Purpose
The aim of the study is to determine the bioactivity and effects of PEGylation on the pharmacokinetics in rabbit aqueous humor and plasma of an aptamer directed against TGFβ2.
Methods
Pharmacological activity of anti-TGFβ2 aptamer in rabbit ocular fluid was demonstrated using a mink lung epithelial cell proliferation assay. For pharmacokinetic analyses, concentrations of aptamers in plasma and aqueous humor were determined over time following bilateral subconjunctival administration to Dutch-belted rabbits using a hybridization-based pseudo-enzyme-linked immunosorbent assay (ELISA) assay.
Results
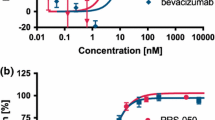
Anti-TGFβ2 aptamer (ARC81) binds to human TGFβ2 with a KD of approximately 5 nM and inhibits the activity of human TGFβ2 in vitro in a cell-based assay with an IC50 of approximately 100 nM. ARC81 blocks endogenously derived TGFβ2 in rabbit aqueous humor in vitro with an IC50 of approximately 200 nM and an IC90 of approximately 1 μM. In vivo in rabbit, ARC81 [no polyethylene glycol (PEG)] entered systemic circulation rapidly (tmax = 1 h in plasma) relative to aptamer conjugates ARC117 (20 kDa PEG) and ARC119 (40 kDa PEG), which showed prolonged residence in the subconjunctival space and aqueous compartment (tmax = 6 and 12 h, respectively, in plasma). Both 20- and 40-kDa aptamer conjugates reached maximal concentrations (Cmax) in aqueous humor of 23–30 nM and remained at or above 1 nM for as long as 12 h.
Conclusions
Pharmacologically active levels of anti-TGFβ2 aptamers can be sustained in the ocular fluid and local tissue environment over a 12-h period after single administration. Daily subconjunctival administration of PEGylated anti-TGFβ2 aptamers should allow further pharmacological evaluation of these agents in a rabbit conjunctival scarring model. Perioperative administration, via subconjunctival injection, may prove to be an effective means to deliver therapeutic quantities of TGFβ2 aptamer conjugates in trabeculectomy procedures.








Similar content being viewed by others
Abbreviations
- AUC:
-
area under the concentration vs. time curve
- C max :
-
highest concentration observed
- MLEC:
-
mink lung epithelial cell
- NCA:
-
noncompartmental analysis
- PEG:
-
polyethylene glycol
- SELEX:
-
systematic evolution of ligands by exponential enrichment
- t 1/2 :
-
terminal half–life
- t max :
-
time at which the highest concentration occurred
- V d :
-
volume of distribution
References
C. Verges J. Cazal C. Lavin (2005) ArticleTitleSurgical strategies in patients with cataract and glaucoma Curr Opin Ophthalmol 16 44–52 Occurrence Handle10.1097/00055735-200502000-00008 Occurrence Handle15650579
R. Bindlish G. P. Condon J. D. Schlosser J. D'Antonio K. B. Lauer R. Lehrer (2002) ArticleTitleEfficacy and safety of mitomycin-C in primary trabeculectomy: five-year follow-up Ophthalmology 109 1336–1341 Occurrence Handle10.1016/S0161-6420(02)01069-2 Occurrence Handle12093659
D. A. Belyea J. A. Dan R. L. Stamper M. F. Lieberman W. H. Spencer (1997) ArticleTitleLate onset of sequential multifocal bleb leaks after glaucoma filtration surgery with 5-fluorouracil and mitomycin C Am J Ophthalmol 124 40–45 Occurrence Handle1:STN:280:ByiA28rjvFA%3D Occurrence Handle9222231
T. H. Kupin M. S. Juzych D. H. Shin A. K. Khatana M. M. Olivier (1995) ArticleTitleAdjunctive mitomycin C in primary trabeculectomy in phakic eyes Am J Ophthalmol 119 30–39 Occurrence Handle1:STN:280:ByqC38nit1M%3D Occurrence Handle7825687
W. L. Membrey D. P. Poinoosawmy C. Bunce F. W. Fitzke R. A. Hitchings (2000) ArticleTitleComparison of visual field progression in patients with normal pressure glaucoma between eyes with and without visual field loss that threatens fixation Br J Ophthalmol 84 1154–1158 Occurrence Handle1:STN:280:DC%2BD3M%2FgvFGhtA%3D%3D Occurrence Handle11004102
Y. Ochiai H. Ochiai (2002) ArticleTitleHigher concentration of transforming growth factor-beta in aqueous humor of glaucomatous eyes and diabetic eyes Jpn J Ophthalmol 46 249–253 Occurrence Handle10.1016/S0021-5155(01)00523-8 Occurrence Handle1:CAS:528:DC%2BD38XksVahsbk%3D Occurrence Handle12063033
M. F. Cordeiro M. B. Reichel J. A. Gay F. D'Esposita R. A. Alexander P. T. Khaw (1999) ArticleTitleTransforming growth factor-beta1, -beta2, and -beta3 in vivo: effects on normal and mitomycin C-modulated conjunctival scarring Investig Ophthalmol Vis Sci 40 1975–1982 Occurrence Handle1:STN:280:DyaK1Mzms1ylsQ%3D%3D
M. F. Cordeiro J. A. Gay P. T. Khaw (1999) ArticleTitleHuman anti-transforming growth factor-beta2 antibody: a new glaucoma anti-scarring agent Investig Ophthalmol Vis Sci 40 2225–2234 Occurrence Handle1:STN:280:DyaK1MvgsVCgtQ%3D%3D
M. F. Cordeiro A. Mead R. R. Ali R. A. Alexander S. Murray C. Chen C. York-Defalco N. M. Dean G. S. Schultz P. T. Khaw (2003) ArticleTitleNovel antisense oligonucleotides targeting TGF-beta inhibit in vivo scarring and improve surgical outcome Gene Ther 10 59–71 Occurrence Handle10.1038/sj.gt.3301865 Occurrence Handle1:CAS:528:DC%2BD3sXivVyksA%3D%3D Occurrence Handle12525838
C. Tuerk L. Gold (1990) ArticleTitleSystematic evolution of ligands by exponential enrichment: RNA ligands to bacteriophage T4 DNA polymerase Science 249 505–510 Occurrence Handle1:CAS:528:DyaK3cXlt1OltLY%3D Occurrence Handle2200121
W. A. Pieken D. B. Olsen F. Benseler H. Aurup F. Eckstein (1991) ArticleTitleKinetic characterization of ribonuclease-resistant 2′-modified hammerhead ribozymes Science 253 314–317 Occurrence Handle1:CAS:528:DyaK3MXlslyjtL0%3D Occurrence Handle1857967
J. M. Healy S. D. Lewis M. Kurz R. M. Boomer K. T. Thompson C. Wilson T. G. McCauley (2004) ArticleTitlePharmacokinetics and biodistribution of novel aptamer compositions Pharm Res 21 2234–2246 Occurrence Handle10.1007/s11095-004-7676-4 Occurrence Handle1:CAS:528:DC%2BD2MXjslyjsA%3D%3D Occurrence Handle15648255
S. R. Watson Y. F. Chang D. O'Connell L. Weigand S. Ringquist D. H. Parma (2000) ArticleTitleAnti-L-selectin aptamers: binding characteristics, pharmacokinetic parameters, and activity against an intravascular target in vivo Antisense Nucleic Acid Drug Dev 10 63–75 Occurrence Handle1:CAS:528:DC%2BD3cXivVKlt7s%3D Occurrence Handle10805157
N. Pagratis, M. Lochrie, and L. Gold. High affinity TGFβ nucleic acid ligands and inhibitors, Gilead Sciences, Inc., USA, United States Patent No. 63,466,111, 2002.
P. E. Burmeister S. D. Lewis R. F. Silva J. R. Preiss L. R. Horwitz P. S. Pendergrast T. G. McCauley J. C. Kurz D. M. Epstein C. Wilson A. D. Keefe (2005) ArticleTitleDirect in vitro selection of a 2′-O-methyl aptamer to VEGF Chem Biol 12 25–33 Occurrence Handle10.1016/j.chembiol.2004.10.017 Occurrence Handle1:CAS:528:DC%2BD2MXmtlKnsQ%3D%3D Occurrence Handle15664512
M. Gibaldi D. Perrier (1982) Pharmacokinetics Marcel Dekker Inc. New York
J. C. Jennings S. Mohan T. A. Linkhart R. Widstrom D. J. Baylink (1988) ArticleTitleComparison of the biological actions of TGF beta-1 and TGF beta-2: differential activity in endothelial cells J Cell Physiol 137 167–172 Occurrence Handle10.1002/jcp.1041370120 Occurrence Handle1:CAS:528:DyaL1cXmt1egtL0%3D Occurrence Handle3170656
S. W. Cousins M. M. McCabe D. Danielpour J. W. Streilein (1991) ArticleTitleIdentification of transforming growth factor-beta as an immunosuppressive factor in aqueous humor Investig Ophthalmol Vis Sci 32 2201–2211 Occurrence Handle1:STN:280:By6A3c7pvVU%3D
A. L. Mead T. T. Wong M. F. Cordeiro I. K. Anderson P. T. Khaw (2003) ArticleTitleEvaluation of anti-TGF-beta2 antibody as a new postoperative anti-scarring agent in glaucoma surgery Investig Ophthalmol Vis Sci 44 3394–3401 Occurrence Handle10.1167/iovs.02-0978
Acknowledgements
The authors thank Dr. Sandra Dennis and Dr. John Nash at CTBR-BioResearch Inc. (Montreal, Quebec, Canada) for support of this study and Dr. Judy Healy (Archemix Corp., Cambridge, MA, USA) for help with preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
McCauley, T.G., Kurz, J.C., Merlino, P.G. et al. Pharmacologic and Pharmacokinetic Assessment of Anti-TGFβ2 Aptamers in Rabbit Plasma and Aqueous Humor. Pharm Res 23, 303–311 (2006). https://doi.org/10.1007/s11095-005-9305-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-005-9305-2