Abstract
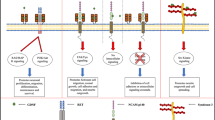
Our understanding of the physiological relevance of unique Damage-induced neuronal endopeptidase (DINE) [also termed Endothelin-converting enzyme-like 1 (ECEL1)] has recently expanded. DINE/ECEL1 is a type II membrane-bound metalloprotease, belonging to a family including the neprilysin (NEP) and endothelin-converting enzyme (ECE). The family members degrade and/or process peptides such as amyloid β and big-endothelins, which are closely associated with pathological conditions. Similar to NEP and ECE, DINE has been expected to play an important role in injured neurons as well as in developing neurons, because of its remarkable transcriptional response to neuronal insults and predominant neuronal expression from the embryonic stage. However, the physiological significance of DINE has long remained elusive. In the last decade, a series of genetically manipulated mice have driven research progress to elucidate the physiological aspects of DINE. The mice ablating Dine fail to arborize the embryonic motor axons in some subsets of muscles, including the respiratory muscles, and die immediately after birth. The abnormal phenotype of motor axons is also caused by one amino acid exchanges of DINE/ECEL1, which are responsible for distal arthrogryposis type 5 in a group of human congenital movement disorders. Furthermore, the mature Dine-deficient mice in which the lethality is rescued by genetic manipulation have shown the involvement of DINE in central nervous system regeneration. Here we describe recent research advances that DINE-mediated proteolytic processes are critical for nerve development, regeneration and pathogenesis, and discuss the future potential for DINE as a therapeutic target for axonal degeneration/disorder.





Similar content being viewed by others
References
Kiryu-Seo S, Sasaki M, Yokohama H, Nakagomi S, Hirayama T, Aoki S, Wada K, Kiyama H (2000) Damage-induced neuronal endopeptidase (DINE) is a unique metallopeptidase expressed in response to neuronal damage and activates superoxide scavengers. Proc Natl Acad Sci USA 97:4345–4350. https://doi.org/10.1073/pnas.070509897
Turner AJ, Fisk L, Nalivaeva NN (2004) Targeting amyloid-degrading enzymes as therapeutic strategies in neurodegeneration. Ann N Y Acad Sci 1035:1–20. https://doi.org/10.1196/annals.1332.001
Turner AJ, Isaac RE, Coates D (2001) The neprilysin (NEP) family of zinc metalloendopeptidases:genomics and function. Bioessays 23: 261–269
Iwata N, Tsubuki S, Takaki Y, Watanabe K, Sekiguchi M, Hosoki E, Kawashima-Morishima M, Lee HJ, Hama E, Sekine-Aizawa Y, Saido TC (2000) Identification of the major Abeta1-42-degrading catabolic pathway in brain parenchyma: suppression leads to biochemical and pathological deposition. Nat Med 6:143–150. https://doi.org/10.1038/72237
Yanagisawa H, Hammer RE, Richardson JA, Emoto N, Williams SC, Takeda S, Clouthier DE, Yanagisawa M (2000) Disruption of ECE-1 and ECE-2 reveals a role for endothelin-converting enzyme-2 in murine cardiac development. J Clin Investig 105:1373–1382. https://doi.org/10.1172/jci7447
Yanagisawa H, Yanagisawa M, Kapur RP, Richardson JA, Williams SC, Clouthier DE, de Wit D, Emoto N, Hammer RE (1998) Dual genetic pathways of endothelin-mediated intercellular signaling revealed by targeted disruption of endothelin converting enzyme-1 gene. Development 125:825–836
Valdenaire O, Richards JG, Faull RL, Schweizer A (1999) XCE, a new member of the endothelin-converting enzyme and neutral endopeptidase family, is preferentially expressed in the CNS. Brain Res Mol Brain Res 64:211–221
Matsumoto S, Kiryu-Seo S, Kiyama H (2016) Motor nerve arborization requires proteolytic domain of damage-induced neuronal endopeptidase (DINE) during development. J Neurosci 36:4744–4757. https://doi.org/10.1523/jneurosci.3811-15.2016
Nagata K, Kiryu-Seo S, Maeda M, Yoshida K, Morita T, Kiyama H (2010) Damage-induced neuronal endopeptidase is critical for presynaptic formation of neuromuscular junctions. J Neurosci 30:6954–6962. https://doi.org/10.1523/jneurosci.4521-09.2010
Dieterich K, Quijano-Roy S, Monnier N, Zhou J, Faure J, Smirnow DA, Carlier R, Laroche C, Marcorelles P, Mercier S, Megarbane A, Odent S, Romero N, Sternberg D, Marty I, Estournet B, Jouk PS, Melki J, Lunardi J (2013) The neuronal endopeptidase ECEL1 is associated with a distinct form of recessive distal arthrogryposis. Hum Mol Genet 22:1483–1492. https://doi.org/10.1093/hmg/dds514
McMillin MJ et al (2013) Mutations in ECEL1 cause distal arthrogryposis type 5D. Am J Hum Genet 92:150–156. https://doi.org/10.1016/j.ajhg.2012.11.014
Nagata K, Kiryu-Seo S, Tamada H, Okuyama-Uchimura F, Kiyama H, Saido TC (2016) ECEL1 mutation implicates impaired axonal arborization of motor nerves in the pathogenesis of distal arthrogryposis. Acta Neuropathol 132:111–126. https://doi.org/10.1007/s00401-016-1554-0
Nagata K, Takahashi M, Kiryu-Seo S, Kiyama H, Saido TC (2017) Distinct functional consequences of ECEL1/DINE missense mutations in the pathogenesis of congenital contracture disorders. Acta Neuropathol Commun 5:83. https://doi.org/10.1186/s40478-017-0486-9
Kaneko A, Kiryu-Seo S, Matsumoto S, Kiyama H (2017) Damage-induced neuronal endopeptidase (DINE) enhances axonal regeneration potential of retinal ganglion cells after optic nerve injury. Cell Death Dis 8:e2847. https://doi.org/10.1038/cddis.2017.212
Kiryu S, Yao GL, Morita N, Kato H, Kiyama H (1995) Nerve injury enhances rat neuronal glutamate transporter expression: identification by differential display PCR. J Neurosci 15:7872–7878
Morita N, Kiryu S, Kiyama H (1996) p53-independent cyclin G expression in a group of mature neurons and its enhanced expression during nerve regeneration. J Neurosci 16:5961–5966
Valdenaire O, Rohrbacher E, Langeveld A, Schweizer A, Meijers C (2000) Organization and chromosomal localization of the human ECEL1 (XCE) gene encoding a zinc metallopeptidase involved in the nervous control of respiration. Biochem J 346 Pt 3:611–616
Shirotani K, Tsubuki S, Iwata N, Takaki Y, Harigaya W, Maruyama K, Kiryu-Seo S, Kiyama H, Iwata H, Tomita T, Iwatsubo T, Saido TC (2001) Neprilysin degrades both amyloid beta peptides 1–40 and 1–42 most rapidly and efficiently among thiorphan- and phosphoramidon-sensitive endopeptidases. J Biol Chem 276:21895–21901. https://doi.org/10.1074/jbc.M008511200
Nagata K, Kiryu-Seo S, Kiyama H (2006) Localization and ontogeny of damage-induced neuronal endopeptidase mRNA-expressing neurons in the rat nervous system. Neuroscience 141:299–310. https://doi.org/10.1016/j.neuroscience.2006.03.032
Schweizer A, Valdenaire O, Koster A, Lang Y, Schmitt G, Lenz B, Bluethmann H, Rohrer J (1999) Neonatal lethality in mice deficient in XCE, a novel member of the endothelin-converting enzyme and neutral endopeptidase family. J Biol Chem 274:20450–20456
Darabid H, Perez-Gonzalez AP, Robitaille R (2014) Neuromuscular synaptogenesis: coordinating partners with multiple functions. Nat Rev Neurosci 15:703–718
Jessen KR, Mirsky R (2005) The origin and development of glial cells in peripheral nerves. Nat Rev Neurosci 6:671–682. https://doi.org/10.1038/nrn1746
Jessen KR, Mirsky R, Lloyd AC (2015) Schwann cells: development and role in nerve repair. Cold Spring Harb Perspect Biol 7:a020487. https://doi.org/10.1101/cshperspect.a020487
Raphael AR, Perlin JR, Talbot WS (2010) Schwann cells reposition a peripheral nerve to isolate it from postembryonic remodeling of its targets. Development 137:3643–3649. https://doi.org/10.1242/dev.057521
Wanner IB, Mahoney J, Jessen KR, Wood PM, Bates M, Bunge MB (2006) Invariant mantling of growth cones by Schwann cell precursors characterize growing peripheral nerve fronts. Glia 54:424–438. https://doi.org/10.1002/glia.20389
Britsch S, Goerich DE, Riethmacher D, Peirano RI, Rossner M, Nave KA, Birchmeier C, Wegner M (2001) The transcription factor Sox10 is a key regulator of peripheral glial development. Genes Dev 15:66–78
Lin W, Sanchez HB, Deerinck T, Morris JK, Ellisman M, Lee KF (2000) Aberrant development of motor axons and neuromuscular synapses in erbB2-deficient mice. Proc Natl Acad Sci USA 97:1299–1304
Riethmacher D, Sonnenberg-Riethmacher E, Brinkmann V, Yamaai T, Lewin GR, Birchmeier C (1997) Severe neuropathies in mice with targeted mutations in the ErbB3 receptor. Nature 389:725–730. https://doi.org/10.1038/39593
Guan Z, Kuhn JA, Wang X, Colquitt B, Solorzano C, Vaman S, Guan AK, Evans-Reinsch Z, Braz J, Devor M, Abboud-Werner SL, Lanier LL, Lomvardas S, Basbaum AI (2016) Injured sensory neuron-derived CSF1 induces microglial proliferation and DAP12-dependent pain. Nat Neurosci 19:94–101. https://doi.org/10.1038/nn.4189
Kanaan NM, Collier TJ, Cole-Strauss A, Grabinski T, Mattingly ZR, Winn ME, Steece-Collier K, Sortwell CE, Manfredsson FP, Lipton JW (2015) The longitudinal transcriptomic response of the substantia nigra to intrastriatal 6-hydroxydopamine reveals significant upregulation of regeneration-associated genes. PLoS ONE 10:e0127768. https://doi.org/10.1371/journal.pone.0127768
Ortuno-Sahagun D, Rivera-Cervantes MC, Gudino-Cabrera G, Junyent F, Verdaguer E, Auladell C, Pallas M, Camins A, Beas-Zarate C (2012) Microarray analysis of rat hippocampus exposed to excitotoxicity: reversal Na(+)/Ca(2+) exchanger NCX3 is overexpressed in glial cells. Hippocampus 22:128–140. https://doi.org/10.1002/hipo.20869
Yasuda M, Tanaka Y, Omodaka K, Nishiguchi KM, Nakamura O, Tsuda S, Nakazawa T (2016) Transcriptome profiling of the rat retina after optic nerve transection. Sci Rep 6:28736. https://doi.org/10.1038/srep28736
Yasuda M, Tanaka Y, Ryu M, Tsuda S, Nakazawa T (2014) RNA sequence reveals mouse retinal transcriptome changes early after axonal injury. PLoS ONE 9:e93258. https://doi.org/10.1371/journal.pone.0093258
Ohba N, Kiryu-Seo S, Maeda M, Muraoka M, Ishii M, Kiyama H (2004) Expression of damage-induced neuronal endopeptidase (DINE) mRNA in peri-infarct cortical and thalamic neurons following middle cerebral artery occlusion. J Neurochem 91:956–964. https://doi.org/10.1111/j.1471-4159.2004.02784.x
Kato R, Kiryu-Seo S, Kiyama H (2002) Damage-induced neuronal endopeptidase (DINE/ECEL) expression is regulated by leukemia inhibitory factor and deprivation of nerve growth factor in rat sensory ganglia after nerve injury. J Neurosci 22:9410–9418
Han SM, Baig HS, Hammarlund M (2016) Mitochondria localize to injured axons to support regeneration. Neuron 92:1308–1323. https://doi.org/10.1016/j.neuron.2016.11.025
Shin JE, Cho Y, Beirowski B, Milbrandt J, Cavalli V, DiAntonio A (2012) Dual leucine zipper kinase is required for retrograde injury signaling and axonal regeneration. Neuron 74:1015–1022. https://doi.org/10.1016/j.neuron.2012.04.028
Tedeschi A, Bradke F (2013) The DLK signalling pathway–a double-edged sword in neural development and regeneration. EMBO Rep 14:605–614. https://doi.org/10.1038/embor.2013.64
Wlaschin JJ, Gluski JM, Nguyen E, Silberberg H, Thompson JH, Chesler AT, Le Pichon CE (2018) Dual leucine zipper kinase is required for mechanical allodynia and microgliosis after nerve injury. Elife 7. https://doi.org/10.7554/eLife.33910
Kummer KK, Kalpachidou T, Kress M, Langeslag M (2017) Signatures of altered gene expression in dorsal root ganglia of a fabry disease mouse model. Front Mol Neurosci 10:449. https://doi.org/10.3389/fnmol.2017.00449
Kiryu-Seo S, Kato R, Ogawa T, Nakagomi S, Nagata K, Kiyama H (2008) Neuronal injury-inducible gene is synergistically regulated by ATF3, c-Jun, and STAT3 through the interaction with Sp1 in damaged neurons. J Biol Chem 283:6988–6996. https://doi.org/10.1074/jbc.M707514200
Chandran V et al (2016) A systems-level analysis of the peripheral nerve intrinsic axonal growth program. Neuron 89:956–970. https://doi.org/10.1016/j.neuron.2016.01.034
Nakagomi S, Suzuki Y, Namikawa K, Kiryu-Seo S, Kiyama H (2003) Expression of the activating transcription factor 3 prevents c-Jun N-terminal kinase-induced neuronal death by promoting heat shock protein 27 expression and Akt activation. J Neurosci 23:5187–5196
Seijffers R, Mills CD, Woolf CJ (2007) ATF3 increases the intrinsic growth state of DRG neurons to enhance peripheral nerve regeneration. J Neurosci 27:7911–7920. https://doi.org/10.1523/jneurosci.5313-06.2007
Kiryu-Seo S, Kiyama H (2011) The nuclear events guiding successful nerve regeneration. Front Mol Neurosci 4:53. https://doi.org/10.3389/fnmol.2011.00053
Benowitz LI, He Z, Goldberg JL (2017) Reaching the brain: advances in optic nerve regeneration. Exp Neurol 287:365–373. https://doi.org/10.1016/j.expneurol.2015.12.015
He Z, Jin Y (2016) Intrinsic control of axon regeneration. Neuron 90:437–451. https://doi.org/10.1016/j.neuron.2016.04.022
Yin Y, Cui Q, Li Y, Irwin N, Fischer D, Harvey AR, Benowitz LI (2003) Macrophage-derived factors stimulate optic nerve regeneration. J Neurosci 23:2284–2293
Sato K, Shiga Y, Nakagawa Y, Fujita K, Nishiguchi KM, Tawarayama H, Murayama N, Maekawa S, Yabana T, Omodaka K, Katayama S, Feng Q, Tsuda S, Nakazawa T (2018) Ecel1 knockdown with an AAV2-mediated CRISPR/Cas9 system promotes optic nerve damage-induced RGC death in the mouse retina. Invest Ophthalmol Vis Sci 59:3943–3951. https://doi.org/10.1167/iovs.18-23784
Turner AJ, Tanzawa K (1997) Mammalian membrane metallopeptidases: NEP, ECE, KELL, and PEX. Faseb j 11:355–364
Hofstra RM, Valdenaire O, Arch E, Osinga J, Kroes H, Loffler BM, Hamosh A, Meijers C, Buys CH (1999) A loss-of-function mutation in the endothelin-converting enzyme 1 (ECE-1) associated with Hirschsprung disease, cardiac defects, and autonomic dysfunction. Am J Hum Genet 64:304–308. https://doi.org/10.1086/302184
Higuchi Y et al (2016) Mutations in MME cause an autosomal-recessive charcot-marie-tooth disease type 2. Ann Neurol 79:659–672. https://doi.org/10.1002/ana.24612
Auer-Grumbach M et al (2016) Rare variants in MME, encoding metalloprotease neprilysin, are linked to late-onset autosomal-dominant axonal polyneuropathies. Am J Hum Genet 99:607–623. https://doi.org/10.1016/j.ajhg.2016.07.008
Barnett CP, Todd EJ, Ong R, Davis MR, Atkinson V, Allcock R, Laing N, Ravenscroft G (2014) Distal arthrogryposis type 5D with novel clinical features and compound heterozygous mutations in ECEL1. Am J Med Genet A 164:1846–1849. https://doi.org/10.1002/ajmg.a.36342
Khan AO, Shaheen R, Alkuraya FS (2014) The ECEL1-related strabismus phenotype is consistent with congenital cranial dysinnervation disorder. J aapos 18:362–367. https://doi.org/10.1016/j.jaapos.2014.03.005
Patil SJ, Rai GK, Bhat V, Ramesh VA, Nagarajaram HA, Matalia J, Phadke SR (2014) Distal arthrogryposis type 5D with a novel ECEL1 gene mutation. Am J Med Genet A 164a:2857–2862. https://doi.org/10.1002/ajmg.a.36702
Shaaban S, Duzcan F, Yildirim C, Chan WM, Andrews C, Akarsu NA, Engle EC (2014) Expanding the phenotypic spectrum of ECEL1-related congenital contracture syndromes. Clin Genet 85:562–567. https://doi.org/10.1111/cge.12224
Shaheen R, Al-Owain M, Khan AO, Zaki MS, Hossni HA, Al-Tassan R, Eyaid W, Alkuraya FS (2014) Identification of three novel ECEL1 mutations in three families with distal arthrogryposis type 5D. Clin Genet 85:568–572. https://doi.org/10.1111/cge.12226
Dohrn N, Le VQ, Petersen A, Skovbo P, Pedersen IS, Ernst A, Krarup H, Petersen MB (2015) ECEL1 mutation causes fetal arthrogryposis multiplex congenita. Am J Med Genet A 167a:731–743. https://doi.org/10.1002/ajmg.a.37018
Todd EJ et al (2015) Next generation sequencing in a large cohort of patients presenting with neuromuscular disease before or at birth. Orphanet J Rare Dis 10:148. https://doi.org/10.1186/s13023-015-0364-0
Bayram Y et al (2016) Molecular etiology of arthrogryposis in multiple families of mostly Turkish origin. J Clin Investig 126:762–778. https://doi.org/10.1172/jci84457
Hamzeh AR, Nair P, Mohamed M, Saif F, Tawfiq N, Khalifa M, Al-Ali MT, Bastaki F (2017) A novel variant in the endothelin-converting enzyme-like 1 (ECEL1) gene in an Emirati child. Med Princ Pract 26:195–198. https://doi.org/10.1159/000456034
Stattin EL, Johansson J, Gudmundsson S, Ameur A, Lundberg S, Bondeson ML, Wilbe M (2018) A novel ECEL1 mutation expands the phenotype of distal arthrogryposis multiplex congenita type 5D to include pretibial vertical skin creases. Am J Med Genet A 176:1405–1410. https://doi.org/10.1002/ajmg.a.38691
Benoit A, Vargas MA, Desgroseillers L, Boileau G (2004) Endothelin-converting enzyme-like 1 (ECEL1) is present both in the plasma membrane and in the endoplasmic reticulum. Biochem J 380:881–888. https://doi.org/10.1042/bj20040215
Acknowledgements
We sincerely thank Professor Anthony J. Turner for his fruitful and warmest advice on this study. This work was financially supported by the Japan Society for the Promotion of Science KAKENHI Grant Number 18K15379 (K.N.), KAKENHI Grant Number 18H02524 (S.K.-S.), KAKENHI Grant Number 16H05117 and 17H05743 (H.K.), Japan Foundation for Applied Enzymology (H.K.), Takeda Science Foundation (S.K.-S) and the 24th General Assembly of the Japanese Association of Medical Sciences (S.K.-S.).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Special issue: In honor of Prof. Anthony J. Turner.
Rights and permissions
About this article
Cite this article
Kiryu-Seo, S., Nagata, K., Saido, T.C. et al. New Insights of a Neuronal Peptidase DINE/ECEL1: Nerve Development, Nerve Regeneration and Neurogenic Pathogenesis. Neurochem Res 44, 1279–1288 (2019). https://doi.org/10.1007/s11064-018-2665-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-018-2665-x