No Heading
Purpose.
The purpose of this research was to characterize nipecotic acid pharmacokinetics in blood and brain after intravenous (i.v.) and nasal administration of nipecotic acid and its n-butyl ester.
Methods.
Nipecotic acid and its n-butyl ester were administered to rats i.v. and intranasally (n = 5 rats/drug per route), and nipecotic acid pharmacokinetics in blood were characterized. Nipecotic acid concentration-time profiles were determined in blood by noncompartmental and compartmental methods. Nipecotic acid was also dosed i.v. and its n-butyl ester was dosed by nasal and i.v. routes, and brain levels of nipecotic acid over the subsequent 4 h (n = 5 rats/time point per route) were assessed.
Results.
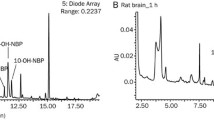
The absolute systemic availability of nipecotic acid after nasal dosing was 14%. After i.v. and nasal dosing of the n-butyl ester, nipecotic acid systemic availability was 97% and 92%, respectively. Both i.v. and nasal administration of the n-butyl ester resulted in a significantly longer terminal half-life and larger mean resident time and volume of distribution for nipecotic acid than was observed after an i.v. nipecotic acid dose. Total brain exposure to nipecotic acid was not significantly different after nasal and i.v. dosing of the n-butyl ester. However, the brain/blood nipecotic acid ratio declined significantly with time after i.v. and nasal dosing of the ester prodrug. Nipecotic acid was not detectable in brain after i.v. dosing of nipecotic acid.
Conclusions.
The use of an ester formulation was crucial to delivering nipecotic acid to the brain. Preliminary evidence strongly suggests ester hydrolysis is rate limiting to nipecotic acid brain delivery. Once nipeoctic acid was formed, it displayed tissue trapping in brain. Parenteral dosing of nipecotic acid esters is unnecessary for systemic or brain delivery of nipecotic acid and possibly other CNS active zwitterion esters.
Similar content being viewed by others
References
1. R. J. Porter. Therapy of epilepsy. Curr. Opin. Neurol. Neurosurg. 1:206–211 (1988).
2. G. A. Baker, A. Jacoby, D. Buck, C. Stalgis, and D. Monnet. Quality of life of people with epilepsy: a European study. Epilepsia 38:353–362 (1997).
3. A. C. Hui, G. M. Joynt, H. Li, and K. S. Wong. Status epilepticus in Hong Kong Chinese: aetiology, outcome and predictors of death and morbidity. Seizure 12:478–482 (2003).
4. L. Illum. Nasal drug delivery - possibilities, problems and solutions. J. Control. Rel. 87:187–198 (2003).
5. A. Hussain. Intranasal drug delivery. Adv. Drug Deliv. Rev. 29:39–49 (1998).
6. L. Li, S. Gorukanti, Y. M. Choi, and K. H. Kim. Rapid-onset intranasal delivery of anticonvulsants: pharmacokinetic and pharmacodynamic evaluation in rabbits. Int. J. Pharm. 199:65–76 (2000).
7. A. M. Crider, T. T. Tita, J. D. Wood, and C. N. Hinko. Esters of nipecotic acid and isonipecotic acids as potential anticonvulsants. J. Pharm. Sci. 71:1212–1219 (1982).
8. A. M. Crider, J. D. Wood, K. D. Tschappat, C. N. Hinko, and K. Seibert. Gamma-aminobutyric acid uptake inhibition and anticonvulsant activity of nipecotic acid esters. J. Pharm. Sci. 73:1612–1616 (1984).
9. C. N. Hinko, A. M. Crider, and J. D. Wood. A comparison of prodrug esters of nipecotic acid. Neuropharmacol 27:475–483 (1988).
10. C. N. Hinko, K. Seibert, and A. M. Crider. Anticonvulsant activity of the nipecotic acid ester, (+/−)-n-nitrophenyl-3-piperidine-carbooxylate. Neuropharmacol. 23:1009–1014 (1984).
11. S. Manfredini, B. Pavan, S. Vertuani, M. Scaglianti, D. Compagnone, C. Biondi, A. Scatturin, S. Tanganelli, L. Ferraro, P. Prasad, and A. Dalpiaz. Design, synthesis and activity of ascorbic acid prodrugs of nipecotic, kynurenic and diclophenamic acid, liable to increase neurotropic activity. J. Med. Chem. 45:559–562 (2002).
12. H. H. Frey, C. Popp, and W. Loscher. Influence of inhibitors of the high affinity GABA uptake on seizure thresholds in mice. Neuropharmacol. 18:581–590 (1979).
13. F. P. Bonina, L. Arenare, F. Palagiano, A. Saija, F. Nava, D. Trombetta, and P. de Caprariis. Synthesis, stability, and pharmacological evaluation of nipecotic acid prodrugs. J. Pharm. Sci. 88:561–567 (1999).
14. L. J. S. Knutsen, K. E. Andersen, J. Lau, B. F. Lundt, R. F. Henry, H. E. Morton, L. Naerum, H. Petersen, H. Stephensen, P. D. Suzdak, M. D. B. Swedberg, C. Thomsen, and P. O. Sorensen. Synthesis of novel GABA uptake inhibitors. 3. Diaryloxime and diarylvinyl ether derivatives of nipecotic acid and guvacine as anticonvulsant agents. J. Med. Chem. 42:3447–3462 (1999).
15. K. E. Andersen, J. L. Sorensen, J. Lau, B. F. Lundt, H. Petersen, P. O. Huusfeldt, P. D. Suzdak, and M. D. B. Swedberg. Synthesis of novel γ-aminobutryic acid (GABA) uptake inhibitors. 5. Preparation and structure-activity studies of tricyclic analogues of known GABA uptake inhibitors. J. Med. Chem. 44:2152–2163 (2001).
16. K. E. Andersen, J. Lau, B. F. Lundt, H. Petersen, P. O. Huusfeldt, P. D. Suzdak, and M. D. B. Swedberg. Synthesis of novel GABA uptake inhibitors, Part 6: preparation and evaluation of N-Ω asymmetrically substituted nipecotic acid derivatives. Biorganic Med. Chem. 9:2773–2785 (2001).
17. H. Wang. 2003. The brain delivery of nipecotic acid by the administration of its ester prodrug via the nasal route: Physicochemical Properties. Ph.D. Thesis, University of Kentucky, Lexington, KY, pp. 71–85.
18. S. Hirai, T. Yashika, T. Matsuzawa, and H. Mima. Absorption of drugs from the nasal mucosa of rat. Int. J. Pharm. 7:317–325 (1981).
19. P. J. Wedlund, S. L. Chang, and R. H. Levy. Steady-state approach to examination of pulmonary metabolism: application to carbamazepine. J. Pharm. Sci. 2:860–862 (1983).
20. S. L. Chang, K. Emmick, and P. J. Wedlund. Characterization of antipyrine autoinduction in rat utilizing a new microsampling technique for serial blood sample collection. J. Pharm. Sci. 74:456–458 (1986).
21. H. Wang, A. A. Hussain, J. S. Pyrek, J. Goodman, and P. J. Wedlund. Assay for nipecotic acid in small blood samples by gas chromatography-mass spectroscopy. J. Pharm. Biomed. Anal. 34/35:1063–1070 (2004).
22. K. S. Pang, W. F. Cherry, J. A. Terrell, and E. H. Ulm. Disposition of enalapril and its diacid metabolite, enalaprilat, in a perfused rat liver preparation. Presence of a diffusional barrier for enalaprilat into hepatocytes. Drug Metab. Dispos. 12:309–313 (1984).
23. T. Haradahira, M. Maeda, A. Kato, Y. Kanazawa, M. Yamada, Y. Torii, Y. Ichiya, and K. Masuda. Metabolic pathway of 2-deoxy-2-[18F]fluoro-D-talose in mice: trapping in tissue after phosphorylation by galactokinase. Nucl. Med. Biol. 21:269–276 (1994).
24. A. M. Evans. Membrane transport as a determinant of the hepatic elimination of drugs and metabolites. Clin. Exp. Pharmacol. Physiol. 23:970–974 (1996).
25. H. D. Kao, A. Traboulsi, S. Itoh, L. Dittert, and A. Hussain. Enhancement of the systemic and CNS specific delivery of L-DOPA by the nasal administration of its water soluble prodrugs. Pharm. Res. 17:978–984 (2000).
26. T. C. A. Kumar, G. F. X. David, A. Sankaranarayanan, V. Puri, and K. R. Sundram. Pharmacokinetics of progesterone after its administration to ovariectomized rhesus monkeys by injection, infusion, or nasal spray. Proc. Natl. Acad. Sci. USA 79:4185–4189 (1982).
27. A. Czerniawska. Experimental investigations on the penetration of 198Au from nasal mucous membrane into cerebrospinal fluid. Acta Otolaryng. 70:58–60 (1970).
28. T. Sakane, M. Akizuki, M. Yoshida, S. Yamashita, T. Nadai, M. Hashida, and H. Sezaki. Transport of cephalexin to the cerebrospinal fluid directly from the nasal cavity. J. Pharm. Pharmacol. 43:449–451 (1991).
29. H. H. S. Chow, Z. Chen, and G. T. Matsuura. Direct transport of cocaine from the nasal cavity to the brain following intranasal cocaine administration in rats. J. Pharm. Sci. 88:754–758 (1999).
30. B. A. Kakee, A. H. Takanaga, H. T. Terasaki, T. M. Naito, M. T. Tsuruo, and Y. Sugiyama. Efflux of a suppressive neurotransmitter, GABA, across the blood-brain barrier. J. Neurochem. 79:110–118 (2001).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, H., Hussain, A. & Wedlund, P. Nipecotic Acid: Systemic Availability and Brain Delivery After Nasal Administration of Nipecotic Acid and n-Butyl Nipecotate to Rats. Pharm Res 22, 556–562 (2005). https://doi.org/10.1007/s11095-005-2491-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-005-2491-0