Abstract
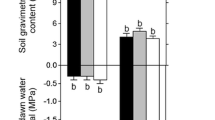
Water stress is a major abiotic constraint leading to serious crop losses. Recently, in the Mediterranean region, water stress has become markedly sensed, especially in Citrus orchards. This study investigated the physiological responses of local sour orange (Citrus aurantium L.) clones to severe water stress. Water stress was applied by withholding irrigation during weeks, followed by a rewatering phase during three months. Under water stress, sour orange clones decreased their stomatal conductance, net photosynthetic rate, and transpiration rate. On the contrary, biomass was stable, especially in the Kliaa clone. In addition, reduced leaf water potentials (-3 MPa) and water contents were measured in most of the clones, except Kliaa which kept the highest water potential (-2.5 MPa). After rewatering, all clones recovered except of the Ghars Mrad (GM) clone. Ultrastructural observations of leaf sections by transmission electron microscopy did not reveal marked alterations in the mesophyll cells and chloroplast structure of Kliaa in comparison to the sensitive clone GM, in which palisade parenchyma cells and chloroplasts were disorganized. This contrasting behavior was mainly attributed to genetic differences as attested by molecular analysis. This study highlighted GM as the drought-sensitive clone and Kliaa as the tolerant clone able to develop an avoidance strategy based on an efficient stomatal regulation. Although a high percentage of polyembryony characterizes C. aurantium and justifies its multiplication by seeds, heterogeneous water-stress responses could be observed within sour orange plants in young orchards.
Similar content being viewed by others
Abbreviations
- C:
-
control, irrigated plants
- C i :
-
intercellular CO2 concentration
- DM:
-
dry mass
- E :
-
transpiration
- EH:
-
equivalent humidity
- FM:
-
fresh mass
- GM:
-
Ghars Mrad
- g s :
-
stomatal conductance
- IBA:
-
indol-butyric acid
- LWC:
-
leaf water content
- P N :
-
net photosynthetic rate
- RAPD:
-
random amplified polymorphic DNA
- REW:
-
rewatered plants
- TEM:
-
transmission electron microscopy
- Wg:
-
gravimetric soil water content
- WS:
-
water-stressed plants
- Ψw :
-
leaf water potential
References
Ali Q., Ashraf M.: Induction of drought tolerance in maize (Zea mays L.) due to exogenous application of trehalose: growth, photosynthesis, water relations and oxidative defense mechanism.–J. Agron. Crop Sci. 194: 258–271, 2011.
Aroca R.: Plant Responses to Drought Stress: from Morphological to Molecular Features. Pp. 417–438. Springer, Berlin Heidelberg 2012.
Aubert B., Vullin G.: [Nurseries and citrus plantations.] Pp. 184. CIRAD, Montpellier 1997. [In French]
Bates L., Waldren R.P., Teare I.D.: Rapid determination of free proline for water-stress studies.–Plant Soil 39: 205–207, 1973.
Ben Salem-Fnayou A., Bouamama B., Ghorbel A., Mliki A.: Investigations on the leaf anatomy and ultrastructure of grapevine (Vitis vinifera L.) under heat stress.–Microsc. Res. Tech. 74: 756–762, 2011.
Bondada B.R., Syvertsen J.P.: Concurrent changes in net CO2 assimilation and chloroplast ultrastructure in nitrogen deficient Citrus leaves.–Environ. Exp. Bot. 54: 41–48, 2005.
Boujnah D., Gouiaa M., Braham M. et al.: [Trials of preservation irrigation of olive trees under extreme drought conditions in the semi-arid: ecophysiological aspects of recovery.]–Ezzaitouna 10:1–2, 2004. [In French]
Challabathula D., Djilianov D., Bartels D.: Photosynthesis in dessication tolerant plants: Energy metabolism and antioxidative stress defense.–Plant Sci. 182: 29–41, 2012.
Chaves M.M., Flexas, J., Pinheiro C.: Photosynthesis under drought and salt stress: regulation mechanisms from whole plant to cell.–Ann. Bot-London 103: 551–560, 2009.
Chaves M.M., Maroco J.P., Pereira J.S.: Understanding plant responses to drought–from genes to the whole plant.–Funct. Plant Biol. 30: 239–264, 2003.
da Silva C.R., Folegatti M.V., da Silva T.J.A. et al.: Water relations and photosynthesis as criteria for adequate irrigation management in “Tahiti” lime trees.–Sci. Agric. 62: 415–422, 2005.
Dellaporta S.L., Wood J., Hicks J.B.: A plant DNA minipreparation: Version II.–Plant Mol. Biol. Rep. 1: 19–21, 1983.
di Ferdinando M., Brunetti C., Fini A., Tattini M.: Flavonoids as antioxidants in plants under abiotic stresses.–In: Ahmad P., Prasad M.N.V. (ed.): Abiotic Stress Responses in Plants, Metabolism, Productivity and Sustainability. Pp. 465. Springer, New York 2012.
Flexas J., Medrano H.: Drought-inhibition of photosynthesis in C3 plants: stomatal and non-stomatal limitations revisited.–Ann. Bot-London 89: 183–189, 2002.
García-Sánchez F., Syvertsen J.P., Gimeno V. et al.: Responses to flooding and drought stress by two citrus rootstock seedlings with different water-use efficiency.–Physiol. Plantarum 130: 532–542, 2007.
Gimeno V., Díaz-López L., Simón-Grao S. et al.: Foliar potassium nitrate application improves the tolerance of Citrus macrophylla L. seedlings to drought conditions.–Plant Physiol. Bioch. 83: 308–315, 2014.
Hummel I., Pantin F., Sulpice R. et al.: Arabidopsis plants acclimate to water deficit at low cost through changes of carbon usage: an integrated perspective using growth, metabolite, enzyme and gene expression analysis.–Plant Physiol. 154: 357–372, 2010.
Isarangkool N.A.S., Do F.C., Pannengpetch K. et al.: Water loss regulation in mature Hevea brasiliensis: effects of intermittent drought in the rainy season and hydraulic limitation.–Tree Physiol. 31: 751–762, 2011.
Kramer P.J., Boyer J.S.: Water Relations in Plants and Soils. Pp. 482. Elsevier Sci. Acad. Press, California 1995.
Liu Z.J., Guo Y.K., Bai J.G.: Exogenous hydrogen peroxide changes antioxidant enzyme activity and protects ultrastructure in leaves of two cucumber ecotypes under osmotic stress.–J. Plant Growth Regul. 29: 171–183, 2010.
Loussert R.: [Mediterranean agricultural techniques.]–Agrumes 1: 3–41, 1989. [In French]
Mohd R.I., Abd Ghani M., Ismail I.: Differential responses in growth, physiological processes and peroxidase activity of young mango (Mangifera indica) and Citrus (Citrus sinensis L.) plants to water deficit.–Pertanika J. Trop. Agric. Sci. 27: 47–55, 2004.
Pérez-Pérez J.G., Syvertsen J.P., Botía P., García-Sánchez F.: Leaf water relations and net gas exchange responses of salinized Carrizo citrange seedlings during drought stress and recovery.–Ann Bot-London 100: 335–345, 2007.
Przybyl K., Idzikowska K.: Ultrastructural changes in chloroplasts of mesophyll cells of chlorotic and prematurely yellowed leaves of Betula pendula Rothr.–Acta Soc. Bot. Pol. 72: 289–293, 2003.
Rejeb M.N.: [Effect of extreme temperatures on plants, responses and adaptation strategies.]–PhD. Thesis. University of Tunis, Tunis 1992. [In French]
Rodríguez-Gamir J., Primo-Millo E., Forner J.B., Forner-Giner M.A.: Citrus rootstock responses to water stress.–Sci. Hortic-Amsterdam. 126: 95–102, 2010.
Shao R.X., Xin L.F., Zheng H.F. et al.: Changes in chloroplast ultrastructure in leaves of drough-stressed maize inbred lines.–Photosynthetica 54: 74–80, 2016
Sokal R.R., Michener C.D.: A statistical method for evaluating systematic relationships.–Univ. Kans. Sci. Bull. 38: 1409–1438, 1958.
Souza R.P., Machado E.C., Silva J.A.B. et al.: Photosynthetic gas exchange, chlorophyll fluorescence and some associated metabolic changes in cowpea (Vigna unguiculata) during water stress and recovery.–Environ. Exp. Bot. 51: 45–56, 2004.
Staub A.M.: [Carbohydrates extraction, identification and dosing in bacteria tissue extracts.]–In: Loiseleur J. (ed.): [Laboratory Techniques.] Pp 1307–1366, Masson, Paris 1963. [In French]
Toumi I.: [Physiological and biochemical responses of grapevine to water and salt stresses: involvement of polyamin metabolism in the tolerance response.]–PhD. Thesis. University of Tunis, Tunis, 2008. [In French]
Wu Q.S., Xia R.X., Zou Y.N.: Improved soil structure and citrus growth after inoculation with three arbuscular mycorrhizal fungi under drought stress.–Eur. J. Soil Biol. 44: 122–128, 2008.
Author information
Authors and Affiliations
Corresponding author
Additional information
Acknowledgements: Authors are grateful to Mrs. Aouatef Ben Ammar (Laboratory of Electron Microscopy, Faculty of Medicine,Tunis) for technical assistance concerning transmission electron microscopy.
Rights and permissions
About this article
Cite this article
Salem-Fnayou, A.B., Belghith, I., Lamine, M. et al. Physiological and ultrastructural responses of sour orange (Citrus aurantium L.) clones to water stress. Photosynthetica 54, 532–541 (2016). https://doi.org/10.1007/s11099-016-0221-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11099-016-0221-y