Abstract
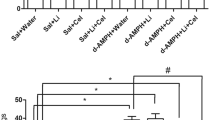
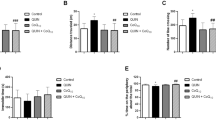
Decreased antioxidant activity is considered as one of the causes of tardive dyskinesia in schizophrenic patients in a prolonged neuroleptic treatment course. Haloperidol (HAL) has been hypothesized to increase oxidative stress, while clozapine (CLO) would produce less oxidative damage. The objective was to determine whether CLO for 28 days could reverse or attenuate HAL-induced oxidative damage in animals previously treated with HAL for 28 days. HAL significantly increased thiobarbituric acid reactive substances levels in the cortex (CX) and striatum and increased protein carbonyls in hippocampus (HP) and CX and this was not attenuated by CLO treatment. In the total radical trapping antioxidant parameter assay there was a decrease in the HP total antioxidant potential induced by HAL and by treatment with HAL + CLO. Our findings demonstrated that the atypical antipsychotic CLO could not revert oxidative damage caused by HAL.




Similar content being viewed by others
References
Morgenstern H, Glazer WM (1993) Identifying risk factors for tardive dyskinesia among long-term outpatients maintained with neuroleptic medications. Arch Gen Psychiatry 50:723–733
Raja M (1995) Tardive dystonia: prevalence, risk factors and comparison with tardive dyskinesia in a population of two hundred acute psychiatric in patients. Eur Arch Psychiatry Clin Neurosci 245:145–151
Andreassen OA et al (2003) Oral dyskinesias and histopathological alterations in substantia nigra after long-term haloperidol treatment of old rats. Neuroscience 122:717–725
Sachdev P, Saharov T, Cathcart S (1999) The preventive role of antioxidants (selegiline and vitamin E) in a rat model of tardive dyskinesia. Biol Psychiatry 46:1672–1681
Jeste DV, Lacro JP, Baily A et al (1999) Lower incidence of tardive dyskinesia with rispiridone compared with haloperidol in older patients. J Am Geriatr Soc 47:716–719
Paulson GW (2005) Historical comments on tardive dyskinesia: a neurologist’s perspective. J Clin Psychiatry 66:260–264
Correll CU, Leucht S, Kane JM (2004) Lower risk for tardive dyskinesia associated with second-generation antipsychotics: a systematic review of 1-year studies. Am J Psychiatry 161:414–425
Westerink BHC, Vries JB (1989) On the mechanism of neuroleptic induced increase in striatal dopamine release: brain dialysis provides direct evidence of mediation by autoreceptors localized on nerve terminals. Neurosci Lett 99:197–202
Wayner DDM, Burton GW, Ingold KU et al (1985) Quantitative measurement of the total, peroxyl radical-trapping antioxidant capability of human blood plasma by controlled peroxidation. The important contribution made by plasma proteins. FEBS Lett 187:33–37
Yokoyama H, Kasai N, Ueda Y et al (1998) In vivo analysis of hydrogen peroxide and lipid radicals in the striatum of rats under long-term administration of a neuroleptic. Free Radic Biol Med 26:1056–1060
Tsai G, Goff DC, Chang RW et al (1998) Markers of glutamatergic neurotransmission and oxidative stress associated with tardive dyskinesia. Am J Psychiatry 155:1207–1213
Gupta S, Mosnik D, Black DW et al (1999) Tardive dyskinesia: review of treatments past, present, and future. Ann Clin Psychiatry 11:257–266
Soares KV, McGrath JJ (1999) The treatment of tardive dyskinesia—a systematic review and meta-analysis. Schizophr Res 39:1–16
Elkashef AM, Wyatt RJ (1999) Tardive dyskinesia: possible involvement of free radicals and treatment with vitamin E. Schizophr Bull 25:731–740
Andreassen OA, Jùrgensenb HA (2000) Neurotoxicity associated with neuroleptic-induced oral dyskinesias in rats: implications for tardive dyskinesia? Prog Neurobiol 61:525–541
Vilner BJ, DeCosta BR, Bowen WD (1995) Cytotoxic effects of sigma ligants: sigma receptor mediated alteration in cellular morphology and viability. J Neurosci 15:117–134
Polydoro M, Schröder N, Noemia M et al (2004) Haloperidol- and clozapine-induced oxidative stress in the rat brain. Pharmacol Biochem Behavior 78:751–756
Reinke A, Martins MR, Lima MS et al (2004) Haloperidol and clozapine, but not olanzapine, induces oxidative stress in rat brain. Neurosci Lett 372:157–160
Creese I, Burt D, Snyder SH (1976) Dopamine receptor binding predicts clinical and pharmacological potencies of antischizophrenic drugs. Science 192:481–483
Fang J, Lai CT, Yu PH (1996) Neurotoxic effect of 4-(4-chlorophenyl)-1-(4-(4-fluorophenyl)-4-oxobutyl(-pyridinium) (HP+), a major metabolite of haloperidol in the dopaminergic system in vitro and in vivo. Biog Amines 12:125–134
See RE (1991) Striatal dopamine metabolism increases during long-term haloperidol administration in rats but shows tolerance in response to acute challenge with raclopride. Neurosci Lett 129:265–268
Shivakumar BR, Ravindranath V (1993) Oxidative stress and thiol modification induced by chronic administration of haloperidol. J Pharmacol Exp Ther 265:1137–1141
Tuunainen A, Wahlbeck K, Gilbody S (2002) Newer atypical antipsychotic medication in comparison to clozapine: a systematic review of randomized trials. Schizophr Res 56:1–10
Daly DA, Moghaddam B (1993) Actions of clozapine and haloperidol on the extracellular levels of excitotory amino acids in the prefrontal cortex and striatum of conscious rats. Neurosci Lett 152:61–64
Bassitt DP, Louza Neto MR (1998) Clozapine efficacy in tardive dyskinesia in schizophrenic patients. Eur Arch Psychiatry Clin Neurosci 248:209–211
Parikh V, Khan MM, Mahadik SP (2003) Differential effects of antipsychotics on expression of antioxidant enzymes and membrane lipid peroxidation in rat brain. J Psychiatry Res 37:43–51
Gama CS, Salvador M, Andreazza AC et al (2006) Elevated serum superoxide dismutase and thiobarbituric acid reactive substances in schizophrenia: a study of patients treated with haloperidol or clozapine. Prog Neuropsychopharmacol Biol Psychiatry 30:512–515
Draper HH, Hadley M (1990) Malondialdehyde determination as index of lipid peroxidation. Methods Enzymol 186:421–431
Levine RL, Garland D, Oliver CN (1990) Determination of carbonyl content in oxidatively modified proteins. Methods Enzymol 186:464–478
Wayner DDM, Burton GW, Ingold KU et al (1985) Quantitative measurement of the total, peroxyl radical-trapping antioxidant capability of human blood plasma by controlled peroxidation. The important contribution made by plasma proteins. FEBS Lett 187:33–37
Lowry OH, Rosebrough AL, Randall RJ (1951) Protein measurement with the folin phenol reagent. J Biol Chem 193:265–275
Bannister JV, Calaberese L (1987) Assays for SOD. Methods Biochem Anal 32:279–312
Dal-Pizzol F, Klamt F, Bernard EA (2001a) Retinol supplementation induces oxidative stress and modulates antioxidant enzyme activities in rat Sertoli cells. Free Radic Res 34:395–404
Dal-Pizzol F, Klamt F, Frota MLC Jr et al (2001b) Neonatal iron exposure induces oxidative stress in adult Wistar rat. Dev Brain Res 130:109–114
Cadet JL, Lohr JB, Jeste DV (1986) Free radicals and tardive dyskinesias. Trends Neurosci 9:107–108
Creese G, Burt D, Synder SIL (1976) Dopamine receptor binding and pharmacological potencies and antischizophrenic drugs. Science 192:481–483
Coyle JT, Puttfarcken P (1993) Oxidative stress glutamate and neurodegenerative disorders. Science 262:689–695
Ravindranath V, Reed DJ (1990) Glutathione depletion and formation of glutathione protein mixed disulfide following exposure of brain mitochondria to oxidative stress. Biochem Biophys Res Commun 169:150–158
Nielsen EB, Lyon M (1978) Evidence for cell loss in corpus striatum after long-term treatment with a neuroleptic drug (flupenithixol) in rats. Psychopharmacology 59:85–89
Subramanyam B, Rollema H, Woolf T (1990) Identification of a potentially neurotoxic pyridinium metabolite of haloperidol in rats. Biochem Biophys Res Commun 166:238–244
Chang WH, Jann MW, Chiang TS (1996) Plasma haloperidol and reduced haloperidol concentrations in a geriatric population. Neuropsychobiology 33:12–16
Galili R, Mosberg, Gil-Ad I, Weizman A (2000) Haloperidol-induced neurotoxicity—possible implications for tardive dyskinesia. J Neural Transm 107(4):479–490
de Leon J, Moral L, Camunas C (1991) Clozapine and jaw dyskinesia: a case report. J Clin Psychiatry 52:494–495
Arnaiz SL, Coronel MF, Boveris A (1999) Nitric oxide, superoxide and hydrogen peroxide production in brain mitochondria after haloperidol treatment. Nitric Oxide 3:235–243
Dal-Pizzol F, Klamt F, Bernard EA et al (2001) Retinol supplementation induces oxidative stress and modulates antioxidant enzyme activities in rat Sertoli cells. Free Radic Res 34:395–404
Dal-Pizzol F, Klamt F, Frota Jr MLC et al (2001) Neonatal iron exposure induces oxidative stress in adult Wistar rat. Dev Brain Res 130:109–114
Dal-Pizzol F, Klamt F, Vianna MMR et al (2000) Lipid peroxidation in hippocampus early and late after status epilepticus induced by pilocarpine or kainic acidin Wistar rats. Neurosci Lett 291:179–182
Polydoro M, Schröder N, Noemia MM et al (2004) Haloperidol- and clozapine-induced oxidative stress in the rat brain. Pharmacol Biochem Behav 78:751–756
Desco M, Gispert JD, Reig S et al (2003) Cerebral metabolic patterns in chronic and recent-onset schizophrenia. Psychiatry Res 122:125–135
Halliwell B, Gutteridge JMC (1989) Lipid peroxidation: a radical chain reaction. In: Free radicals biology and medicine, Oxford University Press, New York, pp 188–276
Phillis J (1994) ‘‘A radical’’ view of cerebral ischemic injury. Prog Neurobiol 42:441–448
Frederickson CJ, Cuajungco CJ, Labuda J (2002) Nitric oxide causes apparent release of zinc from presynaptic boutons. Neuroscience 115:471–474
Acknowledgments
This work was supported by grants from CNPq, FAPESC, Instituto Cérebro e Mente and UNESC (Brazil).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Agostinho, F.R., Jornada, L.K., Schröder, N. et al. Effects of Chronic Haloperidol and/or Clozapine on Oxidative Stress Parameters in Rat Brain. Neurochem Res 32, 1343–1350 (2007). https://doi.org/10.1007/s11064-007-9311-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-007-9311-3