Abstract
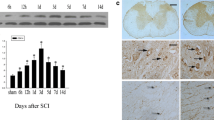
KPC1 (Kip1 ubiquitylation-promoting complex 1) is the catalytic subunit of the ubiquitin ligase KPC, which regulates the degradation of the cyclin-dependent kinase inhibitor p27kip1 at the G1 phase of the cell cycle. To elucidate the expression and role of KPC1 in nervous system lesion and repair, we performed an acute spinal cord contusion injury (SCI) model in adult rats. Western blot analysis showed a significant up-regulation of KPC1 and a concomitant down-regulation of p27kip1 following spinal injury. Immunohistochemistry and immunofluorescence revealed wide expression of KPC1 in the spinal cord, including expression in neurons and astrocytes. After injury, KPC1 expression was increased predominantly in astrocytes, which highly expressed PCNA, a marker for proliferating cells. Co-immunoprecipitation demonstrated increased interactions between p27kip1 and KPC1 4 days after injury. To understand whether KPC1 plays a role in astrocyte proliferation, we applied LPS to induce astrocyte proliferation in vitro. Western blot analysis demonstrated that p27kip1 expression was negatively correlated with KPC1 expression following LPS stimulation. Immunofluorescence analysis showed subcellular localizations of p27kip1 and KPC1 were also changed following the stimulation of astrocytes with LPS. These results suggest that KPC1 is related to the down-regulation of p27kip1; this event may be involved in the proliferation of astrocytes after SCI.







Similar content being viewed by others
References
Becker EB, Bonni A (2004) Cell cycle regulation of neuronal apoptosis in development and disease. Prog Neurobiol 72:1–25
Blight AR (1985) Delayed demyelination and macrophage invasion: a candidate for secondary cell damage in spinal cord injury. Cent Nerv Syst Trauma 2:299–315
Bloom J, Pagano M (2003) Deregulated degradation of the cdk inhibitor p27 and malignant transformation. Semin Cancer Biol 13:41–47
Brahmachari S, Fung YK, Pahan K (2006) Induction of glial fibrillary acidic protein expression in astrocytes by nitric oxide. J Neurosci 26:4930–4939
Byrnes KR, Stoica BA, Fricke S et al (2007) Cell cycle activation contributes to post-mitotic cell death and secondary damage after spinal cord injury. Brain 130:2977–2992
Carrano AC, Eytan E, Hershko A et al (1999) SKP2 is required for ubiquitin-mediated degradation of the CDK inhibitor p27. Nat Cell Biol 1:193–199
Coats S, Flanagan WM, Nourse J et al (1996) Requirement of p27Kip1 for restriction point control of the fibroblast cell cycle. Science 272:877–880
Codeluppi S, Svensson CI, Hefferan MPF et al (2009) The Rheb-mTOR pathway is upregulated in reactive astrocytes of the injured spinal cord. J Neurosci 29:1093–1104
Davies SJ, Field PM, Raisman G (1996) Regeneration of cut adult axons fails even in the presence of continuous aligned glial pathways. Exp Neurol 142:203–216
Di Giovanni S, Movsesyan V, Ahmed F et al (2005) Cell cycle inhibition provides neuroprotection and reduces glial proliferation and scar formation after traumatic brain injury. Proc Natl Acad Sci U S A 102:8333–8338
Dumont RJ, Okonkwo DO, Verma S et al (2001) Acute spinal cord injury, part I: pathophysiologic mechanisms. Clin Neuropharmacol 24:254–264
Fawcett JW (1997) Astrocytic and neuronal factors affecting axon regeneration in the damaged central nervous system. Cell Tissue Res 290:371–377
Fero ML, Rivkin M, Tasch M et al (1996) A syndrome of multiorgan hyperplasia with features of gigantism, tumorigenesis, and female sterility in p27(Kip1)-deficient mice. Cell 85:733–744
Fillies T, Woltering M, Brandt B et al (2007) Cell cycle regulating proteins p21 and p27 in prognosis of oral squamous cell carcinomas. Oncol Rep 17:355–359
Go HS, Shin CY, Lee SH et al (2009) Increased proliferation and gliogenesis of cultured rat neural progenitor cells by lipopolysaccharide-stimulated astrocytes. Neuroimmunomodulation 16:365–376
Gruner JA (1992) A monitored contusion model of spinal cord injury in the rat. J Neurotrauma 9:123–126 discussion 126–128
Hengst L, Reed SI (1996) Translational control of p27Kip1 accumulation during the cell cycle. Science 271:1861–1864
Kamura T, Hara T, Matsumoto M et al (2004) Cytoplasmic ubiquitin ligase KPC regulates proteolysis of p27(Kip1) at G1 phase. Nat Cell Biol 6:1229–1235
Kotoshiba S, Kamura T, Hara T et al (2005) Molecular dissection of the interaction between p27 and Kip1 ubiquitylation-promoting complex, the ubiquitin ligase that regulates proteolysis of p27 in G1 phase. J Biol Chem 280:17694–17700
Kwon BK, Tetzlaff W, Grauer JN et al (2004) Pathophysiology and pharmacologic treatment of acute spinal cord injury. Spine J 4:451–464
McDonald JW, Sadowsky C (2002) Spinal-cord injury. Lancet 359:417–425
McGraw J, Hiebert GW, Steeves JD (2001) Modulating astrogliosis after neurotrauma. J Neurosci Res 63:109–115
McMillian MK, Thai L, Hong JS et al (1994) Brain injury in a dish: a model for reactive gliosis. Trends Neurosci 17:138–142
Minamishima YA, Nakayama K (2002) Recovery of liver mass without proliferation of hepatocytes after partial hepatectomy in Skp2-deficient mice. Cancer Res 62:995–999
Morris GF, Mathews MB (1989) Regulation of proliferating cell nuclear antigen during the cell cycle. J Biol Chem 264:13856–13864
Nakayama K, Ishida N, Shirane M et al (1996) Mice lacking p27(Kip1) display increased body size, multiple organ hyperplasia, retinal dysplasia, and pituitary tumors. Cell 85:707–720
Nieto-Sampedro M (1999) Neurite outgrowth inhibitors in gliotic tissue. Adv Exp Med Biol 468:207–224
Pagano M, Tam SW, Theodoras AM et al (1995) Role of the ubiquitin-proteasome pathway in regulating abundance of the cyclin-dependent kinase inhibitor p27. Science 269:682–685
Raghupathi R (2004) Cell death mechanisms following traumatic brain injury. Brain Pathol 14:215–222
Ridet JL, Malhotra SK, Privat A et al (1997) Reactive astrocytes: cellular and molecular cues to biological function. Trends Neurosci 20:570–577
Sekhon LH, Fehlings MG (2001) Epidemiology, demographics, and pathophysiology of acute spinal cord injury. Spine (Phila Pa 1976) 26:S2–S12
Sgambato A, Cittadini A, Faraglia B et al (2000) Multiple functions of p27(Kip1) and its alterations in tumor cells: a review. J Cell Physiol 183:18–27
Shen A, Liu Y, Zhao J et al (2008) Temporal-spatial expressions of p27kip1 and its phosphorylation on Serine-10 after acute spinal cord injury in adult rat: Implications for post-traumatic glial proliferation. Neurochem Int 52:1266–1275
Shirane M, Harumiya Y, Ishida N et al (1999) Down-regulation of p27(Kip1) by two mechanisms, ubiquitin-mediated degradation and proteolytic processing. J Biol Chem 274:13886–13893
Signoretti S, Di Marcotullio L, Richardson A et al (2002) Oncogenic role of the ubiquitin ligase subunit Skp2 in human breast cancer. J Clin Invest 110:633–641
Silver J, Miller JH (2004) Regeneration beyond the glial scar. Nat Rev Neurosci 5:146–156
Sun LL, Cheng C, Liu HO et al (2007) Src suppressed C kinase substrate regulates the lipopolysaccharide-induced TNF-alpha biosynthesis in rat astrocytes. J Mol Neurosci 32:16–24
Tator CH, Fehlings MG (1991) Review of the secondary injury theory of acute spinal cord trauma with emphasis on vascular mechanisms. J Neurosurg 75:15–26
Tawfik VL, Lacroix-Fralish ML, Bercury KK et al (2006) Induction of astrocyte differentiation by propentofylline increases glutamate transporter expression in vitro: heterogeneity of the quiescent phenotype. Glia 54:193–203
Wyndaele M, Wyndaele JJ (2006) Incidence, prevalence and epidemiology of spinal cord injury: what learns a worldwide literature survey? Spinal Cord 44:523–529
Young W (1993) Secondary injury mechanisms in acute spinal cord injury. J Emerg Med 11(Suppl 1):13–22
Tomoda K, Kubota Y, Kato J (1999) Degradation of the cyclin-dependent-kinase inhibitor p27Kip1 is instigated by Jab1. Nature 398:160–165
Acknowledgments
This work was supported by the National Natural Science Foundation of China (No. 30770488, No. 30870320, No. 31070723, No. 81071487 and No. 81070275), Natural Science Foundation of Jiangsu Province (No. BK2009156, No. BK2009157 and No. BK2009161; BK2009169), the 333 high-level personnel training project of Jiangsu Province (No. BRA2010137) and the Natural Science Foundation of Zhejiang Province (Y2090545).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Jian Zhao and Shuangwei Zhang contributed equally to this work.
Rights and permissions
About this article
Cite this article
Zhao, J., Zhang, S., Wu, X. et al. KPC1 Expression and Essential Role After Acute Spinal Cord Injury in Adult Rat. Neurochem Res 36, 549–558 (2011). https://doi.org/10.1007/s11064-010-0377-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-010-0377-y