrology“ of leaves: co-ordination of structure and function in temperate woody species. - Plant Cell Environ. 26: 1343–1356, 2003.
Sack L., Melcher P.J., Liu W.H. et al.: How strong is intracanopy leaf plasticity in temperate deciduous trees? - Am. J. Bot. 93: 829–839, 2006.
Short M.J.: Cistaceae. - In: Press J.R., Short M.J. (ed.): Flora of Madeira. Pp. 226–227. The Natural History Museum, HMSO, London 1994.
Syvertsen J.P., Lloyd J., McConchie C. et al.: On the relationship between leaf anatomy and CO2 diffusion through the mesophyll of hypostomatous leaves. - Plant Cell Environ. 18: 149–157, 1995.
Valladares F., Niinemets Ü.: Shade tolerance, a key plant feature of complex nature and consequences. - Annu. Rev. Ecol. Evol. S. 39: 237–257, 2008.
Warburg E.F.: Cistus L. - In: Tutin T.G., Heywood V.H., Burges N.A. et al. (ed.): Flora Europaea, Vol. 2. Pp.282–285. Cambridge University Press, Cambridge 1968.
Wright I.J., Westoby M.: Leaves at low versus high rainfall: coordination of structure, lifespan and physiology. - New Phytol. 155: 403–416, 2002.
Yamashita N., Koike N., Ishida A.: Leaf ontogenetic dependence of light acclimation in invasive and native subtropical trees of different successional status. - Plant Cell Environ. 25: 1341–1356, 2002.
Yang S., Sun M., Zhang Y. et al.: Strong leaf morphological, anatomical, and physiological responses of a subtropical woody bamboo (Sinarundinaria nitida) to contrasting light environments. - Plant Ecol. 215: 97–109, 2014.
Zunzunegui M., Ain-Lhout F., Díaz Barradas M.C. et al.: Physiological, morphological and allocation plasticity of a semi-deciduous shrub. - Acta Oecol. 35: 370–379, 2009.
Zunzunegui M., Díaz-Barradas M.C., Jáuregui J. et al.: Seasondependent and independent responses of Mediterranean scrub to light conditions. - Plant Physiol. Bioch. 102: 80–91, 2016.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Puglielli, G., Varone, L., Gratani, L. et al. Specific leaf area variations drive acclimation of Cistus salvifolius in different light environments. Photosynthetica 55, 31–40 (2017). https://doi.org/10.1007/s11099-016-0235-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11099-016-0235-5