Abstract
Purpose
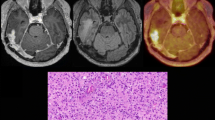
In-field high-grade glioma (HGG) recurrence is a common challenge with limited treatment options, including re-irradiation. The radiotracer 3,4-dihydroxy-6-[18F]-fluoro-l-phenylalanine (18F-DOPA) crosses the blood brain barrier and demonstrates high uptake in tumor, but low uptake in normal tissue. This study investigated whether 18F-DOPA positron emission tomography (PET) and MRI guided re-irradiation for recurrent HGG may improve progression free survival (PFS).
Methods
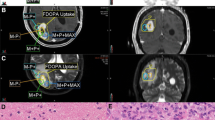
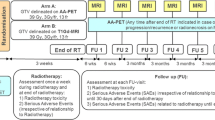
Adults with recurrent or progressive HGG previously treated with radiation were eligible. The primary endpoint was a 20% improvement from the historical control PFS at 3 months (PFS3) of 20% with systemic therapy alone. Re-RT dose was 35 Gy in 10 fractions. The target volume was MRI T1 contrast-enhancement defined tumor plus 18F-DOPA PET defined tumor.
Results
Twenty patients completed treatment per protocol. Diagnosis was most commonly glioblastoma, IDH-wildtype (60%). MRI-defined volumes were expanded by a median 43% (0–436%) by utilizing 18F-DOPA PET. PFS3 was 85% (95% CI 63.2–95.8%), meeting the primary endpoint of PFS3 ≥ 40%. With 9.7 months median follow-up, 17 (85%) had progressed and 15 (75%) had died. Median OS from re-RT was 8.8 months. Failure following re-RT was within both the MRI and PET tumor volumes in 75%, MRI only in 13%, PET only in 0%, and neither in 13%. Four (20%) patients experienced grade 3 toxicity, including CNS necrosis (n = 2, both asymptomatic with bevacizumab initiation for radiographic findings), seizures (n = 1), fatigue (n = 1), and nausea (n = 1). No grade 4–5 toxicities were observed.
Conclusion
18F-DOPA PET-guided re-irradiation for progressive high-grade glioma appears safe and promising for further investigation.




Similar content being viewed by others
Data availability
Data used for this analysis is available upon request from the corresponding author.
References
Chan JL et al (2002) Survival and failure patterns of high-grade gliomas after three-dimensional conformal radiotherapy. J Clin Oncol 20(6):1635–1642
Gebhardt BJ et al (2014) Patterns of failure for glioblastoma multiforme following limited-margin radiation and concurrent temozolomide. Radiat Oncol 9:130
Lundemann M et al (2017) Patterns of failure for patients with glioblastoma following O-(2-[(18) F] fluoroethyl)-L-tyrosine PET- and MRI-guided radiotherapy. Radiother Oncol 122(3):380–386
McDonald MW et al (2011) Pattern of failure after limited margin radiotherapy and temozolomide for glioblastoma. Int J Radiat Oncol Biol Phys 79(1):130–136
NCCN Guidelines Version 1.2021 Central Nervous System Cancers. Available from: https://www.nccn.org/professionals/physician_gls/pdf/cns.pdf. Accessed on 06 sep 2021
Mayer R, Sminia P (2008) Reirradiation tolerance of the human brain. Int J Radiat Oncol Biol Phys 70(5):1350–1360
Shi W et al (2018) Investigating the effect of reirradiation or systemic therapy in patients with glioblastoma after tumor progression: a secondary analysis of nrg oncology/radiation therapy oncology group trial 0525. Int J Radiat Oncol Biol Phys 100(1):38–44
Straube C et al (2019) A second course of radiotherapy in patients with recurrent malignant gliomas: clinical data on re-irradiation, prognostic factors, and usefulness of digital biomarkers. Curr Treat Options Oncol 20(9):71
Tsien C et al (2019) randomized phase ii trial of re-irradiation and concurrent bevacizumab versus bevacizumab alone as treatment for recurrent glioblastoma (nrg oncology/rtog 1205): initial outcomes and rt plan quality report. Int J Radiat Oncol Biol Phys 105(1):S78–S78
Youland RS et al (2018) Modern reirradiation for recurrent gliomas can safely delay tumor progression. Neurooncol Pract 5(1):46–55
de Wit MC et al (2004) Immediate post-radiotherapy changes in malignant glioma can mimic tumor progression. Neurology 63(3):535–537
Taal W et al (2008) Incidence of early pseudo-progression in a cohort of malignant glioma patients treated with chemoirradiation with temozolomide. Cancer 113(2):405–410
Hygino da Cruz LC Jr et al (2011) Pseudoprogression and pseudoresponse: imaging challenges in the assessment of posttreatment glioma. AJNR Am J Neuroradiol 32(11):1978–1985
Chen W et al (2006) 18F-FDOPA PET imaging of brain tumors: comparison study with 18F-FDG PET and evaluation of diagnostic accuracy. J Nucl Med 47(6):904–911
Ledezma CJ et al (2009) 18F-FDOPA PET/MRI fusion in patients with primary/recurrent gliomas: initial experience. Eur J Radiol 71(2):242–248
Pafundi DH et al (2013) Biopsy validation of 18F-DOPA PET and biodistribution in gliomas for neurosurgical planning and radiotherapy target delineation: results of a prospective pilot study. Neuro Oncol 15(8):1058–1067
Laack NN et al (2021) Initial results of a phase 2 trial of (18)F-DOPA PET-guided dose-escalated radiation therapy for glioblastoma. Int J Radiat Oncol Biol Phys 110(5):1383–1395
Armstrong TS et al (2006) Validation of the M.D. Anderson symptom inventory brain tumor module (MDASI-BT). J Neurooncol 80(1):27–35
Wen PY et al (2010) Updated response assessment criteria for high-grade gliomas: response assessment in neuro-oncology working group. J Clin Oncol 28(11):1963–1972
Iwamoto FM et al (2009) Patterns of relapse and prognosis after bevacizumab failure in recurrent glioblastoma. Neurology 73(15):1200–1206
Kreisl TN et al (2009) Phase II trial of single-agent bevacizumab followed by bevacizumab plus irinotecan at tumor progression in recurrent glioblastoma. J Clin Oncol 27(5):740–745
Kim MM et al (2021) Response assessment during chemoradiation using a hypercellular/hyperperfused imaging phenotype predicts survival in patients with newly diagnosed glioblastoma. Neuro Oncol 23(9):1537–1546
Kim MM et al (2021) a phase 2 study of dose-intensified chemoradiation using biologically based target volume definition in patients with newly diagnosed glioblastoma. Int J Radiat Oncol Biol Phys 110(3):792–803
Funding
No funding sources to report.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by WGB, SG, SBJ, and DB. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Dr. Ruff receives royalties and planned/pending patents unrelated to this work. Dr. Kizilbash receives grant funding to the institution and investigational product for preclinical studies, unrelated to this work.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Research involved in human and animal rights
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This study was presented as an oral presentation at the ASTRO 2021 Annual Meeting.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Breen, W.G., Youland, R.S., Giri, S. et al. Initial results of a phase II trial of 18F-DOPA PET-guided re-irradiation for recurrent high-grade glioma. J Neurooncol 158, 323–330 (2022). https://doi.org/10.1007/s11060-022-04011-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-022-04011-w