Abstract
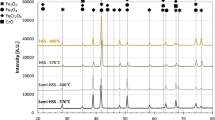
Non-isothermal oxidation kinetics of the high-speed steel (HSS) were studied by thermal gravimetric analysis. The surface and cross-sectional morphology of the HSS oxide film formed at different temperatures and durations were observed by scanning electron microscopy, and the corresponding chemical composition was analyzed by using energy dispersive spectrometer. The composition and structure of the oxide film were also investigated by X-ray diffraction. The results showed that the oxide scale of the HSS is mainly composed of Fe2O3, Fe3O4 and FeCr2O4. Temperature is the main factor on the quality of the oxide film. Below 600 °C, the oxidation rate of the steel is slow and the thickness of the oxide film is below 5 µm. However, the oxidation rate sharply increased as the temperature reaches 600 °C. Cr, Mo and V tend to concentrate at the scale/steel interface and form FeCr2O4 and other oxides.













Similar content being viewed by others
References
C. Vergne, C. Boher, C. Levaillant and R. Gras, Wear 250, 322 (2001).
M. Schütze, Oxidation of Metals 44, 29 (1995).
A. Molinari, G. Straffelini, A. Tomasi, A. Biggi and G. Corbo, Materials Science and Technology 17, 425 (2001).
H. Zhu, Q. Zhu, A. K. Tieu, B. Kosasih and C. Kong, Wear 302, 1310 (2013).
N. F. Garza-Montes-de-Oca, J. H. Ramírez-Ramírez, I. Alvarez-Elcoro, W. M. Rainforth and R. Colás, Oxidation of Metals 80, 191 (2013).
N. F. Garza-Montes-de-Oca, R. Colás and W. M. Rainforth, Oxidation of Metals 76, 451 (2011).
M. J. Monteiro, S. R. J. Saunders and F. C. Rizzo, Oxidation of Metals 75, 57 (2011).
K. Goto, Y. Matsuda, K. Sakamoto and Y. Sugimoto, ISIJ International 32, 1184 (1992).
F. J. Pérez, L. Martínez, M. P. Hierro, C. Gómez, A. L. Portela, G. N. Pucci, D. Duday, J. Lecomte-Beckers and Y. Greday, Corrosion Science 48, 472 (2006).
P. A. Munther and J. G. Lenard, Journal of Materials Processing Technology 88, 105 (1999).
O. Kato, H. Yamamoto, M. Ataka and K. Nakajima, ISIJ International 32, 1216 (1992).
H. H. Kim, J. W. Lim and J. J. Lee, ISIJ International 43, 1983 (2003).
R. Colas, J. Ramırez, I. Sandoval, J. C. Morales and L. A. Leduc, Wear 230, 56 (1999).
R. D. Mercado-Solis, J. Talamantes-Silva, J. H. Beynon and M. A. L. Hemandez-Rodriguez, Wear 263, 1560 (2007).
C. Vergne, C. Boher, R. Gras and C. Levaillant, Wear 260, 957 (2006).
J. H. Lee, J. C. Oh, J. W. Park, H. C. Lee and S. Lee, ISIJ International 41, 859 (2001).
L. Zhou, F. Liu, D. Sun and L. Yao, Journal of Iron and Steel Research 17, 60 (2005).
L. Zhou, F. Liu and D. Sun, Steel Rolling 22, 10 (2005).
Q. Zhu, H. T. Zhu, A. K. Tieu, M. Reid and L. C. Zhang, Corrosion Science 52, 2707 (2010).
Q. Zhu, H. T. Zhu, A. K. Tieu and C. Kong, Corrosion Science 53, 3603 (2011).
L. Zhou, F. Liu, C. S. Liu and D. L. Sun, Transfer Material Heat Treatment 25, 134 (2004).
A. Molinari, M. Pellizzari, A. Biggi, G. Gorbo, A. Tremea, in Proceedings 6th International Tooling Conference (Karlstadt University, Sweden, 2002), p. 365.
A. Molinari, G. Straffelini, A. Tomasi, A. Biggi and G. Corbo, Materials Science and Engineering A 280, 255 (2000).
M. J. Monteiro and F. C. Rizzo, Materials Science Forum 522, 171 (2006).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yin, Y., Sun, J., Teng, S. et al. Oxidation Behavior of High-Speed Steel Used for Hot Rolls. Oxid Met 86, 45–57 (2016). https://doi.org/10.1007/s11085-016-9618-z
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11085-016-9618-z