Abstract
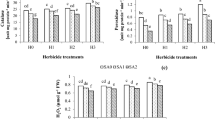
In this work, the injuries caused by clethodim herbicide application as well as the use of exogenous salicylic acid (SA) as a protective agent against clethodim in Zea mays leaves were examined. Although the target for clethodim is the inhibition of acetyl coenzyme A carboxylase (ACCase) which is the key enzyme for fatty acid biosynthesis, it can indirectly affect the photosynthetic machinery, gaseous exchange and some biochemical parameters. Clethodim application caused chlorosis and yellowing of leaf-tip parts. Higher doses caused browning or reddening of leaves and sometimes dead parts of the leaf margins were observed. The rate of photosynthesis was significantly lowered and the pigments content was highly reduced as a response to clethodim spraying. Moreover, other gas-exchange properties were altered. Furthermore, accumulation of high amounts of carbohydrates, proteins and proline were detected. SA spraying three days prior clethodim application caused partially or totally disappearance of clethodim injuries and kept the leaves similar to those of control. Improved photosynthesis and enhanced pigments content were observed in leaves treated with SA. Other analyzed parameters showed values similar to those of the corresponding control. From the experimental work, an evidenced role of SA working against clethodim effects was suggested and discussed in this paper.
Similar content being viewed by others
Abbreviations
- ACCase:
-
acetyl coenzyme A carboxylase
- C i :
-
intercellular CO2 concentration
- Cars:
-
carotenoids
- Chl:
-
chlorophyll
- DM:
-
dry mass
- E :
-
transpiration rate
- g s :
-
stomatal conductance
- P N :
-
photosynthetic rate
- SA:
-
salicylic acid
- WUE:
-
water-use efficiency
References
Ananievaa, E.A., Alexieva, V.S., Popova, L.P.: Treatment with salicylic acid decreases the effects of paraquat on photosynthesis. — J. Plant Physiol. 159: 685–693, 2002.
Ananievaa, E.A., Christov, K.N., Popova, LP.: Exogenous treatment with salicylic acid leads to increased antioxidant capacity in leaves of barley plants exposed to paraquat. — J. Plant Physiol. 161: 319–328, 2004.
Anderson, R.N., Geadelmann, J.L.: The effect of parentage on the control of volunteer corn (Zea mays) in soybeans (Glycine max). — Weed Sci. 30: 127–136, 1982.
Arfan, M., Athar, H.R., Ashraf, M.: Does exogenous application of salicylic acid through the rooting medium modulate growth and photosynthetic capacity in two differently adapted spring wheat cultivars under salt stress? — J. Plant Physiol. 164: 685–694, 2007.
Barkosky, R.R., Einhellig, F.A.: Effects of salicylic acid on plant water relationship — J. Chem. Ecol. 19: 237–47, 1993.
Barry, P., Young, A.J., Britton, G.: Photodestruction in higher plants by herbicide action. — J. Exp. Bot. 41: 123–123, 1990.
Bates, L.S., Waldern, R.P., Teare, I.D.: Rapid determination of free proline for water stress studies. — Plant Soil 39: 205–207, 1973.
Borsani, O., Valpuesta, V., Botella, M.A.: Evidence for a role of salicylic acid in the oxidative damage generated by NaCl and osmotic stress in Arabidopsis seedlings. — Plant Physiol. 126: 1024–1030, 2001.
Croon, K.A., Ketchersid, M.L., Merkle, M.G.: Effect of bentazon, imazaquin, chlorimuron on the absorption and translocation of the methyl ester of haloxyfop. — Weed Sci. 37: 645–650, 1989.
Duncan, D.B.: A significance test for differences between ranked treatments in analyses of variance. — Virginia J. Sci. 2: 171–189, 1951.
El-Tayeb, M.A.: Response of barley grains to the interactive effect of salinity and salicylic acid. — Plant Growth Regul. 45: 215–224, 2005.
Fales, F.W.: The assimilation and degradation of carbohydrates by yeast cells. — J. Biol. Chem 193: 113–113, 1951.
Fariduddin, Q., Hayat, S., Ahmad, A.: Salicylic acid influences net photosynthetic rate, carboxylation efficiency, nitrate reductase activity and seed yield in Brassica juncea. — Photosynthetica 41: 281–284, 2003.
Faville, M.J., Silvester, W.B., Green, T.G.A., Jermyn, W.A.: Photosynthetic characteristics of three asparagus cultivars differing in yield. — Crop Sci. 39: 1070–7, 1999.
Fayez, K.A.: Action of photosynthetic diuron herbicide on cell organelles and biochemical instituents of the leaves of two soybean cultivars. — Pesticide Biochem. Physiol. 66: 105–115, 2000.
Ghai, N., Setia, R.C., Setia, N.: Effects of paclobutrazol and salicylic acid on chlorophyll content, hill activity and yield components in Brassica napus L. (cv. GSL-1). — Phytomorphology 52: 83–87, 2002.
Gunes, A., Inal, A., Alpaslan, M., Eraslan, F., Bagci, E.G., Cicek, N.: Salicylic acid induced changes on some physiological parameters symptomatic for oxidative stress and mineral nutrition in maize (Zea mays L.) grown under salinity. — J. Plant Physiol. 164: 728–736, 2007.
Halim, V.A., Vess, A., Scheel, D., Rosahl, S.: The role of salicylic acid and jasmonic acid in pathogen defence. — Plant Biol. 8: 307–313, 2006.
Harwood J.L.: Fatty acid metabolism. — Annu Rev. Plant Physiol. Plant Mo1. Biol. 39: 101–138, 1988.
Hayat, Q., Hayat, S., Irfan, M., Ahmad, A.: Effect of exogenous salicylic acid under changing environment: A review. — Environ. Exp. Bot. 68: 14–25, 2010.
Hayat, S., Fariduddin, Q., Ali, B., Ahmad, A.: Effect of salicylic acid on growth and enzyme activities of wheat seedlings. — Acta Agron. Hung. 53: 433–437, 2005.
Hellergren, J., Li, P.H.: Survival of solanum tuberosum suspension cultures to −14 °C: The mode of action of proline. — Plant Physiol. 52: 449–453, 1981.
Incledon, B.J., Hall, J.C.: Inhibition of ACCase 220 and ACCase 240 isozymes from sethoxydim resistant and susceptible maize hybrids. — J. Agric. Food Chem. 471: 299–304, 1999.
Iwataki, I.: Cyclohexanedione herbicides: Their activities and properties; — CRC Press: Boca Raton. Vol. XVI: 397–426, 1992.
Janda T, Szalai G, Tari I, Páldi E.: Hydroponic treatment with salicylic acid decreases the effects of chilling injury in maize (Zea mays L.) plants. — Planta 208: 175–180, 1999.
Jordan, L.D., Leon, S., Warren, J.R., et al.: Weed science, Italian ryegrass control with preplant herbicides. — J. Cotton Sci. 5: 268–274, 2001.
Khan, W., Prithviraj, B., Smith, D.L.: Photosynthetic responses of corn and soybean to foliar application of salicylates. — J. Plant Physiol. 160: 485–492, 2003.
Kilili, K.G., Atanassova, N., Vardanyan, A., Clatot, N., Al-Sabarna, K., Kanellopoulos, P.N., Makris, A.M., Kampranis, S.C.: Differential roles of tau class glutathione S-transferases in oxidative stress. — J. Biol. Chem. 279: 24540–24551, 2004.
Kumar, P., Lakshmi, N.J., Mani, V.P.: Interactive effects of salicylic acid and phytohormones on photosynthesis and grain yield of soybean (Glycine max L Merrill). — Physiol. Mol. Biol. Plants 6: 179–186, 2000.
Larque, S.A.: Stomatal closure in response to acetylsalicylic acid treatment. — Z. Pflanzenphysiol. 93: 371–375, 1979.
Lichtenthaler, H.K., Buschmann, C.: Chlorophylls and carotenoids — Measurement and characterization by UV-VIS. — In: Lichtenthaler, H.K. (ed.): Current Protocols in Food Analyticial Chemistry (CPFA), (Supplement 1). Pp. 1–8. Wiley & Sons, New York 2001.
Lowry, O.H., Rosebrough, N.J., Farr, A.I., Randall, R.J.: Protein measurement with the folin phenol reagent. — J. Biol. Chem. 193: 291–297, 1951.
Magné, C., Saladin, G., Clément, C.: Transient effect of the herbicide flazasulfuron on carbohydrate physiology in Vitis vinifera L. — Chemosphere 62: 650–657, 2006.
Maleck, K., Levine, A., Eulgem, T., et al.: The transcriptome of Arabidopsis thaliana during systemic acquired resistance. — Nature Genet. 26: 403–410, 2000.
Moharekar, S.T., Lokhande, S.D., Hara, T., Tanaka, R., Tanaka, A., Chavan, P.D.: Effect of salicylic acid on chlorophyll and carotenoid contents of wheat and moong seedlings. — Photosynthetica 41: 315–317, 2003.
Munné-Bosch, S., Alegre: L Changes in carotenoids, tocopherols and diterpenes during drought and recovery, and the biological significance of chlorophyll loss in Rosmarinus officinalis plants. — Planta 210: 925–931, 2000.
Nakajima, Y., Yoshida, S., Ono, T.: Differential effects of urea/triazine-type and phenol-type photosystem II inhibitors on inactivation of the electron transport and degradation of the D1 protein during photon inhibition. — Plant Cell Physiol. 37: 673–680, 1996.
Nelson, K.A., Renner, K.A.: Soybean growth and development as affected by glyphosate and postemergence herbicide tank mixtures. — Agron. J. 93: 428–434, 2001.
Nikolskaya, T., Zagnitko, O., Tevzadze, G., Haselkorn, R., Gornicki, P.: Herbicide sensitivity determinant of wheat plastid acetyl-CoA carboxylase is located in a 400-amino acid fragment of the carboxyltransferase domain. — Proc. Nat. Acad. Sci. USA 96: 14647–14651, 1999.
Ohlrogge, J.B., Jaworski, J.G.: Regulation of fatty acid synthesis. — Annu. Rev. Plant Physiol. 48: 109–136, 1997.
Radwan, D.E.M., Fayez, K.A., Mahmoud, S.Y., Hamad, A., Lu, G.: Physiological and metabolic changes of Cucurbita pepo leaves in response to zucchini yellow mosaic virus (ZYMV) infection and salicylic acid treatments. — Plant Physiol. Bioch. 45: 480–489, 2007.
Rai, V.K., Sharma, S.S., Sharma, S.: Reversal of ABA-induced stomatal closure by phenolic compounds. — J. Exp. Bot. 37: 129–134, 1986.
Saladin, G., Magné, C., Clément, C.: Impact of flumioxazin herbicide on growth and carbohydrate physiology in Vitis vinifera L. — Plant Cell Rep. 21: 821–827, 2003.
Sandmann, G., Böger, P.: Sites of herbicide action at photosynthetic apparatus. — In: Staehelin, L.A, Arntzen, C.S. (ed.): Photosynthesis III. Pp. 595–602. Springer-Verlag, Berlin — Heidelberg — New York — Tokyo 1986.
Schlegel, H.-G.: [Die Verwertung organischer Säuren durch Chlorella im Licht.] — Planta 47: 510–526, 1956. [In German.]
Schobert, R.A.: Is there an osmotic regulatory mechanism in algae and higher plants? — J. Theor. Biol. 86: 17–26, 1977.
Shakirova, F.M., Sakhabutdinova, A.R., Bezrukova, M.V., Fatkhutdinova, R.A., Fatkhutdinova, D.R.: Changes in the hormonal status of wheat seedlings induced by salicylic acid and salinity. — Plant Sci. 164: 317–322, 2003.
Shuting, D., Rongqi, G., Changltao, H., Qunying, W., Koogjun, W.: Study of canopy photosynthesis properties and high yield potential after anthesis in maize. — Acta Agron. Sin. 23: 318–325, 1997.
Strobel, N.E., Kuc: A Chemical and biological inducers of systemic acquired resistance to pathogens protect cucumber and tobacco from damage caused by paraquat and cupric chloride. — Phytopathology 85: 1306–1310, 1995.
Tomaso, J.M.: Evidence against a direct membrane effect in the mechanism of action of graminicides. — Weed Sci. 42: 302–309, 1994.
Vencill, W.K.: Herbicide Handbook. 8th Ed. — Weed Sci. Soc. Amer., Lawrence 2002.
Watahiki, M.K., Mori, H., Yamamoto, K.T.: Inhibitory effects of auxins and related substances on the activity of an Arabidopsis glutathione S-transferase isozyme expressed in Escherichia coli. — Physiol. Plant. 94: 566–574, 1995.
Webb, S.R., Hall, J.C.: Monoclonal based ELISA for the identification of herbicidal cyclohexanedione analogues that inhibit germinaceous acetyl coenzyme A carboxylase. — J. Agric. Food Chem. 48: 1210–1218, 2000.
William, A.: The effect of cell cycle regulators on protein profiles in cultured root meristems of Pesium sativum. — Environ. Exp. Bot. 29: 317–322, 1989.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Radwan, D.E.M., Soltan, D.M. The negative effects of clethodim in photosynthesis and gas-exchange status of maize plants are ameliorated by salicylic acid pretreatment. Photosynthetica 50, 171–179 (2012). https://doi.org/10.1007/s11099-012-0016-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11099-012-0016-8