ABSTRACT
Purpose
The purpose of this study was to develop a novel, drug-free therapy that can reduce the over-accumulation of cariogenic bacteria on dental surfaces.
Methods
We designed and synthesized a polyethylene glycol (PEG)-based hydrophilic copolymer functionalized with a pyrophosphate (PPi) tooth-binding anchor using “click” chemistry. The polymer was then evaluated for hydroxyapatite (HA) binding kinetics and capability of reducing bacteria adhesion to artificial tooth surface.
Results
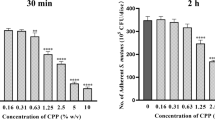
The PPi-PEG copolymer can effectively inhibit salivary protein adsorption after rapid binding to an artificial tooth surface. As a result, the in vitro S. mutans adhesion study showed that the PPi-PEG copolymer can inhibit saliva protein-promoted S. mutans adhesion through the creation of a neutral, hydrophilic layer on the artificial tooth surface.
Conclusions
The results suggested the potential application of a PPi-PEG copolymer as a drug-free alternative to current antimicrobial therapy for caries prevention.




Similar content being viewed by others
Abbreviations
- CFU:
-
Colony forming units
- Click-PEG:
-
Multifunctional polyethylene glycol copolymer synthesized using click chemistry
- DCC:
-
N,N’-dicyclohexylcarbodiimide
- DCM:
-
Dichloromethane
- DMAP:
-
4-dimethylaminopyridine
- DMF:
-
Dimethylformamide
- EDTA:
-
Ethylenediaminetetraacetic acid
- GRAS:
-
Generally regarded as safe
- HA:
-
Hydroxyapatite
- HMHP:
-
PPi-PEG copolymer with high MW and high pyrophosphate content
- HMLP:
-
PPi-PEG copolymer with high MW and low pyrophosphate content
- IRB:
-
Institutional review board
- LMHP:
-
PPi-PEG copolymer with low MW and high pyrophosphate content
- LMLP:
-
PPi-PEG copolymer with low MW and low pyrophosphate content
- MW:
-
Molecular weight
- PDI:
-
Polydispersity index
- PEG:
-
Polyethylene glycol
- PPi:
-
Pyrophosphate
- PPi-PEG:
-
Pyrophosphate modified click-PEG
- S. mutans :
-
Streptococcus mutans
- TBAP:
-
Tris(tetra-n-butylammonium) hydrogen pyrophosphate
- THYE:
-
Todd Hewitt-yeast extract
REFERENCES
Aas JA, Paster BJ, Stokes LN, Olsen I, Dewhirst FE. Defining the normal bacterial flora of the oral cavity. J Clin Microbiol. 2005;43:5721–32.
Faveri M, Mayer MP, Feres M, de Figueiredo LC, Dewhirst FE, Paster BJ. Microbiological diversity of generalized aggressive periodontitis by 16S rRNA clonal analysis. Oral Microbiol Immunol. 2008;23:112–8.
Marsh PD, Moter A, Devine DA. Dental plaque biofilms: communities, conflict and control. Periodontol. 2000;55:16–35.
Baehniand PC, Takeuchi Y. Anti-plaque agents in the prevention of biofilm-associated oral diseases. Oral Dis. 2003;9 Suppl 1:23–9.
Loe H, Theilade E, Jensen SB. Experimental gingivitis in man. J Periodontol. 1965;36:177–87.
Gunsolley JC. A meta-analysis of six-month studies of antiplaque and antigingivitis agents. J Am Dent Assoc. 2006;137:1649–57.
van der Mei HC, White DJ, Atema-Smit J, van de Belt-Gritter E, Busscher HJ. A method to study sustained antimicrobial activity of rinse and dentifrice components on biofilm viability in vivo. J Clin Periodontol. 2006;33:14–20.
Marshand PD, Bradshaw DJ. Dental plaque as a biofilm. J Ind Microbiol. 1995;15:169–75.
Creeth JE, Abraham PJ, Barlow JA, Cummins D. Oral delivery and clearance of antiplaque agents from Triclosan-containing dentifrices. Int Dent J. 1993;43:387–97.
Marsh PD. Are dental diseases examples of ecological catastrophes? Microbiology. 2003;149:279–94.
Marsh PD. Dental plaque as a microbial biofilm. Caries Res. 2004;38:204–11.
Yazdankhah SP, Scheie AA, Hoiby EA, Lunestad BT, Heir E, Fotland TO, et al. Triclosan and antimicrobial resistance in bacteria: an overview. Microb Drug Resist. 2006;12:83–90.
Taubmanand MA, Nash DA. The scientific and public-health imperative for a vaccine against dental caries. Nat Rev Immunol. 2006;6:555–63.
Shimotoyodome A, Koudate T, Kobayashi H, Nakamura J, Tokimitsu I, Hase T, et al. Reduction of Streptococcus mutans adherence and dental biofilm formation by surface treatment with phosphorylated polyethylene glycol. Antimicrob Agents Chemother. 2007;51:3634–41.
Johnston EE, Bryers JD, Ratner BD. Plasma deposition and surface characterization of oligoglyme, dioxane, and crown ether nonfouling films. Langmuir. 2005;21:870–81.
Pidhatika B, Rodenstein M, Chen Y, Rakhmatullina E, Muhlebach A, Acikgoz C, et al. Comparative stability studies of poly(2-methyl-2-oxazoline) and poly(ethylene glycol) brush coatings. Biointerphases. 2012;7:1.
Qi W, Joshi S, Weber CR, Wali RK, Roy HK, Savkovic SD. Polyethylene glycol diminishes pathological effects of Citrobacter rodentium infection by blocking bacterial attachment to the colonic epithelia. Gut Microbes. 2011;2(5):267–73.
Carles J. Colorimetric microdetermination of phosphorus. Bull Soc Chim Biol (Paris). 1956;38:255–7.
Murchison HH, Barrett JF, Cardineau GA, Curtiss 3rd R. Transformation of Streptococcus mutans with chromosomal and shuttle plasmid (pYA629) DNAs. Infect Immun. 1986;54:273–82.
Biswas I, Drake L, Biswas S. Regulation of gbpC expression in Streptococcus mutans. J Bacteriol. 2007;189:6521–31.
Jett BD, Hatter KL, Huycke MM, Gilmore MS. Simplified agar plate method for quantifying viable bacteria. Biotechniques. 1997;23:648–50.
Marsh PD. Controlling the oral biofilm with antimicrobials. J Dent. 2010;38 Suppl 1:S11–5.
Liu XM, Thakur A, Wang D. Efficient synthesis of linear multifunctional poly(ethylene glycol) by copper(I)-catalyzed Huisgen 1,3-dipolar cycloaddition. Biomacromolecules. 2007;8:2653–8.
U.S. FDA Website. http://www.fda.gov.
Hein CD, Liu XM, Chen F, Cullen DM, Wang D. The synthesis of a multiblock osteotropic polyrotaxane by copper(I)-catalyzed huisgen 1,3-dipolar cycloaddition. Macromol Biosci. 2010;10:1544–56.
Kandori K, Oda S, Tsuyama S. Effects of pyrophosphate ions on protein adsorption onto calcium hydroxyapatite. J Phys Chem B. 2008;112:2542–7.
Moreno EC, Kresak M, Hay DI. Adsorption of molecules of biological interest onto hydroxyapatite. Calcif Tissue Int. 1984;36:48–59.
Rosenberg M, Judes H, Weiss E. Cell surface hydrophobicity of dental plaque microorganisms in situ. Infect Immun. 1983;42:831–4.
Shimotoyodome A, Kobayashi H, Tokimitsu I, Matsukubo T, Takaesu Y. Statherin and histatin 1 reduce parotid saliva-promoted Streptococcus mutans strain MT8148 adhesion to hydroxyapatite surfaces. Caries Res. 2006;40:403–11.
Vinogradov AM, Winston M, Rupp CJ, Stoodley P. Rheology of biofilms formed from the dental plaque pathogen Streptococcus mutans. Biofilm. 2004;1:49–56.
Olsson J, Carlen A, Holmberg K. Inhibition of Streptococcus mutans adherence by means of surface hydrophilization. J Dent Res. 1990;69:1586–91.
ACKNOWLEDGMENTS AND DISCLOSURES
This work was supported in part by NIH grants R03 DE019179 (KCR), R01 AI038901 (KWB) and Nebraska Research Initiative (NRI) Proof-of-Concept Award (DW). FC also acknowledges the graduate fellowship support from University of Nebraska Medical Center. The authors of this article declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Fu Chen and Zhenshan Jia contributed equally to this manuscript.
Rights and permissions
About this article
Cite this article
Chen, F., Jia, Z., Rice, K.C. et al. The Development of Drug-Free Therapy for Prevention of Dental Caries. Pharm Res 31, 3031–3037 (2014). https://doi.org/10.1007/s11095-014-1396-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-014-1396-1