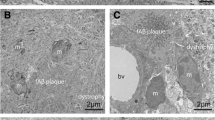
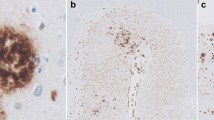
This study confirms the presence of iron, co-localized with Aβ plaques, in PS/APP mouse brain, using Perls‘ stain for Fe3+ supplemented by 3,3′-diaminobenzidine (DAB) and Aβ immunohistochemistry in histological brains sections fixed with formalin or methacarn. In this study, the fixation process and the slice thickness did not interfere with the Perls’ technique. The presence of iron in ß-amyloid plaques in PS/APP transgenic mice, a model of Alzheimer’s disease (AD) pathology, may explain previous reports of reductions of transverse relaxation time (T2) in MRI studies and represent the source of the intrinsic Aß plaque MR contrast in this model.
Similar content being viewed by others
References
J. A. Hill (1988) The distribution of iron in the brain M. B. H. Youdim (Eds) Brain iron: Neurochemistry and behavioural aspects Taylor & Francis London 1–24
J. L. Beard J. R. Connor B. C. Jones (1993) ArticleTitleIron in the brain. Nutr. Rev 51 157–170
A. H. Koeppen (2003) ArticleTitleA brief history of brain iron research. J. Neurol. Scis 207 95–97
A. Takeda (2001) ArticleTitleSignificance of transferring in iron delivery to the brain. J. Health Sci 47 323–331
J. R. Burdo D. A. Antonetti E. B. Wolpert J. R. Connor (2003) ArticleTitleMechanisms and regulation of transferrin and iron transport in a model blood-brain barrier system. Neuroscience 121 883–890
M. A. Smith P. L. Harris L. M. Sayre G. Perry (1997) ArticleTitleIron accumulation in Alzheimer disease is a source of redox-generated free radicals. Proc. Natl. Acad. Sci. USA 94 9866–9868
A. I. Bush (2003) ArticleTitleThe metallobiology of Alzheimer’s disease. Trends Neurosci 26 207–214 Occurrence Handle10.1016/S0166-2236(03)00067-5 Occurrence Handle1:CAS:528:DC%2BD3sXis12nsb8%3D Occurrence Handle12689772
B. M. Todorich J. R. Connor (2004) ArticleTitleRedox metals in Alzheimer’s disease. Ann. NY Acad. Sci 1012 171–178
K. J. Thompson S. Shoham J. R. Connor (2001) ArticleTitleIron and neurodegenerative disorders. Brain Res. Bull 55 155–64
J. C. Sipe P. Lee E. Beutler (2002) ArticleTitleBrain Iron Metabolism and Neurodegenerative Disorders. Dev. Neurosci 24 188–196
G. M. Bishop S. R. Robinson Q. Liu G. Perry C. S. Atwood M. A. Smith (2002) ArticleTitleIron: a pathological mediator of Alzheimer disease? Dev. Neurosci 24 184–187
M. A. Lovell J. D. Robertson W. J. Teesdale J. L. Campbell W. R. Markesbery (1998) ArticleTitleCopper, iron and zinc in Alzheimer’s disease senile plaques. J. Neurol. Sci 158 47–52
C. S. Atwood R. N. Martins M. A. Smith G. Perry (2002) ArticleTitleSenile plaque composition and posttranslational modification of amyloid-beta peptide and associated proteins. Peptides 23 1343–1350
S. M. LeVine (1997) ArticleTitleIron deposits in multiple sclerosis and Alzheimer’s disease brains. Brain Res 760 298–303
J. R. Connor S. L. Menzies S. M. St. Martin E. J. Mufson (1992) ArticleTitleA histochemical study of iron, transferrin, and ferritin in Alzheimer’s diseased brains. J. Neurosci. Res 31 75–83
J. Nguyen-Legros J. Bizot M. Bolesse J. P. Pulicani (1980) ArticleTitle“Diaminobenzidine black” as a new histochemical demonstration of exogenous iron. Histochemistry 66 239–244 Occurrence Handle1:CAS:528:DyaL3cXkvFWnsrc%3D Occurrence Handle7399970
D. P. Perl P. F. Good (1992) ArticleTitleComparative Techniques for Determining Cellular Iron Distribution in Brain Tissues. Ann. Neurol 32 S76–S81
M. A. Smith K. Hirai K. Hsiao M. A. Pappolla P. L. Harris S. L. Siedlak M. Tabaton G. Perry (1998) ArticleTitleAmyloid-beta deposition in Alzheimer transgenic mice is associated with oxidative stress. J. Neurochem 70 2212–2215
S. M. LeVine (1991) ArticleTitleOligodendrocytes and myelin sheaths in normal, quaking and shiverer brains are enriched in iron. J. Neurosci. Res 29 413–419
K. Thompson S. Menzies M. Muckenthaler F. M. Torti T. Wood S. V. Torti M. W. Hentze J. Beard J. Connor (2003) ArticleTitleMouse brains deficient in H-ferritin have normal iron concentration but a protein profile of iron deficiency and increased evidence of oxidative stress. J. Neurosci. Res 71 46–63
J. M. Hill R. C. Switzer Suffix3rd (1984) ArticleTitleThe regional distribution and cellular localization of iron in the rat brain. Neuroscience 11 595–603
A. R. White R. Reyes J. F. Mercer J. Camakaris H. Zheng A. I. Bush G. Multhaup K. Beyreuther C. L. Masters R. Cappai (1999) ArticleTitleCopper levels are increased in the cerebral cortex and liver of APP and APLP2 knockout mice. Brain Res 842 439–44
C. J. Maynard R. Cappai I. Volitakis R. A. Cherny A. R. White K. Beyreuther C. L. Masters A. I. Bush Q. X. Li (2002) ArticleTitleOverexpression of Alzheimer’s disease amyloid-beta opposes the age-dependent elevations of brain copper and iron. J. Biol. Chem 277 44670–44676
J. A. Helpern R. J. Ordidge J. M. Gorell J. C. Deniau K. M. A. Welch (1995) ArticleTitlePreliminary Observations of Transverse Relaxation Rates Obtained at 3 Tesla From the Substantia Nigra of Adult Normal Human Brain. NMR Biomed 8 25–27
J. M. Gorell R. J. Ordidge G. G Brown J. C. Deniau N. M. Buderer J. A. Helpern (1995) ArticleTitleIncreased Iron Related MRI Contrast in the Substantia Nigra in Parkinson’s Disease. Neurology 45 1138–1143
G. Bartzokis D. Sultzer J. Cummings L. E. Holt D. B. Hance V. W. Henderson J. Mintz (2000) ArticleTitleIn vivo evaluation of brain iron in Alzheimer disease using magnetic resonance imaging. Arch. Gen. Psychiatry 57 47–53
J. A. Helpern S. P. Lee M. F. Falangola V. V. Dyakin A. Bogart B. Ardekani K. Duff C. A. Branch T. Wisniewski M. J. Leon Particlede O. T. Wolf J. O’Shea R. A. Nixon (2004) ArticleTitleIn Vivo detection of neuropathology in an animal model of Alzheimer’s disease by magnetic resonance imaging. Magn. Reson. Med 51 794–798
S. P. Lee M. F. Falangola R. A. Nixon K. Duff J. A. Helpern (2004) ArticleTitleVisualization of ß-Amyloid Plaques in a Transgenic Mouse Model of Alzheimer’s Disease using MR Microscopy Without Contrast Reagents. Magn. Reson. Med 52 538–544
E. McGowan S. Sanders T. Iwatsubo A. Takeuchi T. Saido C. Zehr X. Yu S. Uljon R. Wang D. Mann D. Dickson K. Duff (1999) ArticleTitleAmyloid phenotype characterization of transgenic mice overexpressing both mutant amyloid precursor protein and mutant presenilin 1 transgenes. Neurobiol. Dis 6 231–244
Jensen, J. H., Chandra, R. 2002. Method for measuring the magnetic field correlation function for water protons in biological tissues. Pages 2297, in Proceedings of the International Society for Magnetic Resonance in Medicine tenth scientific meeting
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Falangola, M.F., Lee, SP., Nixon, R.A. et al. Histological Co-Localization of Iron in Aß Plaques of PS/APP Transgenic Mice. Neurochem Res 30, 201–205 (2005). https://doi.org/10.1007/s11064-004-2442-x
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11064-004-2442-x