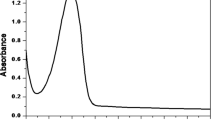
Nifedipine (NIF) is a poorly water-soluble and a high photosensitive drug, which is among most efficient drugs for treating diseases such as hypertension and angina pectoris. The main purpose of this work was the preparation and study of the inclusion complex between NIF and ethylenediamine-β-cyclodextrin (NIF/EDA-CD). The NIF/EDA-CD complex has been prepared using the co-precipitation method and characterized by UV-Vis spectroscopy, dynamic light scattering (DLS), differential scanning calorimetry (DSC), thermogravimetric (TGA), and Fourier-transform infrared (FT-IR) spectroscopy in both solution and solid state. The 1 : 1 molar stoichiometry is confirmed by the continuous-variation Job’s plot and phase solubility diagram. The phase solubility diagram showed a complexation constant of 1019.62 M-1. In addition, supplementary data from solubility tests have demonstrated that the solubility of NIF in water greatly increased through complexation with EDA-CD, which is a result of the water-soluble EDA-CD host.








Similar content being viewed by others
References
R. P. Patel and M. M. Patel, Dhaka Univ. J. Pharm. Sci., 6, 25 – 36 (2007).
J. A. Squella, E. Barnafi, S. Perna, et al., Talanta, 36, 363 – 366 (1989).
K. Kothari, V. Ragoonanan, and R. Suryanarayanan, Mol. Pharm., 12, 1477 – 1484 (2015).
T. Higuchi and K. Connors, Adv. Anal. Chem. Instrum., 4, 117 – 212 (1961).
T. Tao, Y. Zhao, J. Wu, et al., Intern. J. Pharm., 367, 109 – 114 (2009).
P. J. Marsac, T. Li, and L. S. Taylor, Pharm. Res., 26, 139 – 151 (2009).
I. Ghosh, S. Bose, R. Vippagunta, et al., Intern. J. Pharm., 409, 260 – 268 (2011).
V. Patel and J. Sarai, Indian J. Pharm. Sci., 76, 483 (2014).
V. J. Stella and K. W. Nti-Addae, Adv. Drug Deliv. Rev., 59, 677 – 694 (2007).
M. Di Cagno, T. T. Nielsen, K. L. Larsen, et al., Intern. J. Pharm., 468, 258 – 263 (2014).
H. Namazi and A. Heydari, Polym. Intern., 63, 1447 – 1455 (2014).
T. Loftsson and M. E. Brewster, J. Pharm. Sci., 85, 1017 – 25 (1996).
G. Castronuovo and M. Niccoli, Bioorg. Med. Chem., 14, 3883 – 3887 (2006).
J. Szejtli, Carbohydr. Polym., 12, 375 – 392 (1990).
S. Hbaieb, Mater. Sci. Eng. C, 28, 697 – 704 (2008).
S. Menuel, B. Doumert, S. Saitzek, et al., J. Org Chem., 80, 6259 – 6266 (2015).
T. Irie and K. Uekama, J. Pharm. Sci., 86, 147 – 62 (1997).
H. Namazi, F. Fathi, and A. Heydari, Nanoparticles Based on Modified Polysaccharides, in The Delivery of Nanoparticles, A. Hashim (Ed.), InTech (2012), pp. 149 – 184.
H. Namazi, A. Heydari, and A. Pourfarzolla, Intern. J. Polym. Mater. Polym. Biomater., 63, 1 – 6 (2013).
C. Conte, G. Costabile, I. d’Angelo, et al., J. Colloiod. Interface Sci., 454, 112 – 120 (2015).
M. Peppas, Y. Huang, M. Torres-Lugo, et al., Ann. Rev. Biomed. Eng., 2, 9 – 29 (2000).
M. Singh, R. Sharma, and U. C. Banerjee, Biotechnol Adv, 20, 341 – 59 (2002).
T. Nakajima, H. Furukawa, Y. Tanaka, et al., Macromolecules, 42, 2184 – 2189 (2009).
A. Beig, J. M. Miller, and A. Dahan, Eur. J. Pharm. Biopharm., 85, 1293 – 9 (2013).
N. M. Crini and G. Crini, Prog. Polym. Sci., 38, 344 – 368 (2013).
G. Mocanu, D. Vizitiu, and A. Carpov, J. Bioact. Compat. Polym., 16, 315 – 342 (2001).
P. A, L. R, and J. JLV, Intern. J. Pharm., 175, 75 – 84 (1998).
N. Z. Aleš, Intern. J. Pharm., 291, 51 – 58 (2005).
H. Itagaki, T. Kurokawa, H. Furukawa, et al., Macromolecules, 43, 9495 – 9500 (2010).
K. Uekama, F. Kihara, H. Arima, et al., Bioconjug. Chem., 14, 342 – 350 (2003).
K. Uekama, F. Kihara, H. Arima, et al., Bioconjug. Chem., 13, 1211 – 1219 (2002).
S. Hbaieb, R. Kalfat, Y. Chevalier, et al., Mater. Sci. Eng. C, 28, 697 – 704 (2008).
K. S. Novoselov, A. K. Geim, S. V. Morozov, et al., Science, 306, 666 – 9 (2004).
Y. Qin, C. Zou, X. Yan, et al., Chem. Eng. Res. Design, 94, 301 – 306 (2015).
H. Q. Song, R. Q. Li, S. Duan, et al., Nanoscale, 7, 5803 – 5814 (2015).
P. R. Ashton, R. Koniger, J. F. Stoddart, et al., J. Org. Chem., 61, 903 – 908 (1996).
T. Higuchi and K. A. Connors, Advances in Analytical Chemistry and Instrumentation,Willey-Interscience, New York (1965), Vol. 4.
L. Wang, S. Li, P. Tang, et al., Carbohydr. Polym., 129, 9 – 16 (2015).
K. N. Baglole, P. G. Boland, and B. D. Wagner, J. Photochem. Photobiol. A, 173, 230 – 237 (2005).
M. B. M. De Azevedo, J. B. Alderete, A. C. S. Lino, et al., J. Inclus. Phenom. Macrocycl. Chem., 37, 67 – 74 (2000).
X. Ge, J. He, Y. Yang, et al., J. Mol. Struct., 994, 163 – 169(2011).
M. I. Sancho, E. Gasull, S. E. Blanco, et al., Carbohydr. Res., 346, 1978 – 84 (2011).
X. Shen, M. Belletete, and G. Durocher, J. Phys. Chem. B, 102, 1877 – 1883 (1998).
W. Misiuk and M. Zalewska, Carbohydr. Polym., 77, 482 – 488(2009).
S. C. Jagdale, V. N. Jadhav, A. R. Chabukswar, et al., Braz. J.Pharm. Sci., 48, 131 – 145 (2012).
T. Loftsson, D. Hreinsdottir, and M. Masson, Intern. J. Pharm., 302, 18 – 28 (2005).
P. Job, Ann. Chim., 9, 113 – 203 (1928).
E. Fenyvesi, K. Gruiz, S. Verstichel, et al., Chemosphere, 60, 1001 – 1008 (2005).
M. J. Jozwiakowski and K. A. Connors, Carbohydr. Res., 143, 51 – 59 (1985).
Acknowledgments
The authors express appreciation to the Shahid Bahonar University of Kerman Faculty Research Committee for its support of this investigation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Heydari, A., Iranmanesh, M., Doostan, F. et al. Preparation of Inclusion Complex Between Nifedipine and Ethylenediamine-β-Cyclodextrin as Nanocarrier Agent. Pharm Chem J 49, 605–612 (2015). https://doi.org/10.1007/s11094-015-1338-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-015-1338-1