Abstract
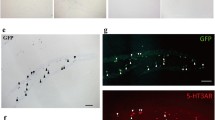
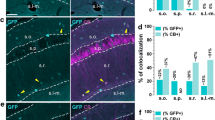
G protein-coupled receptor 50 (GPR50) belongs to the G protein-coupled receptor which is highly homologous with the sequence of melatonin receptor MT1 and MT2. GPR50 expression has previously been reported in many brain regions, like cortex, midbrain, pons, amygdala. But, the distribution of GPR50 in the hippocampus and cortex and the cell types expressing GPR50 is not yet clear. In this study, we examined the distribution of GPR50 in adult male mice by immunofluorescence. Our results showed that GPR50 was localized in the CA1-3 pyramidal cells and the granule cells of the dentate gyrus. GPR50 was also expressed in excitatory and inhibitory neurons. As inhibitory neurons also contain many types, we found that GPR50 was localized in some interneurons in which it was co-expressed with the calcium-binding proteins calbindin, calretinin, and parvalbumin. Besides, similar results were seen in the cortex. The widespread expression of GPR50 in the hippocampus and cortex suggests that GPR50 may be associated with synaptic plasticity and cognitive function.









Similar content being viewed by others
References
Ryan J, Carrière I, Ritchie K, Ancelin ML (2015) Involvement of GPR50 polymorphisms in depression: independent replication in a prospective elderly cohort. Brain Behav 5(3):e00313
Khan MZ, He L (2017) Neuro-psychopharmacological perspective of Orphan receptors of Rhodopsin (class A) family of G protein-coupled receptors. Psychopharmacology 234(8):1181–1207
Grünewald E, Kinnell HL, Porteous DJ, Thomson PA (2009) GPR50 interacts with neuronal NOGO-A and affects neurite outgrowth. Mol Cell Neurosci 42(4):363–371
Khan MZ, He L, Zhuang X (2016) The emerging role of GPR50 receptor in brain. Biomed Pharmacother 78:121–128
Ma YX, Wu ZQ, Feng YJ, Xiao ZC, Qin XL, Ma QH (2015) G protein coupled receptor 50 promotes self-renewal and neuronal differentiation of embryonic neural progenitor cells through regulation of notch and wnt/β-catenin signalings. Biochem Biophys Res Commun 458(4):836–842
Levoye A, Dam J, Ayoub MA, Guillaume JL, Couturier C, Delagrange P et al (2006) The orphan GPR50 receptor specifically inhibits MT1 melatonin receptor function through heterodimerization. EMBO J 25(13):3012–3023
Bhattacharyya S, Luan J, Challis B, Keogh J, Montague C, Brennand J et al (2006) Sequence variants in the melatonin-related receptor gene (GPR50) associate with circulating triglyceride and HDL levels. J Lipid Res 47(4):761–766
Barrett P, Ivanova E, Graham ES, Ross AW, Wilson D, Plé H et al (2006) Photoperiodic regulation of cellular retinol binding protein, CRBP1 [corrected] and nestin in tanycytes of the third ventricle ependymal layer of the Siberian hamster. J Endocrinol 191(3):687–698
Ivanova EA, Bechtold DA, Dupré SM, Brennand J, Barrett P, Luckman SM et al (2008) Altered metabolism in the melatonin-related receptor (GPR50) knockout mouse. Am J Physiol Endocrinol Metab 294(1):E176–182
Li J, Hand LE, Meng QJ, Loudon AS, Bechtold DA (2011) GPR50 interacts with TIP60 to modulate glucocorticoid receptor signalling. PLoS ONE 6(8):e23725
Wojciech S, Ahmad R, Belaid-Choucair Z, Journé AS, Gallet S, Dam J et al (2018) The orphan GPR50 receptor promotes constitutive TGFβ receptor signaling and protectsagainst cancer development. Nat Commun 9(1):1216
Dufourny L, Levasseur A, Migaud M, Callebaut I, Pontarotti P, Malpaux B et al (2008) GPR50 is the mammalian ortholog of Mel1c: evidence of rapid evolution in mammals. BMC Evol Biol 8:105
Wu Q, Zhao CW, Long Z, Xiao B, Feng L (2018) anatomy based networks and topology alteration in seizure-related cognitiveoutcomes. Front Neuroanat 12:25
Scoville WB, Milner B (2000) Loss of recent memory after bilateral hippocampal lesions 1957. J Neuropsychiatr Clin Neurosci 12(1):103–113
Grünewald E, Tew KD, Porteous DJ, Thomson PA (2012) Developmental expression of orphan G protein-coupled receptor 50 in the mouse brain. ACS Chem Neurosci 3(6):459–472
Rocco BR, Sweet RA, Lewis DA, Fish KN (2016) GABA-synthesizing enzymes in calbindin and calretinin neurons in monkey prefrontalcortex. Cereb Cortex 26(5):2191–2204
Denley MCS, Gatford NJF, Sellers KJ, Srivastava DP (2018) Estradiol and the development of the cerebral cortex: an unexpected role? Front Neurosci 12:245
Liu CY, Yang Y, Ju WN, Wang X, Zhang HL (2018) Emerging roles of astrocytes in neuro-vascular unit and the tripartite synapse with emphasis on reactive gliosis in the context of alzheimer's disease. Front Cell Neurosci 12:193
Scofield MD (2017) Exploring the role of astroglial glutamate release and association with synapses in neuronal function and behavior. Biol Psychiatry S0006–3223(17):32159–32165
Devaraju P, Sun MY, Myers TL, Lauderdale K, Fiacco TA (2013) Astrocytic group I mGluR-dependent potentiation of astrocytic glutamate and potassium uptake. J Neurophysiol 109(9):2404–2414
Meyer RC, Giddens MM, Schaefer SA, Hall RA (2013) GPR37 and GPR37L1 are receptors for the neuroprotective and glioprotective factors prosaptide and prosaposin. Proc Natl Acad Sci U S A 110(23):9529–9534
Cartier N, Lewis CA, Zhang R, Rossi FM (2014) The role of microglia in human disease: therapeutic tool or target? Acta Neuropathol 128(3):363–380
Morris GP, Clark IA, Zinn R, Vissel B (2013) Microglia: a new frontier for synaptic plasticity, learning and memory, and neurodegenerative disease research. Neurobiol Learn Mem 105:40–53
Preissler J, Grosche A, Lede V, Le Duc D, Krügel K, Matyash V et al (2015) Altered microglial phagocytosis in GPR34-deficient mice. Glia 63(2):206–215
Reyes-Resina I, Navarro G, Aguinaga D, Canela EI, Schoeder CT, Załuski M et al (2018) Molecular and functional interaction between GPR18 and cannabinoid CB2 G-protein-coupled receptors relevance in neurodegenerative diseases. Biochem Pharmacol S0006–2952(18):30209–30210
Song FE, Huang JL, Lin SH, Wang S, Ma GF, Tong XP (2017) Roles of NG2-glia in ischemic stroke. CNS Neurosci Ther 23(7):547–553
Lieberman OJ, McGuirt AF, Tang G, Sulzer D (2018) Roles for neuronal and glial autophagy in synaptic pruning during development. Neurobiol Dis S0969–9961(18):30134–30137
Selten M, van Bokhoven H, Nadif Kasri N. (2018) Inhibitory control of the excitatory/inhibitory balance in psychiatric disorders. F1000Res, 7:23
Ferraguti F, Cobden P, Pollard M, Cope D, Shigemoto R, Watanabe M et al (2004) Immunolocalization of metabotropic glutamate receptor 1alpha (mGluR1alpha) in distinct classes of interneuron in the CA1 region of the rat hippocampus. Hippocampus 14(2):193–215
Le Duigou C, Kullmann DM (2011) Group I mGluR agonist-evoked long-term potentiation in hippocampal oriens interneurons. J Neurosci 31(15):5777–5781
Benes FM, Berretta S (2001) GABAergic interneurons: implications for understanding schizophrenia and bipolar disorder. Neuropsychopharmacology 25(1):1–27
Kelsom C, Lu W (2013) Development and specification of GABAergic cortical interneurons. Cell Biosci 3(1):19
Moran LV, Hong LE (2011) High vs low frequency neural oscillations in schizophrenia. Schizophr Bull 37(4):659–663
Kann O, Papageorgiou IE, Draguhn A (2014) Highly energized inhibitory interneurons are a central element for information processing in cortical networks. J Cereb Blood Flow Metab 34(8):1270–1282
Schmidt MJ, Mirnics K (2015) Neurodevelopment, GABA system dysfunction, and schizophrenia. Neuropsychopharmacology 40(1):190–206
Maglóczky Z, Freund TF (2005) Impaired and repaired inhibitory circuits in the epileptic human hippocampus. Trends Neurosci 28(6):334–340
Petryszyn S, Parent A, Parent M (2018) The calretinin interneurons of the striatum: comparisons between rodents and primates under normal and pathological conditions. J Neural Transm (Vienna) 125(3):279–290
Hack NJ, Wride MC, Charters KM, Kater SB, Parks TN (2000) Developmental changes in the subcellular localization of calretinin. J Neurosci 20(7):RC67
Christel CJ, Schaer R, Wang S, Henzi T, Kreiner L, Grabs D et al (2012) Calretinin regulates Ca2+-dependent inactivation and facilitation of Ca(v)2.1 Ca2+ channels through a direct interaction with the α12.1 subunit. J Biol Chem 287(47):39766–39775
Pangršič T, Gabrielaitis M, Michanski S, Schwaller B, Wolf F, Strenzke N et al (2015) EF-hand protein Ca2+ buffers regulate Ca2+ influx and exocytosis in sensory hair cells. Proc Natl Acad Sci USA 112(9):E1028–1037
Cauli B, Zhou X, Tricoire L, Toussay X, Staiger JF (2014) Revisiting enigmatic cortical calretinin-expressing interneurons. Front Neuroanat 8:52
Gulyás AI, Hájos N, Freund TF (1996) Interneurons containing calretinin are specialized to control other interneurons in the rat hippocampus. J Neurosci 16(10):3397–3411
Xu L, Zhang XH (2015) Distribution of D1 and D2-dopamine receptors in calcium-binding-protein expressing interneurons in rat anterior cingulate cortex. Sheng Li Xue Bao 67(2):163–172
Caballero A, Tseng KY (2016) GABAergic function as a limiting factor for prefrontal maturation during adolescence. Trends Neurosci 39(7):441–448
Ping Ascoli GA, Alonso-Nanclares L, Anderson SA, Barrionuevo G, Benavides-Piccione R et al (2008) Petilla terminology: nomenclature of features of GABAergic interneurons of the cerebral cortex. Nat Rev Neurosci. 9(7):557–568
Packer AM, Yuste R (2011) Dense, unspecific connectivity of neocortical parvalbumin-positive interneurons: a canonical microcircuit for inhibition? J Neurosci 31(37):13260–13271
Acknowledgements
This work was supported by grants from Scientific Study Project for Institutes of Higher Learning, Ministry of Education, Liaoning Province (LZ2019072, LZ2017001, L2016002) and Natural science foundation of Liaoning province of China (2015020672).
Author information
Authors and Affiliations
Contributions
SL, JY and XW contributed to the conception and design of the project. QL, BG, HS, NL and YZ contributed to the experiments. QL and YS analyzed and interpreted the data. JY, MN, and SL wrote the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing financial interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, Q., Zhang, Y., Ge, BY. et al. GPR50 Distribution in the Mouse Cortex and Hippocampus. Neurochem Res 45, 2312–2323 (2020). https://doi.org/10.1007/s11064-020-03089-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-020-03089-w