Abstract
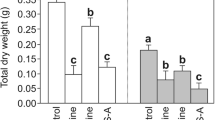
The effects of submergence on chlorophyll (Chl) a fluorescence were compared in seven Oryza sativa (L.) cultivars, namely FR 13A, Khoda, Khadara, Kalaputia (tolerant), Sabita, and Hatipanjari (avoiding type), and IR 42 (susceptible). Seedlings were submerged for 4 d under complete darkness. Oxygen concentration of flood water decreased with the period of submergence with concomitant increase in concentration of carbon dioxide. Submergence caused diminution in the amount of total Chl. Genotypic differences were observed for Chl content and survival percentage. Quantification of the Chl a fluorescence transients (JIP-test) revealed large cultivar differences in the response of photosystem 2 (PS2) to submergence. The kinetics of Chl a fluorescence rise showed complex changes in the magnitudes and rise of O-J, J-I, and I-P phases caused by submergence. The selective suppression of the J-I phase of fluorescence especially after 2 d of submergence provided evidence for weakened electron donation from the oxygen evolving complex whereas under severe submergence stress (4 d) both O-J and J-I steps were suppressed greatly with highly suppressed P-step, which resulted in lowering of variable fluorescence. Grouping probability or energetic connectivity between PS2 obtained through JIP-test from the data after 2 d of submergence showed a direct relation with survival percentage, i.e. fluorescence measurements contained the information of the survival chance of a plant under submerged conditions. The information could be used in identifying the submergence tolerant cultivars when the damage is not very severe.
Similar content being viewed by others
Abbreviations
- area:
-
the space above the fluorescence curve between F0 and Fm
- Chl:
-
chlorophyll
- DAS:
-
days after submergence
- DI0/CS:
-
dissipation of energy in a PS2 cross-section
- ET0/CS:
-
electron transport in a PS2 cross-section
- F0 :
-
minimal fluorescence
- Fm :
-
maximal fluorescence
- Fv (= Fm − F0):
-
variable fluorescence
- Fv/Fm :
-
maximum photochemical efficiency of PS2
- F50µs, F300µs, and F2ms :
-
fluorescence at 50 or 300 µs, and 2 ms, respectively
- OEC:
-
oxygen evolving complex
- PG:
-
grouping probability
- PIABS :
-
performance index on the basis of utilization of absorbed energy
- PQ:
-
plastoquinone
- PS:
-
photosystem
- QA :
-
primary electron acceptor of PS2
- QB :
-
secondary electron acceptor of PS2
- RC/CS0 :
-
number of reaction centres per excited cross-section
- VJ :
-
(F2ms − F0)/(Fm − F0)
References
Aro, E.-M., Virgin, I., Andersson, B.: Photoinhibition of Photosystem II. Inactivation, protein damage and turnover. — Biochim. biophys. Acta 1143: 113–134, 1993.
Calatayud, A., Barreno, E.: Chlorophyll a fluorescence, antioxidant enzymes and lipid peroxidation in tomato in response to ozone and benomyl. — Environ. Pollut. 115: 283–289, 2001.
Crawford, R.M.M., Braendle, R.: Oxygen deprivation stress in a changing climate. — J. exp. Bot. 47: 145–159, 1996.
Das, K.K., Sarkar, R.K.: Post flood changes on the status of chlorophyll, carbohydrate and nitrogen content and its association with submergence tolerance. — Plant Arch. 1: 15–19, 2001.
Dennis, E.S., Dolferus, R., Ellis, M., Rahman, M., Wu, Y., Hoeren, F.U., Grover, A., Ismond, K.P., Good, A.G., Peacock, W.J.: Molecular strategies for improving waterlogging tolerance in plants. — J. exp. Bot. 51: 89–97, 2000.
Drew, M.C.: Oxygen deficiency and root metabolism: injury and acclimation under hypoxia and anoxia. — Annu. Rev. Plant Physiol. Plant mol. Biol. 48: 223–250, 1997.
Force, L., Critchley, C., van Rensen, J.J.S.: New fluorescence parameters for monitoring photosynthesis in plants. 1. The effect of illumination on the fluorescence parameters of the JIP-plants. — Photosynth. Res. 90: 1–19, 2003.
Govindachary, S., Bukhov, N.G., Joly, D., Carpentier, R.: Photosystem II inhibition by moderate light under low temperature in intact leaves of chilling-sensitive and-tolerant plants. — Physiol. Plant. 121: 322–333, 2004.
Guisse, B., Srivastava, A., Strasser, R.J.: Effects of high temperature and water stress on the polyphasic chlorophyll a fluorescence transient of potato leaves. — In: Mathis, P. (ed.): Photosynthesis: From Light to Biosphere. Vol. IV. Pp. 913–916. Kluwer Academic Publ., Dordrecht — Boston — London 1995.
Joliot, P., Joliot, A.: Cyclic electron transport in plant leaf. — Proc. nat. Acad. Sci. USA 99: 10209–10214, 2002.
Lazar, D., Ilik, P.: High-temperature induced chlorophyll fluorescence changes in barley leaves. Comparison of the critical temperatures determined from fluorescence induction and from fluorescence temperature curve. — Plant Sci. 124: 159–164, 1997.
Porra, R.J.: The chequered history of the development and use of simultaneous equations for the accurate determination of chlorophylls a and b. — Photosynth. Res. 73: 149–156, 2002.
Prakash, J.S.S., Srivastava, A., Strasser, R.J., Mohanty, P.: Senescence-induced alternation in the photosystem II functions of Cucumis sativus cotyledons: probing of senescence driven alternation of photosystem II by chlorophyll a fluorescence induction O-J-I-P transients. — Indian J. Biochem. Biophys. 40: 160–168, 2003.
Ramakrishnayya, G., Setter, T.L., Sarkar, R.K., Krishnan, P., Ravi, I.: Influence of P application to floodwater on oxygen concentrations and survival of rice during complete submergence. — Exp. Agr. 35: 167–180, 1999.
Sarkar, R.K.: Saccharide content and growth parameters in relation with flooding tolerance in rice. — Biol. Plant. 40: 597–603, 1998.
Sarkar, R.K., Das, S., Ravi, I.: Changes in certain antioxidative enzymes and growth parameters as a result of complete submergence and subsequent re-aeration of rice cultivars differing in submergence tolerance. — J. Agron. Crop Sci. 187: 69–74, 2001.
Sarkar, R.K., De, R.N., Reddy, J.N., Ramakrishnayya, G.: Studies on the submergence tolerance mechanism in relation to carbohydrate, chlorophyll and specific leaf weight in rice (Oryza sativa L.). — J. Plant Physiol. 149: 623–625, 1996.
Sayed, O.H.: Chlorophyll fluorescence as a tool in cereal crop research. — Photosynthetica 41: 321–330, 2003.
Schreiber, U., Neubauer, C.: The polyphasic rise of chlorophyll fluorescence upon onset of strong continuous illumination: Partial control by the photosystem II donor side and possible ways of interpretation. — Z. Naturforsh. 42c: 1255–1264, 1987.
Setter, T.L., Ramakrishnayya, G., Ram, P.C., Singh, B.B.: Environmental characteristics of floodwater in eastern India: relevance to flooding tolerance of rice. — Indian J. Plant Physiol. 38: 34–40, 1995.
Setter, T.L., Waters, I.: Dissolved gas measurements for experiments on waterlogging and flooding tolerance of plants. — In: Miscellaneous Pubblications 89/1. 2nd Ed. Univ. Western Australia, Nedlands 1990.
Srivastava, A., Guisse, B., Greppin, H., Strasser, R.J.: Regulation of antenna structure and electron transport in Photosystem II of Pisum sativum under elevated temperature probed by the fast polyphasic chlorophyll a fluorescence transient OKJIP. — Biochim. biophys. Acta 1320: 95–106, 1997.
Strasser, B.J., Strasser, R.J.: Measuring fast fluorescence transients to address environmental questions: The JIP-test. — In: Mathis, P. (ed.): Photosynthesis: From Light to Biosphere. Vol. V. Pp. 977–980. Kluwer Academic Publ., Dordrecht — Boston — London 1995.
Strasser, R.J., Srivastava, A., Govindjee: Polyphasic chlorophyll a fluorescence transient in plants and cyanobacteria. — Photochem. Photobiol. 61: 32–42, 1995.
Strasser, R.J., Srivastava, A., Tsimilli-Michael, M.: The fluorescence transient as a tool to characterize and screen photosynthetic samples. — In: Yunus, M., Pathre, U., Mohanty, P. (ed.): Probing Photosynthesis: Mechanisms, Regulation and Adaptation. Pp. 445–483. Tayor and Francis, London — New York 2000.
Strasser, R.J., Stirbet, A.D.: Estimation of the energetic connectivity of PS II centres in plants using the fluorescence rise O-J-I-P. Fitting of experimental data to three different PS II models. — Mathem. Comput. Simulat. 56: 451–461, 2001.
Strasser, R.J., Tsimilli-Michael, M.: Stress in plants, from daily rhythm to global changes, detected and quantified by the JIP-test. — Chimie nouvelle (SRC) 75: 3321–3326, 2001.
Styring, S., Virgin, I., Ehrenerg, A., Andersson, B.: Strong light photoinhibition of electron transport in Photosystem II. Impairment of the function of the first quinone acceptor, QA. — Biochim. biophys. Acta 1015: 269–278, 1990.
van Heerden, P.D.R., Tsimilli-Michael, M., Kruger, G.H., Strasser, R.J.: Dark chilling effects on soybean genotypes during vegetative development; parallel studies of CO2 assimilation, chlorophyll a fluorescence kinetics O-J-I-P and nitrogen fixation. — Physiol. Plant. 117: 476–491, 2003.
Zhang, S., Gao, R.: Diurnal changes of gas exchange, chlorophyll fluorescence, and stomatal aperture of hybrid poplar clones subjected to midday light stress. — Photosynthetica 37: 559–571, 1999.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Panda, D., Rao, D.N., Sharma, S.G. et al. Submergence effects on rice genotypes during seedling stage: Probing of submergence driven changes of photosystem 2 by chlorophyll a fluorescence induction O-J-I-P transients. Photosynthetica 44, 69–75 (2006). https://doi.org/10.1007/s11099-005-0200-1
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11099-005-0200-1