ABSTRACT
Aim
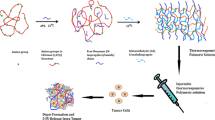
Intravesical instillation is commonly used to decrease the tumor recurrence after transurethral resection. However, most drug solutions would be eliminated from bladder after the first voiding of urine, so its clinical efficacy is limited. To overcome this obstacle, we developed a floating hydrogel system for controlled delivery of antitumor drugs.
Methods
The floating hydrogel was made of Adriamycin, thermo-sensitive polymer (Poloxamer 407) and NH4HCO3, which was liquid at low temperature while forming hydrogel at high temperature. Meanwhile, at high temperature, NH4HCO3 decomposed to produce CO2 bubbles, which helped hydrogel float in bladder without urinary obstruction.
Results
The mixture containing 45% P407 and 6% NH4HCO3 was selected as the optimal formulation. At 37°C, the mixture formed hydrogel and produced many bubbles which could be observed by B ultrasound. The vitro study showed that the antitumor drug Doxorubicin was released in a controlled manner. After the mixture was instilled into rabbit bladder, it formed hydrogel and floated in the bladder. The bladder stimuli was reduced and antitumor drugs could be released continuously in the bladder.
Conclusion
Our results suggested that the floating hydrogel was a feasible intravesical drug delivery system and may have application prospects in intravesical therapy for bladder cancer.








Similar content being viewed by others
REFERENCES
Jemal A, Siegel R, Xu J, Ward E. Cancer statistics, 2010. CA Cancer J Clin. 2010;60:277–300.
Kirkali Z, Chan T, Manoharan M, Algaba F, Busch C, Cheng L, et al. Bladder cancer: epidemiology, staging and grading, and diagnosis. Urology. 2005;66:4–34.
Kurth K, Bouffioux C, Sylvester R, Van der Meijden A, Oosterlinck W, Brausi M. Treatment of superficial bladder tumors: achievements and needs. Eur Urol. 2000;37:1–9.
Gontero P, Cattel L, Paone TC, Milla P, Berta G, Fiorito C, et al. Pharmacokinetic study to optimize the intravesical administration of gemcitabine. BJU Int. 2010;106:1652–6.
Wientjes MG, Badalament RA, Au JL-S. Use of pharmacologic data and computer simulations to design an efficacy trial of intravesical mitomycin C therapy for superficial bladder cancer. Cancer Chemother Pharmacol. 1993;32:255–62.
Lu Z, Yeh T-K, Tsai M, Au JL-S, Wientjes MG. Paclitaxel-loaded gelatin nanoparticles for intravesical bladder cancer therapy. Clin Cancer Res. 2004;10:7677–84.
Burjak M, Bogataj M, Velnar M, Grabnar I, Mrhar A. The study of drug release from microspheres adhered on pig vesical mucosa. Int J Pharm. 2001;224:123–30.
Tyagi P, Li Z, Chancellor M, De Groat WC, Yoshimura N, Huang L. Sustained intravesical drug delivery using thermosensitive hydrogel. Pharm Res. 2004;21:832–7.
Men K, Liu W, Li L, Duan X, Wang P, Gou M, et al. Delivering instilled hydrophobic drug to the bladder by a cationic nanoparticle and thermo-sensitive hydrogel composite system. Nanoscale. 2012;4:6425–33.
Gou M, Li X, Dai M, Gong C, Wang X, Xie Y, et al. A novel injectable local hydrophobic drug delivery system: biodegradable nanoparticles in thermo-sensitive hydrogel. Int J Pharm. 2008;359:228–33.
Collado A, Chechile G, Salvador J, Vicente J. Early complications of endoscopic treatment for superficial bladder tumors. J Urol. 2000;164:1529–32.
Veyries ML, Couarraze G, Geiger S, Agnely F, Massias L, Kunzli B, et al. Controlled release of vancomycin from poloxamer 407 gels. Int J Pharm. 1999;192:183–93.
Paavola A, Kilpelainen I, Yliruusi J, Rosenberg P. Controlled release injectable liposomal gel of ibuprofen for epidural analgesia. Int J Pharm. 2000;199:85–93.
Chung HJ, Lee Y, Park TG. Thermo-sensitive and biodegradable hydrogels based on stereocomplexed Pluronic multi-block copolymers for controlled protein delivery. J Control Release. 2008;127:22–30.
Min B, Bae IY, Lee HG, Yoo S-H, Lee S. Utilization of pectin-enriched materials from apple pomace as a fat replacer in a model food system. Bioresour Technol. 2010;101:5414–8.
Yehand AC, Bai H. Comparison of ammonia and monoethanolamine solvents to reduce CO2 greenhouse gas emissions. Sci Total Environ. 1999;228:121–33.
Sharmaand PK, Bhatia SR. Effect of anti-inflammatories on Pluronic® F127: micellar assembly, gelation and partitioning. Int J Pharm. 2004;278:361–77.
Dumortier G, Grossiord JL, Agnely F, Chaumeil JC. A review of poloxamer 407 pharmaceutical and pharmacological characteristics. Pharm Res. 2006;23:2709–28.
Ricci E, Lunardi L, Nanclares D, Marchetti J. Sustained release of lidocaine from Poloxamer 407 gels. Int J Pharm. 2005;288:235–44.
Lin T, Wu J, Zhao X, Lian H, Yuan A, Tang X, et al. In situ floating hydrogel for intravesical delivery of adriamycin without blocking urinary tract. J Pharm Sci. 2014;103:927–36.
Wangand P, Johnston T. Kinetics of sol‐to‐gel transition for poloxamer polyols. J Appl Polym Sci. 1991;43:283–92.
Alexandridisand P, Alan Hatton T. Poly(ethylene oxide)-poly(propylene oxide)-poly(ethylene oxide) block copolymer surfactants in aqueous solutions and at interfaces: thermodynamics, structure, dynamics, and modeling. Colloids Surf A Physicochem Eng Asp. 1995;96:1–46.
Zhang L, Parsons DL, Navarre C, Kompella UB. Development and in-vitro evaluation of sustained release Poloxamer 407 (P407) gel formulations of ceftiofur. J Control Release. 2002;85:73–81.
Costaand P, Sousa Lobo JM. Modeling and comparison of dissolution profiles. Eur J Pharm Sci. 2001;13:123–33.
ACKNOWLEDGMENTS AND DISCLOSURES
This paper was supported by Research Fund for the Doctoral Program of Higher Education of China (No. 20110091120044), Natural Science Foundation of Jiangsu BK2011572 and BK2011539, National Natural Science Foundation (No. 81202474, 30973651, 81171786), Changzhou Special Project of Biotechnology and Biopharmacy (No. CE20105006) Postdoctoral Foundation (2012M521051).
Tingsheng Lin and Yifan Zhang contributed equally to the article.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
(WMV 8802 kb)
Rights and permissions
About this article
Cite this article
Lin, T., Zhang, Y., Wu, J. et al. A Floating Hydrogel System Capable of Generating CO2 Bubbles to Diminish Urinary Obstruction After Intravesical Instillation. Pharm Res 31, 2655–2663 (2014). https://doi.org/10.1007/s11095-014-1362-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-014-1362-y