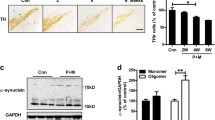
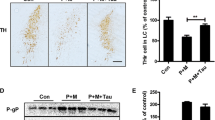
Abstract
Parkinson’s disease (PD) is a common neurodegenerative disease, and its etiology remains obscure. Increasing evidence has suggested an important role for environmental factors such as exposure to pesticides in increasing the risk of developing PD and inflammation is the early incident during the process of PD. In this study, we measure the pro-inflammatory cytokines by enzyme-linked immunosorbnent assay and RT-PCR methods; analyze the reactive oxygen species by DCFH-DA; detected nuclear factor κB (NFκB) translocation by western blot and immunofluorescence methods; and analyze the phosphorylation of mitogen-activated protein (MAP) kinase and protein level of Nurr1 by western blot. Results showed that rotenone could induce tumor neurosis factor α (TNFα) and interleukin 1β (IL-1β) release from BV-2 cells, enhance TNFα and IL-1β mRNA levels in substantia nigra lesioned by rotenone; also, rotenone could increase the phosphorylation of inhibitor of κB (IκB), extracellular regulated protein kinase , c-Jun N-terminal kinase, p38 MAP kinases and promote p65 subunit of NFκB translocation to nuclear; at the same time, rotenone could decrease the protein level of Nurr1 in nuclear. So, rotenone exerted toxicity through activating microglia, and its mechanism might be associated with NFκB signal pathway.






Similar content being viewed by others
References
Block ML, Zecca L, Hong JS (2007) Microglia-mediated neurotoxicity: uncovering the molecular mechanisms. Nat Rev Neurosci 8:57–69. doi:10.1038/nrn2038
Saijo K, Winner B, Carson CT, Collier JG, Boyer L, Rosenfeld MG, Gage FH, Glass CK (2009) A Nurr1/CoREST pathway in microglia and astrocytes protects dopaminergic neurons from inflammation-induced death. Cell 137:47–59. doi:10.1016/j.cell.2009.01.038
Mayeux R (2003) Epidemiology of neurodegeneration. Annu Rev Neurosci 26:81–104. doi:10.1146/annurev.neuro.26.043002.094919
Betarbet R, Sherer TB, MacKenzie G, Garcia-Osuna M, Panov AV, Greenamyre JT (2000) Chronic systemic pesticide exposure reproduces features of Parkinson’s disease. Nat Neurosci 3:1301–1306. doi:10.1038/81834
Bashkatova V, Alam M, Vanin A, Schmidt WJ (2004) Chronic administration of rotenone increases levels of nitric oxide and lipid peroxidation products in rat brain. Exp Neurol 186:235–241. doi:10.1016/j.expneurol.2003.12.005
Nagatsu T, Sawada M (2005) Inflammatory process in Parkinson’s disease: role for cytokines. Curr Pharm Des 11:999–1016. doi:10.2174/1381612053381620
Couch Y, Alvarez-Erviti L, Sibson NR, Wood MJ, Anthony DC (2011) The acute inflammatory response to intranigral alpha-synuclein differs significantly from intranigral lipopolysaccharide and is exacerbated by peripheral inflammation. J Neuroinflammation 8:166–179. doi:10.1186/1742-2094-8-166
Tergaonkar V (2006) NFkappaB pathway: a good signaling paradigm and therapeutic target. Int J Biochem Cell Biol 38:1647–1653. doi:10.1016/j.biocel.2006.03.023
Song JX, Shaw PC, Sze CW, Tong Y, Yao XS, Ng TB, Zhang YB (2010) Chrysotoxine a novel bibenzyl compound inhibits 6-hydroxydopamine induced apoptosis in SH-SY5Y cells via mitochondria protection and NF-kappaB modulation. Neurochem Int 57:676–689. doi:10.1016/j.neuint.2010.08.007
Li B, Chauvin C, DePaulis D, DeOliveira F, Gharib A, Vial G, Lablanche S, Leverve X, Bernardi P, Ovize M, Fontaine E (2012) Inhibition of complex I regulates the mitochondrial permeability transition through a phosphate-sensitive inhibitory site masked by cyclophilin D. Biochim Biophys Acta 1817:1628–1634. doi:10.1016/j.bbabio.2012.05.011
Zorov DB, Filburn CR, Klotz LO, Zweier JL, Sollott SJ (2000) Reactive oxygen species (ROS)-induced ROS release: a new phenomenon accompanying induction of the mitochondrial permeability transition in cardiac myocytes. J Exp Med 192:1001–1014. doi:10.1084/jem.192.7.1001
Batandier C, Leverve X, Fontaine E (2004) Opening of the mitochondrial permeability transition pore induces reactive oxygen species production at the level of the respiratory chain complex I. J Biol Chem 279:17197–17204. doi:10.1074/jbc.M310329200
Tsai HH, Lee WR, Wang PH, Cheng KT, Chen YC, Shen SC (2013) Propionibacterium acnes-induced iNOS and COX-2 protein expression via ROS-dependent NF-kappaB and AP-1 activation in macrophages. J Dermatol Sci 69:122–131. doi:10.1016/j.jdermsci.2012.10.009
Torres M, Forman HJ (2003) Redox signaling and the MAP kinase pathways. BioFactors 17:287–296. doi:10.1002/biof.5520170128
Liu S, Chen ZJ (2011) Expanding role of ubiquitination in NF-kappaB signaling. Cell Res 21:6–21. doi:10.1038/cr.2010.170
Moynagh PN (2005) The NF-kappaB pathway. J Cell Sci 118:4589–4592. doi:10.1242/jcs.02579
Singh AP et al (2012) CXCL12/CXCR4 protein signaling axis induces sonic hedgehog expression in pancreatic cancer cells via extracellular regulated kinase- and akt kinase-mediated activation of nuclear factor kappaB: implications for bidirectional tumor-stromal interactions. J Biol Chem 287:39115–39124
Zhang G, He JL, Xie XY, Yu C (2012) LPS-induced iNOS expression in N9 microglial cells is suppressed by geniposide via ERK p38 and nuclear factor-kappaB signaling pathways. Int J Mol Med 30:561–568. doi:10.1074/jbc.M112.409581
Guma M, Stepniak D, Shaked H, Spehlmann ME, Shenouda S, Cheroutre H, Vicente-Suarez I, Eckmann L, Kagnoff MF, Karin M (2011) Constitutive intestinal NF-kappaB does not trigger destructive inflammation unless accompanied by MAPK activation. J Exp Med 208:1889–1900. doi:10.1084/jem.20110242
Gantke T, Sriskantharajah S, Sadowski M, Ley SC (2012) IkappaB kinase regulation of the TPL-2/ERK MAPK pathway. Immunol Rev 246:168–182. doi:10.1111/j.1600-065X.2012.01104.x
Perlmann T, Wallen-Mackenzie A (2004) Nurr1 an orphan nuclear receptor with essential functions in developing dopamine cells. Cell Tissue Res 318:45–52. doi:10.1007/s00441-004-0974-7
Acknowledgments
This work was supported by National Natural Science Foundation of China Grants (No. 81274122, 81102831, 81073078), Special Purpose for New Drug Development (2012ZX09301002-004), Studies on Structure and function of Bioactive Substances from Natural Medicines (IRT1007), National Natural Science Foundation of Beijing Grants (7131013), Research Fund for the Doctoral Program of Higher Education of China (20121106130001).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yuan, Yh., Sun, Jd., Wu, Mm. et al. Rotenone Could Activate Microglia Through NFκB Associated Pathway. Neurochem Res 38, 1553–1560 (2013). https://doi.org/10.1007/s11064-013-1055-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-013-1055-7