Abstract
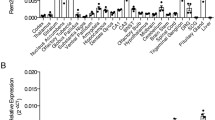
Phospholipases A2 (PLA2) are enzymes that cleave the sn-2 bond of membrane phospholipids to yield free fatty acids and lysophospholipids. Secretory PLA2-III (sPLA2-III) has been suggested to be important for neuronal differentiation, growth and survival, and is highly expressed in the spinal cord. The aim of this study is to elucidate its expression and distribution in different regions of the adult rat CNS. Quantitative RT-PCR analyses showed high levels of sPLA2-III mRNA expression in the brainstem and spinal cord and low expression in the olfactory bulb. Western blot analyses showed high level of expression in the brainstem, spinal cord and cerebral neocortex. A dense band corresponding to the catalytically active, mature/cleaved form, and a faint band corresponding to the full length sPLA2-III were detected in post-mitochondrial supernatants, from different parts of the CNS. Subcellular fractionation of spinal cord homogenates showed that sPLA2-III protein is present in the ‘light membrane/cytosol’ fraction, but not the nucleus, synaptosomal membrane or synaptic vesicle-enriched fractions. sPLA2-III was immunolocalized to neurons in the cerebral neocortex, Purkinje neurons in the cerebellar cortex, periaqueductal gray, red nucleus, spinal trigeminal nucleus and dorsal horn of the spinal cord. Electron microscopy of the spinal cord and cerebral neocortex showed that sPLA2-III was localized in dendrites or dendritic spines, that formed asymmetrical synapses with unlabeled, putatively glutamatergic, axon terminals. The localization of mature/cleaved form of sPLA2-III in postsynaptic structures suggest a physiological role of the enzyme in neurotransmission or synaptic plasticity.





Similar content being viewed by others
References
Sun GY, Shelat PB, Jensen MB, He Y, Sun AY, Simonyi A (2010) Phospholipases A2 and inflammatory responses in the central nervous system. Neuromolecular Med 12(2):133–148. doi:10.1007/s12017-009-8092-z
Dennis EA, Cao J, Hsu YH, Magrioti V, Kokotos G (2011) Phospholipase A2 enzymes: physical structure, biological function, disease implication, chemical inhibition, and therapeutic intervention. Chem Rev 111(10):6130–6185. doi:10.1021/cr200085w
Gelb MH, Valentin E, Ghomashchi F, Lazdunski M, Lambeau G (2000) Cloning and recombinant expression of a structurally novel human secreted phospholipase A2. J Biol Chem 275(51):39823–39826. doi:10.1074/jbc.C000671200
Ho IC, Arm JP, Bingham CO 3rd, Choi A, Austen KF, Glimcher LH (2001) A novel group of phospholipase A2s preferentially expressed in type 2 helper T cells. J Biol Chem 276(21):18321–18326. doi:10.1074/jbc.M008837200
Six DA, Dennis EA (2000) The expanding superfamily of phospholipase A2 enzymes: classification and characterization. Biochim Biophys Acta 1488:1–19
Murakami M, Taketomi Y, Sato H, Yamamoto K (2011) Secreted phospholipase A2 revisited. J Biochem 150(3):233–255. doi:10.1093/jb/mvr088
Goracci G, Ferrini M, Nardicchi V (2010) Low molecular weight phospholipases A2 in mammalian brain and neural cells: roles in functions and dysfunctions. Mol Neurobiol 41(2–3):274–289. doi:10.1007/s12035-010-8108-6
Valentin E, Ghomashchi F, Gelb MH, Lazdunski M, Lambeau G (2000) Novel human secreted phospholipase A2 with homology to the group III bee venom enzyme. J Biol Chem 275:7492–7496
Beck S, Beck G, Ostendorf T, Floege J, Lambeau G, Nevalainen T, Radeke HH, Gurrieri S, Haas U, Thorwart B, Pfeilschifter J, Kaszkin M (2006) Upregulation of group IB secreted phospholipase A2 and its M-type receptor in rat ANTI-THY-1 glomerulonephritis. Kidney Int 70:1251–1260
Murakami M, Masuda S, Shimbara S, Bezzine S, Lazdunski M, Lambeau G, Gelb MH, Matsukura S, Kokubu F, Adachi M, Kudo I (2003) Cellular arachidonate-releasing function of novel classes of secretory phospholipase A2s (groups III and XII). J Biol Chem 278(12):10657–10667. doi:10.1074/jbc.M211325200
Masuda S, Yamamoto K, Hirabayashi T, Ishikawa Y, Ishii T, Kudo I, Murakami M (2008) Human group III secreted phospholipase A2 promotes neuronal outgrowth and survival. Biochem J 409(2):429–438. doi:10.1042/BJ20070844
Titsworth WL, Onifer SM, Liu NK, Xu XM (2007) Focal phospholipases A2 group III injections induce cervical white matter injury and functional deficits with delayed recovery concomitant with Schwann cell remyelination. Exp Neurol 207(1):150–162. doi:10.1016/j.expneurol.2007.06.010
Sato H, Kato R, Isogai Y, Saka G, Ohtsuki M, Taketomi Y, Yamamoto K, Tsutsumi K, Yamada J, Masuda S, Ishikawa Y, Ishii T, Kobayashi T, Ikeda K, Taguchi R, Hatakeyama S, Hara S, Kudo I, Itabe H, Murakami M (2008) Analyses of group III secreted phospholipase A2 transgenic mice reveal potential participation of this enzyme in plasma lipoprotein modification, macrophage foam cell formation, and atherosclerosis. J Biol Chem 283:33483–33497
Sato H, Taketomi Y, Isogai Y, Masuda S, Kobayashi T, Yamamoto K, Murakami M (2009) Group III secreted phospholipase A2 transgenic mice spontaneously develop inflammation. Biochem J 421(1):17–27. doi:10.1042/BJ20082429
Nicolas JP, Lin Y, Lambeau G, Ghomashchi F, Lazdunski M, Gelb MH (1997) Localization of structural elements of bee venom phospholipase A2 involved in N-type receptor binding and neurotoxicity. J Biol Chem 272(11):7173–7181
Thwin MM, Ong WY, Fong CW, Sato K, Kodama K, Farooqui AA, Gopalakrishnakone P (2003) Secretory phospholipase A2 activity in the normal and kainate injected rat brain, and inhibition by a peptide derived from python serum. Exp Brain Res 150(4):427–433. doi:10.1007/s00221-003-1476-7
Ma MT, Yeo JF, Shui G, Wenk MR, Ong WY (2012) Systems wide analyses of lipids in the brainstem during inflammatory orofacial pain—evidence of increased phospholipase A2 activity. Eur J Pain 16(1):38–48
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25:402–408
Murakami M, Masuda S, Shimbara S, Ishikawa Y, Ishii T, Kudo I (2005) Cellular distribution, post-translational modification, and tumorigenic potential of human group III secreted phospholipase A2. J Biol Chem 280:24987–24998
Hallett PJ, Collins TL, Standaert DG, Dunah AW (2008) Biochemical fractionation of brain tissue for studies of receptor distribution and trafficking. Current protocols in neuroscience/editorial board, Jacqueline N Crawley [et al] Chapter 1:Unit 1, vol 16. doi:10.1002/0471142301.ns0116s42
Edwards FA (1995) Anatomy and electrophysiology of fast central synapses lead to a structural model for long-term potentiation. Physiol Rev 75:759–787
Macchioni L, Corazzi L, Nardicchi V et al (2004) Rat brain cortex mitochondria release group II secretory phospholipase A2 under reduced membrane potential. J Biol Chem 279:37860–37869
Nardicchi V, Macchioni L, Ferrini M et al (2007) The presence of a secretory phospholipase A2 in the nuclei of neuronal and glial cells of rat brain cortex. Biochim Biophys Acta 1771:1345–1352
Ferrini M, Nardicchi V, Mannucci R et al (2010) Effects of NGF on the subcellular localization of group IIA secretory phospholipase A2 (GIIA) in PC12 cells: role in neuritogenesis. Neurochem Res 35:2168–2174
Ma MT, Nevalainen TJ, Yeo JF, Ong WY (2010) Expression profile of multiple secretory phospholipase A2 isoforms in the rat CNS: enriched expression of sPLA2-IIA in brainstem and spinal cord. J Chem Neuroanat 39:242–247
Than A, Tan Y, Ong WY, Farooqui AA, Chen P (2012) Kainate receptors mediate regulated exocytosis of secretory phospholipase A2 in SH-SY5Y neuroblastoma cells. Neurosignals 20(2):72–85
Wei S, Ong WY, Thwin MM, Fong CW, Farooqui AA, Gopalakrishnakone P, Hong W (2003) Group IIA secretory phospholipase A2 stimulates exocytosis and neurotransmitter release in pheochromocytoma-12 cells and cultured rat hippocampal neurons. Neuroscience 121(4):891–898
Dorandeu F, Pernot-Marino I, Veyret J, Perrichon C, Lallement G (1998) Secreted phospholipase A2-induced neurotoxicity and epileptic seizures after intracerebral administration: an unexplained heterogeneity as emphasized with paradoxin and crotoxin. J Neurosci Res 54:848–862
Florea A, Puica C, Vintan M, Benga I, Craciun C (2011) Electrophysiological and structural aspects in the frontal cortex after the bee (Apis mellifera) venom experimental treatment. Cell Mol Neurobiol 31(5):701–714. doi:10.1007/s10571-011-9667-4
DeCoster MA, Lambeau G, Lazdunski M, Bazan NG (2002) Secreted phospholipase A2 potentiates glutamate-induced calcium increase and cell death in primary neuronal cultures. J Neurosci Res 67(5):634–645
Martínez-García A, Sastre I, Recuero M, Aldudo J, Vilella E, Mateo I, Sánchez-Juan P, Vargas T, Carro E, Bermejo-Pareja F, Rodríguez-Rodriguez E, Combarros O, Rosich-Estrago M, Frank A, Valdivieso F, Bullido MJ (2010) PLA2G3, a gene involved in oxidative stress induced death, is associated with Alzheimer’s disease. J Alzheimers Dis 22(4):1181–1187
Yang X, Sheng W, He Y, Cui J, Haidekker M, Sun GY, Lee JC (2010) Secretory phospholipase A2 type III enhances alpha-secretase-dependent amyloid precursor protein processing through alterations in membrane fluidity. J Lipid Res 51(5):957–966
Acknowledgments
This work was supported by the Neurobiology and Ageing Research Programme and the National University Health System of the National University of Singapore. The authors extend their appreciation to the Deanship of Scientific Research, King Saud University, for funding this work through the Visiting Professorship Program to WYO. We thank Professor Makoto Murakami, from the Tokyo Metropolitan Institute of Medical Science, Tokyo, Japan, for his kind gift of sPLA2-III antibody.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, H., Siddiqi, N.J., Alhomida, A.S. et al. Expression and Localization of sPLA2-III in the Rat CNS. Neurochem Res 38, 753–760 (2013). https://doi.org/10.1007/s11064-013-0974-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-013-0974-7