Abstract
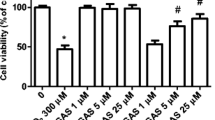
The contribution of oxidative stress to the pathophysiology of depression has been described in numerous studies. Particularly, an increased production of reactive oxygen species (ROS) caused by mitochondrial dysfunction can lead to neuronal cell death. Human neuroblastoma SH-SY5Y cells were used to investigate the neuroprotective effect of the antidepressant duloxetine against rotenone-induced oxidative stress. SH-SY5Y cells were pretreated with duloxetine (1–5 µM) for 24 h followed by a 24-h rotenone exposure (10 µM). The phosphatidylinositol 3-kinase/protein kinase B (PI3K/Akt) inhibitor LY294002 (10 µM) and the heme oxygenase 1 (HO-1) inhibitor zinc protoporphyrin IX-ZnPP (5 µM) were added to cultures 1 h prior duloxetine treatments. After treatments cell viability and ROS generation were assessed. NF-E2-related factor-2 (Nrf2) nuclear translocation was assessed by immunofluorescent staining after 4 and 8 h of duloxetine incubation. Furthermore, the Nrf2 and HO-1 mRNA expression was carried out after 4–48 h of duloxetine treatment by qRT-PCR. Duloxetine pretreatment antagonized rotenone-induced overproduction of ROS and cell death in SH-SY5Y cells. In addition, a 1-h pretreatment with LY294002 abolished duloxetine’s protective effect. Duloxetine also induced nuclear translocation of the Nrf2 and the expression of its target gene, HO-1. Finally, the HO-1 inhibitor, ZnPP, suppressed the duloxetine protective effect. Overall, these results indicate that the mechanism of duloxetine neuroprotective action against oxidative stress and cell death might rely on the Akt/Nrf2/HO-1 pathways.






Similar content being viewed by others
References
Campbell S, Marriott M, Nahmias C, MacQueen GM (2004) Lower hippocampal volume in patients suffering from depression: a meta-analysis. Am J Psychiatry 161:598–607
Nakamura T, Cho D-H, Lipton SA (2012) Redox regulation of protein misfolding, mitochondrial dysfunction, synaptic damage, and cell death in neurodegenerative diseases. Exp Neurol 238:12–21
Fukui H, Moraes CT (2008) The mitochondrial impairment, oxidative stress and neurodegeneration connection: reality or just an attractive hypothesis? Trends Neurosci 31:251–256
Ray PD, Huang B-W, Tsuji Y (2012) Reactive oxygen species (ROS) homeostasis and redox regulation in cellular signaling. Cell Signal 24:981–990
Valko M, Leibfritz D, Moncol J, Cronin MTD, Mazur M, Telser J (2007) Free radicals and antioxidants in normal physiological functions and human disease. Int J Biochem Cell Biol 39:44–84
Behr GA, Moreira JCF, Frey BN (2012) Preclinical and clinical evidence of antioxidant effects of antidepressant agents: implications for the pathophysiology of major depressive disorder. Oxid Med Cell Longev 2012:1–13
Maes M, Mihaylova I, Kubera M, Uytterhoeven M, Vrydags N, Bosmans E (2010) Increased plasma peroxides and serum oxidized low density lipoprotein antibodies in major depression: markers that further explain the higher incidence of neurodegeneration and coronary artery disease. J Affect Disord 125:287–294
Khanzode SD, Dakhale GN, Khanzode SS, Saoji A, Palasodkar R (2003) Oxidative damage and major depression: the potential antioxidant action of selective serotonin re-uptake inhibitors. Redox Rep 8:365–370
Lee CH, Park JH, Yoo KY, Choi JH, Hwang IK, Ryu PD et al (2011) Pre- and post-treatments with escitalopram protect against experimental ischemic neuronal damage via regulation of BDNF expression and oxidative stress. Exp Neurol 229:450–459
Moretti M, Colla A, Balen GO, dos Santos DB, Budni J, de Freitas AE et al (2012) Ascorbic acid treatment, similarly to fluoxetine, reverses depressive-like behavior and brain oxidative damage induced by chronic unpredictable stress. J Psychiatr Res 46:331–340
Bilici M, Efe H, Köroğlu MA, Uydu HA, Bekaroğlu M, Değer O (2001) Antioxidative enzyme activities and lipid peroxidation in major depression: alterations by antidepressant treatments. J Affect Disord 64:43–51
Michel TM, Frangou S, Thiemeyer D, Camara S, Jecel J, Nara K et al (2007) Evidence for oxidative stress in the frontal cortex in patients with recurrent depressive disorder—a postmortem study. Psychiatry Res 151:145–150
Grant MM, Barber VS, Griffiths HR (2005) The presence of ascorbate induces expression of brain derived neurotrophic factor in SH-SY5Y neuroblastoma cells after peroxide insult, which is associated with increased survival. Proteomics 5:534–540
Parcellier A, Tintignac LA, Zhuravleva E, Hemmings BA (2008) PKB and the mitochondria: AKTing on apoptosis. Cell Signal 20:21–30
Kitagishi Y, Kobayashi M, Kikuta K, Matsuda S (2012) Roles of pi3k/akt/gsk3/mtor pathway in cell signaling of mental illnesses. Depress Res Treat 2012:1–8
Hsiung S, Adlersberg M, Arango V, Mann JJ, Tamir H, Liu K (2003) Attenuated 5-HT1A receptor signaling in brains of suicide victims: involvement of adenylyl cyclase, phosphatidylinositol 3-kinase, Akt and mitogen-activated protein kinase. J Neurochem 87:182–194
Karege F, Perroud N, Burkhardt S, Schwald M, Ballmann E, La Harpe R et al (2007) Alteration in kinase activity but not in protein levels of protein kinase B and glycogen synthase kinase-3beta in ventral prefrontal cortex of depressed suicide victims. Biol Psychiatry 61:240–245
Hunziker ME, Suehs BT, Bettinger TL, Crismon ML (2005) Duloxetine hydrochloride: a new dual-acting medication for the treatment of major depressive disorder. Clin Ther 27:1126–1143
Calabrese F, Molteni R, Maj PF, Cattaneo A, Gennarelli M, Racagni G et al (2007) Chronic duloxetine treatment induces specific changes in the expression of BDNF transcripts and in the subcellular localization of the neurotrophin protein. Neuropsychopharmacology 32:2351–2359
Molteni R, Calabrese F, Cattaneo A, Mancini M, Gennarelli M, Racagni G et al (2009) Acute stress responsiveness of the neurotrophin BDNF in the rat hippocampus is modulated by chronic treatment with the antidepressant duloxetine. Neuropsychopharmacology 34:1523–1532
Prickaerts J, De Vry J, Boere J, Kenis G, Quinton MS, Engel S, Melnick L, Schreiber R (2012) Differential BDNF responses of triple versus dual reuptake inhibition in neuronal and astrocytoma cells as well as in rat hippocampus and prefrontal cortex. J Mol Neurosci 48:167–175
Engel D, Zomkowski ADE, Lieberknecht V, Rodrigues AL, Gabilan NH (2013) Chronic administration of duloxetine and mirtazapine downregulates proapoptotic proteins and upregulates neurotrophin gene expression in the hippocampus and cerebral cortex of mice. J Psychiatr Res 47:802–808
Isenberg JS, Klaunig JE (2000) Role of the mitochondrial membrane permeability transition (MPT) in rotenone-induced apoptosis in liver cells. Toxicol Sci Off J Soc Toxicol 53:340–351
Panov A, Dikalov S, Shalbuyeva N, Taylor G, Sherer T, Greenamyre JT (2005) Rotenone model of parkinson disease: multiple brain mitochondria dysfunctions after short term systemic rotenone intoxication. J Biol Chem 280:42026–42035
Assis LC, Straliotto MR, Engel D, Hort MA, Dutra RC, de Bem AF (2014) β-Caryophyllene protects the C6 glioma cells against glutamate-induced excitotoxicity through the Nrf2 pathway. Neuroscience 279:220–231
Watabe M, Nakaki T (2007) Mitochondrial complex I inhibitor rotenone-elicited dopamine redistribution from vesicles to cytosol in human dopaminergic SH-SY5Y cells. J Pharmacol Exp Ther 323:499–507
Bryan HK, Olayanju A, Goldring CE, Park BK (2013) The Nrf2 cell defence pathway: Keap1-dependent and -independent mechanisms of regulation. Biochem Pharmacol 85:705–717
Scapagnini G, Sonya V, Nader AG, Calogero C, Zella D, Fabio G (2011) Modulation of Nrf2/ARE pathway by food polyphenols: a nutritional neuroprotective strategy for cognitive and neurodegenerative disorders. Mol Neurobiol 44:192–201
Banasr M, Dwyer JM, Duman RS (2011) Cell atrophy and loss in depression: reversal by antidepressant treatment. Curr Opin Cell Biol 23:730–737
Filomeni G, Graziani I, De Zio D, Dini L, Centonze D, Rotilio G et al (2012) Neuroprotection of kaempferol by autophagy in models of rotenone-mediated acute toxicity: possible implications for Parkinson’s disease. Neurobiol Aging 33:767–785
Xiong Y, Ding H, Xu M, Gao J (2009) Protective effects of asiatic acid on rotenone- or H2O2-induced injury in SH-SY5Y Cells. Neurochem Res 34:746–754
Wu Y, Li X, Zhu JX, Xie W, Le W, Fan Z et al (2011) Resveratrol-activated AMPK/SIRT1/Autophagy in cellular models of Parkinson’s Disease. Neurosignals 19:163–174
Park HJ, Kim HJ (2013) Inhibitory effect of nicardipine on rotenone-induced apoptosis in SH-SY5Y human neuroblastoma cells. Mol Med Rep 7:941–946
Kolla N, Wei Z, Richardson JS, Li X-M (2005) Amitriptyline and fluoxetine protect PC12 cells from cell death induced by hydrogen peroxide. J Psychiatry Neurosci 30:196–201
Zhang F, Zhou H, Wilson BC, Shi J-S, Hong J-S, Gao H-M (2012) Fluoxetine protects neurons against microglial activation-mediated neurotoxicity. Parkinsonism Relat Disord 18:S213–S217
Schmidt AJ, Heiser P, Hemmeter UM, Krieg J-C, Vedder H (2008) Effects of antidepressants on mRNA levels of antioxidant enzymes in human monocytic U-937 cells. Prog Neuropsychopharmacol Biol Psychiatry 32:1567–1573
Khanam R, Najfi H, Akhtar M, Vohora D (2012) Evaluation of venlafaxine on glucose homeostasis and oxidative stress in diabetic mice. Hum Exp Toxicol 31:1244–1250
Kumar P, Kalonia H, Kumar A (2010) Nitric oxide mechanism in the protective effect of antidepressants against 3-nitropropionic acid-induced cognitive deficit, glutathione and mitochondrial alterations in animal model of Huntington’s disease. Behav Pharmacol 21:217–230
Leibrock C, Ackermann TF, Hierlmeier M, Lang F, Borgwardt S, Lang UE (2013) Akt2 deficiency is associated with anxiety and depressive behavior in mice. Cell Physiol Biochem 32:766–777
Park J-A, Lee C-H (2017) Neuroprotective effect of duloxetine on chronic cerebral hypoperfusion-induced hippocampal neuronal damage. Biomol Ther. https://doi.org/10.4062/biomolther.2016.248
Lv Q, Gu C, Chen C (2014) Venlafaxine protects methylglyoxal-induced apoptosis in the cultured human brain microvascular endothelial cells. Neurosci Lett 569:99–103
Chopra VS, Chalifour LE, Schipper HM (1995) Differential effects of cysteamine on heat shock protein induction and cytoplasmic granulation in astrocytes and glioma cells. Brain Res Mol Brain Res 31:173–184
Guzmán-Beltrán S, Espada S, Orozco-Ibarra M, Pedraza-Chaverri J, Cuadrado A (2008) Nordihydroguaiaretic acid activates the antioxidant pathway Nrf2/HO-1 and protects cerebellar granule neurons against oxidative stress. Neurosci Lett 447:167–171
Hamdulay SS, Wang B, Birdsey GM, Ali F, Dumont O, Evans PC et al (2010) Celecoxib activates PI-3K/Akt and mitochondrial redox signaling to enhance heme oxygenase-1-mediated anti-inflammatory activity in vascular endothelium. Free Radic Biol Med 48:1013–1023
Martin D, Rojo AI, Salinas M, Diaz R, Gallardo G, Alam J et al (2004) Regulation of heme oxygenase-1 expression through the phosphatidylinositol 3-kinase/Akt pathway and the Nrf2 transcription factor in response to the antioxidant phytochemical carnosol. J Biol Chem 279:8919–8929
Kim D-W, Lee K, Kwon J, Lee HJ, Lee D, Mar W (2015) Neuroprotection against 6-OHDA-induced oxidative stress and apoptosis in SH-SY5Y cells by 5,7-dihydroxychromone: activation of the Nrf2/ARE pathway. Life Sci 130:25–30
Nakaso K, Nakamura C, Sato H, Imamura K, Takeshima T, Nakashima K (2006) Novel cytoprotective mechanism of anti-parkinsonian drug deprenyl: PI3K and Nrf2-derived induction of antioxidative proteins. Biochem Biophys Res Commun 339:915–922
Lin H-Y, Yeh W-L, Huang B-R, Lin C, Lai C-H, Lin H et al (2012) Desipramine protects neuronal cell death and induces heme oxygenase-1 expression in Mes23.5 dopaminergic neurons. PLoS ONE 7:e50138
Lin T-K, Chen S-D, Chuang Y-C, Lin H-Y, Huang C-R, Chuang J-H et al (2014) Resveratrol partially prevents rotenone-induced neurotoxicity in dopaminergic SH-SY5Y cells through induction of heme oxygenase-1 dependent autophagy. Int J Mol Sci 15:1625–1646
Dal-Cim T, Molz S, Egea J, Parada E, Romero A, Budni J et al (2012) Guanosine protects human neuroblastoma SH-SY5Y cells against mitochondrial oxidative stress by inducing heme oxigenase-1 via PI3K/Akt/GSK-3β pathway. Neurochem Int 61:397–404
Giordano S, Lee J, Darley-Usmar VM, Zhang J (2012) Distinct effects of rotenone, 1-methyl-4-phenylpyridinium and 6-hydroxydopamine on cellular bioenergetics and cell death. PLoS ONE 7:e44610
Innamorato NG, Rojo AI, García-Yagüe AJ, Yamamoto M, de Ceballos ML, Cuadrado A (2008) The transcription factor Nrf2 is a therapeutic target against brain inflammation. J Immunol 181:680–689
Lee I-T, Luo S-F, Lee C-W, Wang S-W, Lin C-C, Chang C-C et al (2009) Overexpression of HO-1 Protects against TNF-α-mediated airway inflammation by down-regulation of TNFR1-dependent oxidative stress. Am J Pathol 175:519–532
Schipper HM, Song W, Zukor H, Hascalovici JR, Zeligman D (2009) Heme oxygenase-1 and neurodegeneration: expanding frontiers of engagement. J Neurochem 110:469–485
Yirmiya R (2000) Depression in medical illness: the role of the immune system. West J Med 173:333–336
Martín-de-Saavedra MD, Budni J, Cunha MP, Gómez-Rangel V, Lorrio S, del Barrio L, Lastres-Becker I et al (2013) Nrf2 participates in depressive disorders through an anti-inflammatory mechanism. Psychoneuroendocrinology 38:2010–2022
Maes M, Fišar Z, Medina M, Scapagnini G, Nowak G, Berk M (2012) New drug targets in depression: inflammatory, cell-mediated immune, oxidative and nitrosative stress, mitochondrial, antioxidant, and neuroprogressive pathways. And new drug candidates—Nrf2 activators and GSK-3 inhibitors. Inflammopharmacology 20:127–150
Choi H-S, Park JH, Ahn JH, Hong S, Cho JH, Won M-H et al (2015) The anti-inflammatory activity of duloxetine, a serotonin/norepinephrine reuptake inhibitor, prevents kainic acid-induced hippocampal neuronal death in mice. J Neurol Sci 358:390–397
Acknowledgements
This study was supported by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), FAPESC (TO 22.333/2010-6). Rede IBN-Net/CNPq (IBN 01.06.0842-00), CAPES-PROCAD, CAPES-REUNI, Coordenação de Aperfeiçoamento de Pessoal de Nível Superior-CAPES. INCT-National Institute of Science and Technology for Excitotoxicity and Neuroprotection.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Engel, D.F., de Oliveira, J., Lieberknecht, V. et al. Duloxetine Protects Human Neuroblastoma Cells from Oxidative Stress-Induced Cell Death Through Akt/Nrf-2/HO-1 Pathway. Neurochem Res 43, 387–396 (2018). https://doi.org/10.1007/s11064-017-2433-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-017-2433-3