Abstract
Introduction
Supratotal resection (SpTR) of glioblastoma may be associated with improved survival, but published results have varied in part from lack of consensus on the definition and appropriate use of SpTR. A previous small survey of neurosurgical oncologists with expertise performing SpTR found resection 1–2 cm beyond contrast enhancement was an acceptable definition and glioblastoma involving the right frontal and bilateral anterior temporal lobes were considered most amenable to SpTR. The general neurosurgical oncology community has not yet confirmed the practicality of this definition.
Methods
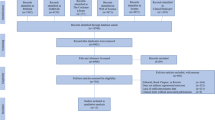
Seventy-six neurosurgical oncology members of the AANS/CNS Tumor Section were surveyed, representing 34.0% of the 223 members who were administered the survey. Participants were presented with 11 definitions of SpTR and rated each definition’s appropriateness. Participants additionally reviewed magnetic resonance imaging for 10 anatomically distinct glioblastomas and assessed the tumor location’s eloquence, perceived equipoise of enrolling patients in a randomized trial comparing gross total to SpTR, and their personal treatment plans.
Results
Most neurosurgeons surveyed agree that gross total plus resection of some non-contrast enhancement (n = 57, 80.3%) or resection 1–2 cm beyond contrast enhancement (n = 52, 73.2%) are appropriate definitions for SpTR. Cases were divided into three anatomically distinct groups by perceived equipoise between gross total and SpTR. The best clinical trial candidates were thought to be right anterior temporal (n = 58, 76.3%) and right frontal (n = 55, 73.3%) glioblastomas.
Conclusion
Support exists among neurosurgical oncologists with varying familiarity performing SpTR to adopt the proposed consensus definition of SpTR of glioblastoma and to potentially investigate the utility of SpTR to treat right anterior temporal and right frontal glioblastomas in a clinical trial. A smaller proportion of general neurosurgical oncologists than SpTR experts would personally treat a left anterior temporal glioblastoma with SpTR.


Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding authors on reasonable request.
References
Omuro A, DeAngelis L (2013) Glioblastoma and other malignant gliomas: a clinical review. JAMA 310(17):1842–1850. https://doi.org/10.1001/jama.2013.280319
Ostrom QT, Gittleman H, Liao P et al (2014) CBTRUS statistical report: Primary brain and central nervous system tumors diagnosed in the United States in 2007–2011. Neuro Oncol 16:iv1–iv63. https://doi.org/10.1093/neuonc/nou223
Louis DN, Perry A, Reifenberger G et al (2016) The 2016 World Health Organization classification of tumors of the central nervous system: a summary. Acta Neuropathol 131(6):803–820. https://doi.org/10.1007/s00401-016-1545-1
Stupp R, Mason W, van den Bent M et al (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352(10):987–996. https://doi.org/10.4137/cmo.s390
Price SJ, Gillard JH (2011) Imaging biomarkers of brain tumour margin and tumour invasion. Br J Radiol 84(SPEC. ISSUE 2):159–167. https://doi.org/10.1259/bjr/26838774
Simpson JR, Horton J, Scott C et al (1993) Influence of location and extent of surgical resection on survival of patients with glioblastoma multiforme: results of three consecutive radiation therapy oncology group (RTOG) clinical trials. Int J Radiat Oncol Biol Phys 26(2):239–244. https://doi.org/10.1016/0360-3016(93)90203-8
Jeremic B, Grujicic D, Antunovic V, Djuric L, Stojanovic M, Shibamoto Y (1994) Influence of extent of surgery and tumor location on treatment outcome of patients with glioblastoma multiforme treated with combined modality approach. J Neurooncol 21(2):177–185. https://doi.org/10.1007/BF01052902
Ushio Y, Kochi M, Hamada JI, Kai Y, Nakamura H (2005) Effect of surgical removal on survival and quality of life in patients with supratentorial glioblastoma. Neurol Med Chir (Tokyo) 45(9):454–460. https://doi.org/10.2176/nmc.45.454
Stummer W, Reulen H, Meinel T (2008) Extent of resection and survival in glioblastoma multiforme: indentification of and adjustment for bias. Neurosurgery 62(3):564–576. https://doi.org/10.1227/01.NEU.0000297118.47076.5E
Davis ME (2016) Glioblastoma: overview of disease and treatment. Clin J Oncol Nurs 20(5):S2–S8
De Leeuw CN, Vogelbaum MA (2019) Supratotal resection in glioma: a systematic review. Neuro Oncol 21(2):179–188. https://doi.org/10.1093/neuonc/noy166
Duffau H (2012) Awake surgery for incidental WHO grade II gliomas involving eloquent areas. Acta Neurochir (Wien) 154(4):575–584. https://doi.org/10.1007/s00701-011-1216-x
Incekara F, Koene S, Vincent AJPE, van den Bent MJ, Smits M (2019) Association between supratotal glioblastoma resection and patient survival: a systematic review and meta-analysis. World Neurosurg 127:617-624.e2. https://doi.org/10.1016/j.wneu.2019.04.092
Jackson C, Choi J, Khalafallah AM et al (2020) A systematic review and meta-analysis of supratotal versus gross total resection for glioblastoma. J Neurooncol 148(3):419–431. https://doi.org/10.1007/s11060-020-03556-y
Khalafallah AM, Huq S, Jimenez AE, Serra R, Bettegowda C, Mukherjee D (2021) “zooming in” on glioblastoma: understanding tumor heterogeneity and its clinical implications in the era of single-cell ribonucleic acid sequencing. Neurosurgery 88(3):477–486. https://doi.org/10.1093/neuros/nyaa305
Binabaj MM, Bahrami A, ShahidSales S et al (2018) The prognostic value of MGMT promoter methylation in glioblastoma: a meta-analysis of clinical trials. J Cell Physiol 233(1):378–386. https://doi.org/10.1002/jcp.25896
Zou P, Xu H, Chen P et al (2013) IDH1/IDH2 mutations define the prognosis and molecular profiles of patients with gliomas: a meta-analysis. PLoS ONE 8(7):1–7. https://doi.org/10.1371/journal.pone.0068782
Kitabayashi T, Nakada M, Furuta T et al (2014) NC-09: the impact of supratotal resection for glioblastoma. Neuro Oncol 16:v134–v137
Pessina F, Navarria P, Cozzi L et al (2017) Maximize surgical resection beyond contrast-enhancing boundaries in newly diagnosed glioblastoma multiforme: is it useful and safe? A single institution retrospective experience. J Neurooncol 135(1):129–139. https://doi.org/10.1007/s11060-017-2559-9
Aldave G, Tejada S, Pay E et al (2013) Prognostic value of residual fluorescent tissue in glioblastoma patients after gross total resection in 5-aminolevulinic acid-guided surgery. Neurosurgery 72(6):915–920. https://doi.org/10.1227/NEU.0b013e31828c3974
Spetzler RF, Martin NA (2008) A proposed grading system for arteriovenous malformations. J Neurosurg 108(1):186–193. https://doi.org/10.3171/JNS/2008/108/01/0186
Friedlein K, Bozhkov Y, Hore N et al (2015) A new functional classification system (FGA/B) with prognostic value for glioma patients. Sci Rep 5:1–11. https://doi.org/10.1038/srep12373
Kahn E, Lane M, Sagher O (2017) Eloquent: history of a word’s adoption into the neurosurgical lexicon. J Neurosurg 127(6):1461–1466. https://doi.org/10.3171/2017.3.JNS17659
Fried I (1993) The myth of eloquent cortex, or what is non-eloquent cortex? J Neurosurg 78:1009–1010. https://doi.org/10.3171/2014.12.JNS142826
Molinaro AM, Hervey-Jumper S, Morshed RA et al (2020) Association of maximal extent of resection of contrast-enhanced and non-contrast-enhanced tumor with survival within molecular subgroups of patients with newly diagnosed glioblastoma. JAMA Oncol 6(4):495–503. https://doi.org/10.1001/jamaoncol.2019.6143
Li YM, Suki D, Hess K, Sawaya R (2016) The influence of maximum safe resection of glioblastoma on survival in 1229 patients: can we do better than gross-total resection? J Neurosurg 124(4):977–988. https://doi.org/10.3171/2015.5.JNS142087
Mampre D, Ehresman J, Pinilla-Monsalve G et al (2018) Extending the resection beyond the contrast-enhancement for glioblastoma: feasibility, efficacy, and outcomes. Br J Neurosurg 32(5):528–535. https://doi.org/10.1080/02688697.2018.1498450
De Bonis P, Anile C, Pompucci A et al (2013) The influence of surgery on recurrence pattern of glioblastoma. Clin Neurol Neurosurg 115(1):37–43. https://doi.org/10.1016/j.clineuro.2012.04.005
Glenn CA, Baker CM, Conner AK et al (2018) An examination of the role of supramaximal resection of temporal lobe glioblastoma multiforme. World Neurosurg 114:e747–e755. https://doi.org/10.1016/j.wneu.2018.03.072
Esquenazi Y, Friedman E, Liu Z, Zhu JJ, Hsu S, Tandon N (2017) The survival advantage of “supratotal” resection of glioblastoma using selective cortical mapping and the subpial technique. Neurosurgery 81(2):275–288. https://doi.org/10.1093/neuros/nyw174
Hamada S, Abou-Zeid A (2016) Anatomical resection in glioblastoma: extent of resection and its impact on duration of survival. Egypt J Neurol Psychiatry Neurosurg 53(3):135–145. https://doi.org/10.4103/1110-1083.192655
Lu M, Fu ZH, He XJ et al (2020) T2 fluid-attenuated inversion recovery resection for glioblastoma involving eloquent brain areas facilitated through awake craniotomy and clinical outcome. World Neurosurg 135:e738–e747. https://doi.org/10.1016/j.wneu.2019.12.130
Ghogawala Z, Schwartz JS, Benzel EC et al (2016) Increased patient enrollment to a randomized surgical trial through equipoise polling of an expert surgeon panel. Ann Surg 264(1):81–86. https://doi.org/10.1097/SLA.0000000000001483
Khalafallah AM, Rakovec M, Bettegowda C et al (2021) A crowdsourced consensus on supratotal resection versus gross total resection for anatomically distinct primary glioblastoma. Neurosurgery 89(4):712–719. https://doi.org/10.1093/neuros/nyab257
Sonabend AM, Zacharia BE, Cloney MB et al (2017) Defining glioblastoma resectability through the wisdom of the crowd: a proof-of-principle study. Neurosurgery 80(4):590–601. https://doi.org/10.1227/NEU.0000000000001374
Choucair A, Levin V, Gutin P et al (1986) Development of multiple lesions during radiation and chemotherapy in patients with gliomas. J Neurosurg 65:654–658
Hou LC, Veeravagu A, Hsu AR, Tse VCK (2006) Recurrent glioblastoma multiforme: a review of natural history and management options. Neurosurg Focus 20(4):E3. https://doi.org/10.3171/foc.2006.20.4.2
Gaspar LE, Fisher BJ, Macdonald DR et al (1992) Supratentorial malignant glioma: Patterns of recurrence and implications for external beam local treatment. Int J Radiat Oncol Biol Phys 24(1):55–57. https://doi.org/10.1016/0360-3016(92)91021-E
Burger PC, Dubois PJ, Schold SC et al (1983) Computerized tomographic and pathologic studies of the untreated, quiescent, and recurrent glioblastoma multiforme. J Neurosurg 58(2):159–169. https://doi.org/10.3171/jns.1983.58.2.0159
Petrecca K, Guiot MC, Panet-Raymond V, Souhami L (2013) Failure pattern following complete resection plus radiotherapy and temozolomide is at the resection margin in patients with glioblastoma. J Neurooncol 111(1):19–23. https://doi.org/10.1007/s11060-012-0983-4
Darmanis S, Sloan SA, Croote D et al (2017) Single-cell RNA-Seq analysis of infiltrating neoplastic cells at the migrating front of human glioblastoma. Cell Rep 21(5):1399–1410. https://doi.org/10.1016/j.celrep.2017.10.030
Schneider M, Potthoff AL, Keil VC et al (2019) Surgery for temporal glioblastoma: lobectomy outranks oncosurgical-based gross-total resection. J Neurooncol 145(1):143–150. https://doi.org/10.1007/s11060-019-03281-1
Shah AH, Mahavadi A, Di L et al (2020) Survival benefit of lobectomy for glioblastoma: moving towards radical supramaximal resection. J Neurooncol 148(3):501–508. https://doi.org/10.1007/s11060-020-03541-5
Roh TH, Kang SG, Moon JH et al (2020) Survival benefit of lobectomy over gross-total resection without lobectomy in cases of glioblastoma in the noneloquent area: a retrospective study. J Neurosurg 132(3):895–901. https://doi.org/10.3171/2018.12.JNS182558
McGirt MJ, Mukherjee D, Chaichana KL, Than KD, Weingart JD, Quinones-Hinojosa A (2009) Association of surgically acquired motor and language deficits on overall survival after resection of glioblastoma multiforme. Neurosurgery 65(3):463–469. https://doi.org/10.1227/01.NEU.0000349763.42238.E9
Rahman M, Abbatematteo J, De Leo EK et al (2017) The effects of new or worsened postoperative neurological deficits on survival of patients with glioblastoma. J Neurosurg 127(1):123–131. https://doi.org/10.3171/2016.7.JNS16396
Bu LH, Zhang J, Lu JF, Wu JS (2021) Glioma surgery with awake language mapping versus generalized anesthesia: a systematic review. Neurosurg Rev 44(4):1997–2011. https://doi.org/10.1007/s10143-020-01418-9
Sanai N, Berger M (2012) Recent surgical management of gliomas. In: Yamanaka R (ed) Glioma. Advances in experimental medicine and biology. Springer, New York, pp 12–25
Hervey-Jumper SL, Berger MS (2019) Insular glioma surgery: an evolution of thought and practice. J Neurosurg 130(1):9–16. https://doi.org/10.3171/2018.10.JNS181519
Sanai N, Polley MY, Berger MS (2010) Insular glioma resection: assessment of patient morbidity, survival, and tumor progression—clinical article. J Neurosurg 112(1):1–9. https://doi.org/10.3171/2009.6.JNS0952
Alimohamadi M, Shirani M, Shariat Moharari R et al (2016) Application of awake craniotomy and intraoperative brain mapping for surgical resection of insular gliomas of the dominant hemisphere. World Neurosurg 92:151–158. https://doi.org/10.1016/j.wneu.2016.04.079
Acknowledgements
The authors would like to thank Erinma Elibe, MPH and Kristin Zerfas for their contributions toward creating and distributing the online survey, respectively. We would also like to acknowledge members of the Johns Hopkins Neuro-Oncology Surgical Outcomes Lab for their valuable input that has helped inform this project.
Funding
The authors acknowledge assistance for clinical data coordination and retrieval from the Core for Clinical Research Data Acquisition, supported in part by the Johns Hopkins Institute for Clinical and Translational Research (UL1TR001079). The authors received no remuneration for the authorship and/or publication of this article.
Author information
Authors and Affiliations
Contributions
MR: Methodology, Formal Analysis, Writing—Original Draft, Review and Editing. AMK: Conceptualization, Methodology. OW: Formal Analysis, Writing—Original Draft, Review and Editing. DD: Formal Analysis, Writing—Original Draft, Review and Editing. JPS: Data Acquisition, Writing—Review and Editing. JHS: Data Acquisition, Writing—Review and Editing. DM: Conceptualization, Supervision, Writing—Review and Editing.
Reporting guidelines
We found no applicable reporting guidelines that would apply to this article. By following the EQUATOR reporting guidelines decision tree, (http://www.equatornetwork.org/wp-content/uploads/2013/11/20160226-RG-decision-tree-for-Wizard-CC-BY-26-February-2016.pdf), we found that none of the most popular checklists are appropriate for our study design.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
This study was reviewed and approved by the Johns Hopkins institutional review board (IRB00196609).
Consent to participate
This study was exempt from obtaining patient consent from patients whose radiographic data was used to generate the survey, as all data was de-identified as required by Health Insurance Portability and Accountability Act (HIPAA) regulations and institutional review board protocol (IRB00196609).
Informed consent
Informed consent was obtained from all participants who completed the survey.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rakovec, M., Khalafallah, A.M., Wei, O. et al. A consensus definition of supratotal resection for anatomically distinct primary glioblastoma: an AANS/CNS Section on Tumors survey of neurosurgical oncologists. J Neurooncol 159, 233–242 (2022). https://doi.org/10.1007/s11060-022-04048-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-022-04048-x