Abstract
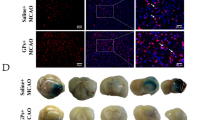
Geniposide, an iridoid glycoside isolated from Gardenia, has neuroprotective activities against oxidative stress and inflammation. The present study investigated the in vivo protective effect of geniposide on ischemia/reperfusion-injured rats by middle cerebral artery occlusion (MCAO), and the inhibitory effects of geniposide and mechanisms against activation of microglial cells by oxygen-glucose deprivation (OGD) in vitro. Male SD rats were subjected to treatment with geniposide at 15, 30 and 60 mg/kg immediately after MCAO. Cerebral infarct volume and microglial cell activation were assessed following 24 h reperfusion. Cultured primary rat microglial cells were exposed to geniposide at the concentrations of 12.5, 25 and 50 μg/mL during 4 h of OGD. The effects of geniposide were evaluated in terms of (1) cell viability; (2) secretion of TNF-α, IL-1β, IL-6, IL-8 and IL-10 into culture media; (3) TLR4 mRNA expression; (4) protein expression of TLR4, p-ERK1/2, p-IκB, p-p38, nuclear and cytoplasmic fraction NF-κB p65; and (5) nuclear transfer of NF-κB p65. Geniposide reduced the infarct volume and inhibited the activation of microglial cells in ischemic penumbra in vivo. OGD increased cell viability and release of TNF-α, IL-1β, IL-6, IL-8 and IL-10, these effects were suppressed by geniposide. Geniposide also attenuated the increases in the OGD-induced TLR4 mRNA and protein levels. In addition, geniposide at 25 and 50 μg/mL downregulated the phosphorylation of ERK, IκB and p38, as well as inhibited nuclear transcriptional activity triggered via NF-κB p65 in microglial cells by OGD. In conclusion, geniposide displays a neuroprotective effect on ischemia/reperfusion-injured rats in vivo and inhibits OGD-induced activation of microglial cells by attenuating inflammatory factors and NF-κB activation in vitro.









Similar content being viewed by others
References
Wang J, Li PT, Du H, Hou JC, Li WH, Pan YS, Chen HC (2012) Tong Luo Jiu Nao injection, a traditional Chinese medicinal preparation, inhibits MIP-1 expression in brain microvascular endothelial cells injured by oxygen-glucose deprivation. J Ethnopharmacol 141:151–157
Wang SW, Lai CY, Wang CJ (1992) Inhibitory effect of geniposide on aflatoxin B1-induced DNA repair synthesis in primary cultured rat hepatocytes. Cancer Lett 65:133–137
Chen QC, Zhang WY, Kim H, Lee IS, Ding Y, Youn UJ, Lee SM, Na M, Min BS, Bae K (2010) Effects of Gardeniae Fructus extract and geniposide on promoting ligament cell proliferation and collagen synthesis. Phytother Res Suppl 1:S1–S5
Liu J, Yin F, Zheng X, Jing J, Hu Y (2007) Geniposide, a novel agonist for GLP-1 receptor, prevents PC12 cells from oxidative damage via MAP kinase pathway. Neurochem Int 51:361–369
Peng CH, Huang CN, Hsu SP, Wang CJ (2007) Penta-acetyl geniposideinduced apoptosis involving transcription of NGF/p75 via MAPK-mediated AP-1 activation in C6 glioma cells. Toxicology 238:130–139
Liaw J, Chao YC (2001) Effect of in vitro and in vivo aerosolized treatment with geniposide on tracheal permeability in ovalbumininduced guinea pigs. Eur J Pharmacol 433:115–121
Wu SY, Wang GF, Liu ZQ, Rao JJ, Lü L, Xu W, Wu SG, Zhang JJ (2009) Effect of geniposide, a hypoglycemic glucoside, on hepatic regulating enzymes in diabetic mice induced by a high-fat diet and streptozotocin. Acta Pharmacol Sin 30:202–208
Lee P, Lee J, Choi SY, Lee SE, Lee S, Son D (2006) Geniposide from Gardenia jasminoides attenuates neuronal cell death in oxygen and glucose deprivation-exposed rat hippocampal slice culture. Biol Pharm Bull 29:174–176
Yin F, Liu J, Zheng X, Guo L, Xiao H (2010) Geniposide induces the expression of heme oxygenase-1 via PI3 K/Nrf2-signaling to enhance the antioxidant capacity in primary hippocampal neurons. Biol Pharm Bull 33:1841–1846
Liu JH, Yin F, Guo LX, Deng XH, Hu YH (2009) Neuroprotection of geniposide against hydrogen peroxide induced PC12 cells injury: involvement of PI3 kinase signal pathway. Acta Pharmacol Sin 30:159–165
Aaron YL, Kathryn GT (2006) Microglia in cerebral ischemia: molecular actions and interactions. Parmacology 84:49–59
Moon JB, Lee CH, Park CW, Cho JH, Hwang IK, Yoo KY, Choi JH, Shin HC, Won MH (2009) Neuronal degeneration and microglial activation in the ischemic dentate gyrus of the gerbil. J Vet Med Sci 71:1381–1386
Dheen ST, Kaur C, Ling EA (2007) Microglial activation and its implications in the brain diseases. Curr Med Chem 14:1189–1197
Yamazaki M, Chiba K, Satoh K (2008) Neuro2a cell death induced by 6-hydroxydopamine is attenuated by genipin. J Health Sci 54:638–644
Longa EZ, Weinstein PR, Carlson S, Cummins R (1989) Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke 20:84–91
Gulian D, Baker TJ (1986) Characterization of amoeboid microglia isolated from developing mammalian brain. J Neurosci 6:2163–2178
McCarthy KD, Devellis J (1980) Preparation of separate astroglial and oligodendroglial cell cultures from rat cerebral tissue. J Cell Biol 85:890–902
Zhang W, Smith C, Shapiro A, Monette R, Hutchison J, Stanimirovic D (1999) Increased expression of bioactive chemokines in human cerebromicrovascular endothelial cells and astrocytes subjected to simulated ischemia in vitro. J Neuroimmunol 101:148–160
Inouye H, Takeda Y, Nhishimura H, Kanomi A, Okuda T, Puff C (1988) Chemotaxonomic studies of Rubiaceous plants containing iridoid glucosides. Phytochemistry 27:2591–2598
Kostova MB, Konaklieva MI, Alipieva KI, Popov SS, Handjieva NV (2005) ESI-MS study of some C10 iridoid glycosides. Instrum Sci Technol 33:691–702
Zheng X, Yang D, Liu X, Wang N, Li B, Cao H, Lu Y, Wei G, Zhou H, Zheng J (2010) Identification of a new anti-LPS agent, geniposide, from Gardenia jasminoides Ellis, and its ability of direct binding and neutralization of lipopolysaccharide in vitro and in vivo. Int Immunopharmacol 10:1209–1219
Nam KN, Choi Y, Jung HJ, Park GH, Park JM, Moon SK, Cho KH, Kang C, Kang I, Oh MS, Lee EH (2010) Genipin inhibits the inflammatory response of rat brain microglial cells. Int Immunopharmacol 10:493–499
Jeon WK, Hong HY, Kim BC (2011) Genipin up-regulates heme oxygenase-1 via PI3-kinase-JNK1/2-Nrf2 signaling pathway to enhance the anti-inflammatory capacity in RAW264.7 macrophages. Arch Biochem Biophys 512:119–125
Vilhardt F (2005) Microglia: phagocyte and glia cell. Int J Biochem Cell Biol 37:17–21
Smith JA, Das A, Ray SK, Banik NL (2012) Role of pro-inflammatory cytokines released from microglia in neurodegenerative diseases. Brain Res Bull 87:10–20
Nishi T, Maier CM, Hayashi T, Saito A, Chan PH (2005) Superoxide dismutase 1 overexpression reduces MCP-1 and MIP-1 alpha expression after transient focal cerebral ischemia. J Cereb Blood Flow Metab 25:1312–1324
Wang J, Li PT, Du H, Hou JC, Li WH, Pan YS, Hua Q, Chen HC (2011) Impact of paracrine signals from brain microvascular endothelial cells on microglial proliferation and migration. Brain Res Bull 86:53–59
Kouadir M, Yang L, Tan R, Shi F, Lu Y, Zhang S, Yin X, Zhou X, Zhao D (2012) CD36 participates in PrP(106-126)-induced activation of microglia. PLoS One 7:e30756
Tynan RJ, Weidenhofer J, Hinwood M, Cairns MJ, Day TA, Walker FR (2012) A comparative examination of the anti-inflammatory effects of SSRI and SNRI antidepressants on LPS stimulated microglia. Brain Behav Immun 26:469–479
Wang JY, Shum AY, Chao CC, Kuo JS (2000) Production of macrophage inflammatory protein-2 following hypoxia/reoxygenation in glial cells. Glia 32:155–164
You Y, Kaur C (2000) Expression of induced nitric oxide synthase in amoeboid microglia in postnatal rats following an exposure to hypoxia. Neurosci Lett 279:101–104
Ock J, Jeong J, Choi WS, Lee WH, Kim SH, Kim IK, Suk KJ (2007) Regulation of Toll-like receptor 4 expression and its signaling by hypoxia in cultured microglia. Neurosci Res 85:1989–1995
Pradillo JM, Fernández-López D, García-Yébenes I, Sobrado M, Hurtado O, Moro MA, Lizasoain I (2009) Toll-like receptor 4 is involved in neuroprotection afforded by ischemic preconditioning. J Neurochem 109:287–294
Weinstein JR, Koerner IP, Möller T (2010) Microglia in ischemic brain injury. Future Neurol 5:227–246
Lehnardt S, Massillon L, Follett P, Jensen FE, Ratan R, Rosenberg PA, Volpe JJ, Vartanian T (2003) Activation of innate immunity in the CNS triggers neurodegeneration through a Toll-like receptor 4-dependent pathway. Proc Natl Acad Sci USA 100:8514–8519
Tanga FY, Nutile-McMenemy N, DeLeo JA (2005) The CNS role of Toll-like receptor 4 in innate neuroimmunity and painful neuropathy. Proc Natl Acad Sci USA 102:5856–5861
Beutler B (2004) Toll-like receptors and their place in immunology. Where does the immune response to infection begin? Natl Rev Immunol 4:498
Muzio M, Ni J, Feng P, Dixit VM (1997) IRAK (Pelle) family member IRAK-2 and MyD88 as proximal mediators of IL-1 signaling. Science 278:1612–1615
McDermott EP, O’Neill LA (2002) Ras participates in the activation of p38 MAPK by interleukin-1 by associating with IRAK, IRAK2, TRAF6, and TAK-1. J Biol Chem 277:7808–7815
Hoshino K, Kaisho T, Iwabe T, Takeuchi O, Akira S (2002) Differential involvement of IFN-beta in Toll-like receptor-stimulated dendritic cell activation. Int Immunol 10:1225–1231
Porter AG, Jänicke RU (1999) Emerging roles of caspase-3 in apoptosis. Cell Death Differ 2:99–104
Toshchakov V, Jones BW, Perera PY, Thomas K, Cody MJ, Zhang S, Williams BR, Major J, Hamilton TA, Fenton MJ, Vogel SN (2002) TLR4, but not TLR2, mediates IFN-beta-induced STAT1alpha/beta-dependent gene expression in macrophages. Nat Immunol 4:392–398
Karin M (2006) Nuclear factor-kappaB in cancer development and progression. Nature 441:431–436
Zhang Z, Rigas B (2006) NF-kappaB, inflammation and pancreatic carcinogenesis: NF-kappaB as a chemoprevention target (review). Int J Oncol 29:185–192
Wong ET, Tergaonkar V (2009) Roles of NF-kappaB in health and disease: mechanisms and therapeutic potential. Clin Sci (Lond) 116:451–465
Baeuerle PA, Baltimore D (1988) I kappa B: a specific inhibitor of the NF-kappa B transcription factor. Science 242:540–546
Hayden MS, Ghosh S (2008) Shared principles in NF-kappaB signaling. Cell 132:344–362
O’Neill LA (2003) The role of MyD88-like adapters in Toll-like receptor signal transduction. Biochem Soc Trans 31:643–647
Jordan MS, Singer AL, Koretzky GA (2003) Adaptors as central mediators of signal transduction in immune cells. Nat Immunol 4:110–116
O’Neill LA (2002) Toll-like receptor signal transduction and the tailoring of innate immunity: a role for Mal? Trends Immunol 23:296–300
Akira S, Takeda K (2004) Toll-like receptor signalling. Nat Rev Immunol 4:499–511
Kawai T, Adachi O, Ogawa T, Takeda K, Akira S (1999) Unresponsiveness of MyD88-deficient mice to endotoxin. Immunity 11:115–122
Acknowledgments
This research was supported by grants from the National Natural Science Foundation of China (Grant Nos. 81102679, 81073085), and the China National Science & Technology Major Project (Grant Nos. 2009ZX09103-317).
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
The authors Jun Wang and Jincan Hou contributed equally to this paper.
Rights and permissions
About this article
Cite this article
Wang, J., Hou, J., Zhang, P. et al. Geniposide Reduces Inflammatory Responses of Oxygen-Glucose Deprived Rat Microglial Cells via Inhibition of the TLR4 Signaling Pathway. Neurochem Res 37, 2235–2248 (2012). https://doi.org/10.1007/s11064-012-0852-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-012-0852-8