Abstract
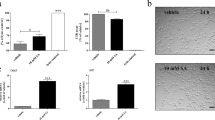
In order to further characterize the still unknown mechanism of cuprizone-induced demyelination, we investigated its effect on rat primary oligodendroglial cell cultures. Cell viability was not significantly affected by this treatment. However, when concentrations of IFNγ and/or TNFα having no deleterious effects per se on cell viability were added together with cuprizone, cell viability decreased significantly. In mitochondria isolated from cuprizone-treated glial cells, we observed a marked decrease in the activities of the various complexes of the respiratory chain, indicating a disruption of mitochondrial function. An enhancement in oxidant production was also observed in cuprizone and/or TNFα-treated oligodendroglial cells. In in vivo experiments, inhibition of microglial activation with minocycline prevented cuprizone-induced demyelination. Based on the above-mentioned results we suggest that these microglial cells appear to have a very active role in cuprizone-induced oligodendroglial cell death and demyelination, through the production and secretion of pro-inflammatory cytokines.









Similar content being viewed by others
References
Blakemore WF (1972) Observations on oligodendrocyte degeneration, the resolution of status spongiosus and remyelination in cuprizone intoxication in mice. J Neurocytol 1:413–426
Blakemore WF (1973) Remyelination of the superior cerebellar peduncle in the mouse following demyelination induced by feeding cuprizone. J Neurol Sci 20:73–83
Morell P, Barrett CV, Mason JL, Toews AD, Hostettler JD, Knapp GW, Matsushima GK (1998). Gene expression in brain during cuprizone induced demyelination and remyelination. Mol Cell Neurosci 12:220–227
Suzuki K, Kikkawa Y (1969) Status spongiosus of CNS and hepatic changes induced by cuprizone (biscyclohexanone oxalyldihydrazone). Am J Pathol 54:307–325
Hemm RD, Carlton WW, Welser JR (1971) Ultrastructural changes of cuprizone encephalopathy in mice. Toxicol Appl Pharmacol 18:869–882
Ludwin SK (1978) Central nervous system demyelination and remyelination in the mouse: an ultrastructural study of cuprizone toxicity. Lab Invest 39:597–612
Carleton WW (1967) Studies on the induction of hydrocephalus and spongy degeneration by cuprizone feeding and attempts to antidote the toxicity. Life Sci 6:11–19
Carey EM, Freeman NM (1983) Biochemical changes in cuprizone induced spongiform encephalopathy. I. Changes in the activities of 2’,3’-cyclic nucleotide 3’-phosphohydrolase, oligodendroglial ceramide galactosyl transferase, and the hydrolysis of the alkenyl group of alkenyl, acyl-glycerophospholipids by plasmalogenase in different regions of the brain. Neurochem Res 6:1029–1044
Hiremath MM, Saito Y, Knapp GW, Ting JP, Suzuki K, Matsushima GK (1998) Microglial/macrophage accumulation during cuprizone-induced demyelination in C57BL/6 mice. J Neuroimmunol 92:38–49
Shohami E, Ginis I, Hallenbeck JM (1999) Dual role of tumor necrosis factor alpha in brain injury. Cytokine Growth Factor Rev 10:119–130
Liu C, Li Wan Po A, Blumhardt LD (1998) “Summary measure” statistic for assessing the outcome of treatment trials in relapsing-remitting multiple sclerosis. J Neurol Neurosurg Psychiatry 64:726–729
Kassiotis G, Kollias G (2001) TNF and receptors in organ-specific autoimmune disease: multi-layered functioning mirrored in animal models. J Clin Invest 107:1507–1508
Buntinx M, Moreels M, Vandenabeele F, Lambrichts I, Raus J, Steels P, Stinissen P, Ameloot M (2004) Cytokine-induced cell death in human oligodendroglial cell lines: I. Synergistic effects of IFN-gamma and TNF-alpha on apoptosis. J Neurosci Res 76:834–845
Cammer W (2002) Protection of cultured oligodendrocytes against tumor necrosis factor-alpha by the antioxidants coenzymes Q (10) and N-acetyl cysteine. Brain Res Bull 58:587–592
Jurewicz A, Matysiak M, Tybor K, Selmaj K (2003) TNF-induced death of adult human oligodendrocytes is mediated by c-jun NH2-terminal kinase-3 Brain 126:1358–1370
Cammer W (1999) The neurotoxicant cuprizone retards the differentiation of oligodendrocytes in vitro. J Neurol Sci 168:116–120
McCarthy KD, de Vellis J (1980) Preparation of separate astroglial and oligodendroglial cell cultures from rat cerebral tissue. J Cell Biol 85:890–902
Casaccia-Bonnefil P, Aibel L, Chao MV (1996) Central glial and neuronal populations display differential sensitivity to ceramide-dependent cell death. J Neurosci Res 43:382–389
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65:55–63
Oberhammer F, Fritsch G, Schmied M, Pavelka M, Printz D, Purchio T, Lassmann H, Schulte-Hermann R (1993) Condensation of the chromatin at the membrane of an apoptotic nucleus is not associated with activation of an endonuclease. J Cell Sci 104:317–326
De Robertis EDF, Pellegrino de Iraldi A, Rodriguez de Lorez Arnaiz G, Salganicoff L (1962) Cholinergic and non-cholinergic nerve ending in rat brain. J Neurochem 9:23–35
Morais Cardoso S, Pereira C, Resende Oliveira C (1999) Mitochondrial function is differentially affected upon oxidative stress. Free Radic Biol Med 26:3–13
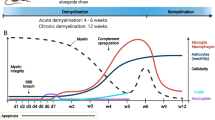
Matsushima GK, Morell P (2001) The neurotoxicant, cuprizone, as a model to study demyelination and remyelination in the central nervous system. Brain Pathol 11:107–116
Giuliani F, Fu SA, Metz LM, Yong VW (2005) Effective combination of minocycline and interferon-beta in a model of multiple sclerosis. J Neuroimmunol 165:83–91
Adamo AM, Paez PM, Escobar Cabrera OE, Wolfson M, Franco PG, Pasquini JM, Soto EF (2006) Remyelination after cuprizone-induced demyelination in the rat is stimulated by apotransferrin. Exp Neurol 198:519–529
Arnett H, Mason J, Marino M, Suzuki K, Matsushima GK, Ting JP (2001) TNF alpha promotes proliferation of oligodendrocyte progenitors and remyelination. Nat Neurosci 4:1116–1122
Selmaj K, Raine CS, Cannella B, Brosnan CF (1991) Identification of lymphotoxin and tumor necrosis factor in multiple sclerosis lesions. J Clin Invest 87:949–954
Vartanian T, Li Y, Zhao M, Stefansson K (1995) Interferon-gamma-induced oligodendrocyte cell death: implications for the pathogenesis of multiple sclerosis. Mol Med 1:732–743
D’Souza SD, Bonetti B, Balasingam V, Cashman NR, Barker PA, Troutt AB, Raine CS, Antel JP (1996) Multiple sclerosis: Fas signaling in oligodendrocyte cell death. J Exp Med 184:2361–2370
Martino G, Consiglio A, Franciotta DM, Corti A, Filippi M, Vandenbroeck K, Sciacca FL, Comi G, Grimaldi LM (1997) Tumor necrosis factor alpha and its receptors in relapsing-remitting multiple sclerosis. J Neurol Sci 152:51–61
Debruyne J, Philippe J, Dereuck J, Willems A, Leroux-Roels G (1998) Relapse markers in multiple sclerosis: are in vitro cytokine production changes reflected by circulatory T-cell phenotype alterations? Mult Scler 4:193–197
Vandevyver C, Motmans K, Stinissen P, Zhang J, Raus J (1998) Cytokine mRNA profile of myelin basic protein reactive T-Cell clones in patients with multiple sclerosis. Autoimmunity 28:77–89
Merrill JE (1991) Effects of interleukin-1 and tumor necrosis factor-alpha on astrocytes, microglia, oligodendrocytes, and glial precursors in vitro. Dev Neurosci 13:130–137
Giulian D, Ingeman JE (1988) Colony-stimulating factors as promoters of ameboid microglia. J Neurosci 8:4707–4717
Théry C, Mallat M (1993) Influence of interleukin-1 and tumor necrosis factor alpha on the growth of microglial cells in primary cultures of mouse cerebral cortex: involvement of colony-stimulating factor 1. Neurosci Lett 150:195–199
Dopp JM, Mackenzie-Graham A, Otero GC, Merrill JE (1997) Differential expression, cytokine modulation, and specific functions of type-1 and type-2 tumor necrosis factor receptors in rat glia. J Neuroimmunol 75:104–112
Merrill JE, Ignarro LJ, Sherman MP, Melinek J, Lane TE (1993) Microglial cell cytotoxicity of oligodendrocytes is mediated through nitric oxide. J Immunol 151:2132–2141
Soliven B, Szuchet S (1995) Signal transduction pathways in oligodendrocytes: role of tumor necrosis factor-alpha. Int J Dev Neurosci 13:351–367
Selmaj KW, Raine CS (1988) Tumor necrosis factor mediates myelin and oligodendrocyte damage in vitro. Ann Neurol 23:339–346
Goossens V, Grooten J, De Vos K, Fiers W (1995) Direct evidence for tumor necrosis factor-induced mitochondrial reactive oxygen intermediates and their involvement in cytotoxicity. Proc Natl Acad Sci USA 92:8115–8119
Green DR, Reed JC (1998) Mitochondria and apoptosis. Science 281:1309–1312
Chew LJ, King WC, Kennedy A, Gallo V (2005) Interferon-gamma inhibits cell cycle exit in differentiating oligodendrocyte progenitor cells. Glia 52:127–143
Hisahara S, Shoji S, Okano H, Miura M (1997) ICE/CED-3 family executes oligodendrocyte apoptosis by tumor necrosis factor. J Neurochem 69:10–20
Hsu H, Shu H, Pan M, Goeddel D (1996) TRADDTRAF2 and TRADD-FADD interactions define two distinct TNF receptor 1 signal transduction pathways. Cell 84:299–308
Rothe M, Wong S, Henzel W, Goeddel D (1994) A novel family of putative signal transducers associated with the cytoplasmic domain of the 75 kDa tumor necrosis factor receptor. Cell 78:681–692
Yrjanheikki J, Keinanen R, Pellikka M, Hokfelt T, Koistinaho J (1998) Tetracyclines inhibit microglial activation and are neuroprotective in global brain ischemia. Proc Natl Acad Sci USA 95:15769–15774
Yrjanheikki J, Tikka T, Keinanen R, Goldsteins G, Chan PH, Koistinaho J (1999) A tetracycline derivative, minocycline, reduces inflammation and protects against focal cerebral ischemia with a wide therapeutic window. Proc Natl Acad Sci USA 96:13496–13500
Brundula V, Rewcastle NB, Metz LM, Bernard CC, Yong VW (2002) Targeting leukocyte MMPs and transmigration: minocycline as a potential therapy for multiple sclerosis. Brain 125:1297–1308
Power C, Henry S, Del Bigio MR, Larsen PH, Corbett D, Imai Y, Yong VW, Peeling J (2003) Intracerebral hemorrhage induces macrophage activation and matrix metalloproteinases. Ann Neurol 53:731–742
Chen M, Ona VO, Li M, Ferrante RJ, Fink KB, Zhu S, Bian J, Guo L, Farrell LA, Hersch SM, Hobbs W, Vonsattel JP, Cha JH, Friedlander RM (2000) Minocycline inhibits caspase-1 and caspase-3 expression and delays mortality in a transgenic mouse model of Huntington disease. Nat Med 6:797–801
Zhu S, Stavrovskaya IG, Drozda M, Kim BYS, Ona V, Li M, Sarang S, Liu AS, Hartley DM, Wu DC, Gullans S, Ferrante RJ, Przedborski S, Kristal BS, Friedlander RM (2002) Minocycline inhibits cytochrome c release and delays progression of amyotrophic lateral sclerosis in mice. Nature 417:74–78
Wang X, Zhu S, Drozda M, Zhang W, Stavrovskaya IG, Cattaneo E, Ferrante RJ, Kristal BS, Friedlander RM (2003) Minocycline inhibits caspase-independent and -dependent mitochondrial cell death pathways in models of Huntington’s disease. Proc Natl Acad Sci USA 100:10483–10487
Fan LW, Pang Y, Lin S, Tien LT, Ma T, Rhodes PG, Cai Z (2005) Minocycline reduces lipopolysaccharide induced neurological dysfunctions and brain injury in the neonatal rat. J Neurosci Res 82:71–82
Cai Z, Lin S, Fan LW Pang Y, Rhodes PG (2006) Minocycline alleviates hypoxic-ischemic injury to developing oligodendrocytes in the neonatal rat brain. Neuroscience 137:425–435
Acknowledgments
Contract grant sponsor: Universidad de Buenos Aires B114 and Agencia Nacional de Promoción de Ciencia y Tecnología (PICT 12282). The authors wish to express their gratitude to Dra. Laura Pardo from FUNDALEU, for her help with the flow cytometry data.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work is dedicated with sincere friendship to Celia and Tony Campagnoni.
Rights and permissions
About this article
Cite this article
Pasquini, L.A., Calatayud, C.A., Bertone Uña, A.L. et al. The Neurotoxic Effect of Cuprizone on Oligodendrocytes Depends on the Presence of Pro-inflammatory Cytokines Secreted by Microglia. Neurochem Res 32, 279–292 (2007). https://doi.org/10.1007/s11064-006-9165-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-006-9165-0