Abstract
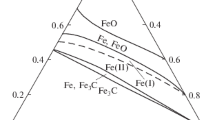
Kinetics of the oxidation of iron in fluxes of atomic and molecular oxygen have been studied over a wide temperature range from 573 to 1273 K. The influence of oxygen pre-dissociation on the oxidation rate was found negligible at 573–1073 K. At temperatures above 1073 K, when only FeO is formed, the rate of iron oxidation in atomic oxygen is substantially higher than that in molecular oxygen. Decarburization occurs during the first stage of oxidation of iron containing carbon. The rate of carbon elimination in atomic oxygen exceeds that in molecular oxygen due to the higher chemical potential of atomic gas.
Similar content being viewed by others
REFERENCES
A. De Roij, European Space Agency Journal 13, 363 (1989).
M. L. Zheludkevich, A. G. Gusakov, A. G. Voropaev, A. A. Vecher, E. N. Kozyrskiy, and S. A. Raspopov, Oxidation of Metals 61, 39, (2004).
A. G. Gusakov, A. G. Voropaev, M. L. Zheludkevich, A. A. Vecher, and S. A. Raspopov, Physical Chemistry Chemical Physics 1, 5311 (1999).
P. G. Dickens, R. Heckingbottom, and J. W. Linett, Transactions of the Faraday Society 65, 2235 (1969).
V. I. Bolobov and K. M. Makarov, Zashchita Metallov 28, 1007 (1992).
A. Vesel, M. Mozetic, and A. Zalar, Applied Surface Science 200, 94 (2002).
S. A. Raspopov, A. G. Gusakov, A. G. Voropaev, A. A. Vecher, M. L. Zheludkevich, O. A. Lukyanchenko, A. G. Gritsovets, and V. K Grishin, Journal of Chemical Society, Faraday Transactions 93, 2113 (1997).
R. Y. Chen and W. Y. D. Yuen, Oxidation of Metals 59, 433 (2003).
E. Fromm and E. Gebhardt, Gases and Carbon in Metals (Springer-Verlag, Berlin, 1976).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zheludkevich, M.L., Gusakov, A.G., Voropaev, A.G. et al. Influence of Oxygen Dissociation on the Oxidation of Iron. Oxidation of Metals 62, 223–235 (2004). https://doi.org/10.1007/s11085-004-7809-5
Issue Date:
DOI: https://doi.org/10.1007/s11085-004-7809-5