Abstract
Purpose
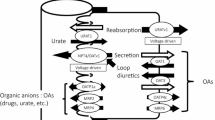
To assess the contribution of organic anion transporting polypeptide 1a5 (Oatp1a5/Oatp3) in the intestinal absorption of an orally active endothelin receptor antagonist, (+)-(5S,6R,7R)-2-butyl-7-[2-((2S)-2-carboxypropyl)-4-methoxyphenyl]-5-(3,4-methylene-dioxyphenyl)cyclopenteno[1,2-b]pyridine-6-carboxylic acid (compound-A) in rats.
Methods
Uptakes of [14C]compound-A by Oatp1a5-expressing Xenopus laevis oocytes and isolated rat enterocytes were evaluated.
Results
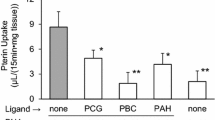
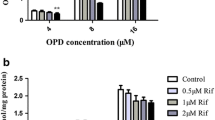
The uptake of compound-A by Oatp1a5-expressing oocytes was significantly higher than that by water-injected oocytes and Oatp1a5-mediated uptake was saturable with K m value of 116 μM. Compound-A was taken up into isolated enterocytes in time- and concentration-dependent manners and the estimated K m value was 83 μM, which was close to that for the Oatpt1a5-mediated uptake in oocytes. Both uptakes of compound-A by Oatp1a5-expressing oocytes and enterocytes were pH-sensitive with significantly higher uptake at acidic pH than those at neutral pH. Uptakes of compound-A into Oatp1a5-expressing oocytes and enterocytes were significantly decreased in the presence of Oatp1a5 substrates such as bromosulfophthalein and taurocholic acid.
Conclusions
These results consistently suggested that Oatp1a5 is contributing to the intestinal absorption of compound-A at least in part, and the transporter-mediated absorption seems to be maximized at the acidic microenvironment of epithelial cells in the small intestine in rats.









Similar content being viewed by others
Abbreviations
- BSP:
-
bromosulfophthalein
- Caco-2:
-
human colon carcinoma cell line
- cRNA:
-
complementary ribonucleic acid
- ET:
-
endothelin
- HEPES:
-
N-2-hydroxyethylpiperazine-N′-2-ethanesulfonic acid
- K m :
-
Michaelis–Menten constant
- k ns :
-
nonsaturable uptake clearance
- LLC-PK1:
-
porcine kidney epithelial cell
- MES:
-
2-N-morpholinoethanesulfonic acid
- OATP/Oatp:
-
organic anion transporting polypeptide
- TCA:
-
taurocholic acid
- V max :
-
maximum uptake rate
References
A. Tsuji, and I. Tamai. Carrier-mediated intestinal transport of drugs. Pharm. Res 13:963–977 (1996).
T. Katsura, and K. Inui. Intestinal absorption of drugs mediated by drug transporters: mechanisms and regulation. Drug Metab. Pharmacokinet 18:1–15 (2003).
I. Tamai, J. Nezu, H. Uchino, Y. Sai, A. Oku, M. Shimane, and A. Tsuji. Molecular identification and characterization of novel members of the human organic anion transporter (OATP) family. Biochem. Biophys. Res. Commun 273:251–260 (2000).
B. Hagenbuch, and P.J. Meier. Organic anion transporting polypeptides of the OATP/SLC21 family: phylogenetic classification as OATP/SLCO superfamily, new nomenclature and molecular/functional properties. Pflugers Arch 447:653–665 (2004).
D. Kobayashi, T. Nozawa, K. Imai, J. Nezu, A. Tsuji, and I. Tamai. Involvement of human organic anion transporting polypeptide OATP-B (SLC21A9) in pH-dependent transport across intestinal apical membrane. J. Pharmacol. Exp. Ther 306:703–708 (2003).
T. Nozawa, K. Imai, J. Nezu, A. Tsuji, and I. Tamai. Functional characterization of pH-sensitive organic anion transporting polypeptide OATP-B in human. J. Pharmacol. Exp. Ther 308:438–445 (2004).
A. Kikuchi, T. Nozawa, T. Wakasawa, T. Maeda, and I. Tamai. Transporter-mediated intestinal absorption of fexofenadine in rats. Drug Metab. Pharmacokinet 21:308–314 (2006).
T. Maeda, K. Takahashi, N. Ohtsu, T. Oguma, T. Ohnishi, R. Atsumi, and I. Tamai. Identification of influx transporter for the quinolone antibacterial agent levofloxacin. Mol. Pharm 4:85–94(2007).
G. K. Dresser, D. G. Bailey, B. F. Leake, U. I. Schwarz, P. A. Dawson, D. J. Freeman, and R. B. Kim. Fruit juices inhibit organic anion transporting polypeptide-mediated drug uptake to decrease the oral availability of fexofenadine. Clin. Pharmacol. Ther 71:11–20 (2002).
H. Glaeser, D. G. Bailey, G. K. Dresser, J. C. Gregor, U. I. Schwarz, J. S. McGrath, E. Jolicoeur, W. Lee, B. F. Leake, R. G. Tirona, and R. B. Kim. Intestinal drug transporter expression and the impact of grapefruit juice in humans. Clin. Pharmacol. Ther 81:362–370 (2007).
M. Nishikibe, H. Ohta, M. Okada, K. Ishikawa, T. Hayama, T. Fukuroda, K. Noguchi, M. Saito, T. Kanoh, S. Ozaki, T. Kamei, K. Hara, D. William, S. Kivlighn, S. Krause, R. Gabel, G. Zingaro, N. Nolan, J. O’brien, F. Clayton, J. Lynch, D. Pettibone, and P. Siegl. Pharmacological properties of J-104132 (L-753,037), a potent, orally active, mixed ETA/ETB endothelin receptor antagonist. J. Pharmacol. Exp. Ther 289:1262–1270 (1999).
N. Kobayashi, T. Tani, A. Hisaka, K. Hara, and T. Yasumori. Hepatobiliary transport of a nonpeptidic endothelin antagonist, (+)-(5S,6R,7R)-2-butyl-7-[2((2S)-2-carboxypropyl)-4-methoxy phenyl]5-(3,4-methylenedioxyphenyl)cyclopentenol[1,2-b]pyridine-6-carboxylic acid: uptake by isolated rat hepatocytes and canalicular membrane vesicles. Pharm. Res 20:89–95 (2003).
H. C. Walters, A. L. Craddock, H. Fusegawa, M. C. Willingham, and P. A. Dawson. Expression, transport properties, and chromosomal location of organic anion transporter subtype 3. Am. J. Physiol 279:G1188–G1200 (2000).
V. Cattori, J. E. Van Montfoort, B. Stieger, L. Landmann, D. K. F. Meijer, K. H. Winterhalter, P. J. Meier, and B. Hagenbuch. Localization of organic anion transporting polypeptide 4 (Oatp4) in rat liver and comparison of its substrate specificity with Oatp1, Oatp2 and Oatp3. Pflugers Arch 443:188–195 (2001).
G. A. Kimmich. Preparation and properties of mucosal epithelial cells isolated from small intestine of the chicken. Biochemistry 9:3659–3669 (1970).
K. Yamaoka, Y. Tanigawara, T. Nakagawa, and T. Uno. A pharmacokinetic analysis program (multi) for microcomputer. J Pharmacobiodyn 4:879–885 (1981).
M. Lucus. Determination of acid surface pH in vivo in rat proximal jejunum. Gut 24:734–739 (1983).
T. Nishio, H. Adachi, R. Nakagomi, T. Tokui, E. Sato, M. Tanemoto, K. Fujiwara, M. Okabe, T. Onogawa, T. Suzuki, D. Nakai, K. Shiiba, M. Suzuki, H. Ohtani, Y. Kondo, M. Unno, S. Ito, K. Iinuma, K. Nunoki, S. Matsuno, and T. Abe. Molecular identification of a rat novel organic anion transporter moat1, which transports prostaglandin D2, leukotriene C4, and taurocholate. Biochem. Biophys. Res. Commun 275:831–837 (2000).
D. T. Thwaites and C. M. Anderson. H-coupled nutrient, micronutrient and drug transporters in the mammalian small intestine. Exp. Physiol. 92:603–619 (2007).
I. Tamai, T. Nakanishi, K. Hayashi, T. Terao, Y. Sai, K. Miyamoto, E. Takeda, H. Higashida, and A. Tsuji. The predominant contribution of oligopeptide transporter PepT1 to intestinal absorption of beta-lactam antibiotics in the rat small intestine. J. Pharm. Pharmacol 49:796–801 (1997).
T. Nozawa, S. Sugiura, Y. Hashino, A. Tsuji, and I. Tamai. Role of anion exchange transporter PAT1 (SLC26A6) in intestinal absorption of organic anions. J. Drug Target 12:97–104 (2004).
I. Tamai, Y. Sai, A. Ono, Y. Kido, H. Yabuuchi, H. Takanaga, E. Satoh, T. Ogihara, O. Amano, S. Izeki, and A. Tsuji. Immunohistochemical and functional characterization of pH-dependent intestinal absorption of weak organic acids by the monocarboxylic acid transporter MCT1. J. Pharm. Pharmacol 51:1113–1121 (1999).
Acknowledgements
The Drug Research Academy at University of Copenhagen are acknowledged for financial support, as well as Brorsons travelfund and OTICON foundation are acknowledged for funding external tuition fee and hence enable the collaboration behind this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tani, T., Gram, L.K., Arakawa, H. et al. Involvement of Organic Anion Transporting Polypeptide 1a5 (Oatp1a5) in the Intestinal Absorption of Endothelin Receptor Antagonist in Rats. Pharm Res 25, 1085–1091 (2008). https://doi.org/10.1007/s11095-007-9472-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-007-9472-4