Abstract
Purpose
Patients suffering from recurrent and residual grade 2 (WHO) meningiomas after subtotal excision should be considered as high-risk groups with an uncertain prognosis. Adjuvant radiotherapy seems to be the best approach to reduce disease progression. The primary aim of this phase II explorative, monocentric, single arm study was to evaluate the safety of adjuvant multisession radiosurgery (mRS) in this group of patients; the efficacy in terms of tumour local control was the secondary endpoint.
Methods
Patients recruited from April 2017 to May 2019 were over 18 years old, had a histologically-documented intracranial recurrent or residual Grade 2 meningioma (WHO 2016) and a KPS > 70. Patients with NF2, concomitant neoplasm or pregnancy were excluded. Descriptive statistics were provided for categorical variables. Progression free survival (PFS) was modelled using the Kaplan–Meier method.
Results
Twenty-four patients were enrolled. All 24 patients underwent mRS: twenty-two patients received 28 Gy in 4 fractions, 2 patients received 24 Gy in 4 Treatment related adverse events (CTCAE 4.3) were limited to grade 2 in 1 patient (4.1%). At a median follow-up of 28 months, 8 patients (33.3%) had disease progression, either out-of-field or infield, compared with the planning target volume. Considering both infield and out-of-field progressions, 3-year PFS was 47% (95% confidence interval, CI, 22–69%); considering only the infield ones, 3-year PFS was 86% (95% CI 55–96%), and local control at last follow-up was 92%.
Conclusion
mRS provides good local control of the tumour volume (TV) and is associated with a low rate of toxicity. These results call for further investigation to confirm favourable outcomes in patients with high-risk meningioma.
Trial information
NCT05081908, October 18, 2021, retrospectively registered.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Meningiomas account for approximately 15–20% of all primary brain tumours in adults [1], among them, grade 2 meningiomas account for approximately 5–7% of the total [1, 2]. These meningiomas are associated with aggressive growth patterns, in line with their clinical and histopathological features of malignancy. Therefore, over the years, the World Health Organization (WHO) Classification System for Meningioma has been optimized to correlate with clinical behaviour [3]. The latest WHO classification of meningiomas has transformed many previously considered grade 1 tumours into higher grades, based solely upon histopathologic criteria [3,4,5]. Although grade 2 meningiomas can be considered rare, they are associated with severely debilitating and life-threatening patterns and symptoms, hence optimal diagnosis and management of these fast-growing lesions is necessary [4, 5]. Complete resection is the goal of the treatment of atypical meningiomas [6], however it is very difficult to achieve especially in the case of skull-based locations. Some groups favour a wait-and-scan approach after surgery, while others believe that long-term local control can be achieved with adjuvant radiotherapy (RT). However, atypical meningiomas can be completely resected and then definitively cured in only 16–18% of cases [7]. For incompletely resected grade 2 meningiomas the overall prognosis is poor with 5- and 10-year PFS of 38–59% and 19–22%, respectively [8, 9], and a significantly reduced overall survival (OS) [8,9,10]. Recurrent and residual grade 2 meningiomas after subtotal excision should be considered as high-risk groups with a bad prognosis [11]. In these cases, RT appears to be the best approach to reduce disease progression, even though the results in terms of disease-free survival are quite poor. So far, most of the published studies have assessed the efficacy of conventional RT, few studies have reported the results of SRS in a single fraction [6,7,8,9,10, 12,13,14,15,16,17,18,19,20,21,22,23,24]. A recent multi-centre international cohort study has reported a 33.5% actuarial PFS at 5 years, which is quite low when compared with SRS results in benign meningiomas [23]. Only a limited number of recent publications describe the outcomes of the use of multisession radiosurgery [24,25,26]: RS in multiple sessions combines the possibility to increase local control by delivering higher total doses over a short time with the reduction of treatment-associated side effects by limiting the irradiated brain volume. Bearing this in mind, at our Institute, a prospective phase II study was designed to evaluate the tolerance of a multisession radiosurgery (mRS) schedule in this subgroup of patients suffering from high risk grade 2 meningiomas (residual and recurrent).
Methods and design
This is a phase II explorative, monocentric, single arm study. Modelling has shown that an increased daily dose and decreased total treatment time results in higher tumour kill without the development of a predominant radiation-resistant clone. The model indicates that hypofractionation induces less radioresistance for the same calculated BED [27, 28]. Consequently, we have hypothesized that a schedule of mRS delivering 24–32 Gy in 4 fractions could achieve good local control. Moreover, considering the sharp dose fall-off, a low rate of radiation related neurological toxicities has been assumed.
Our Institute’s eligible patients were recruited in the study from April 2017 to May 2019 according to the following selection criteria: adults 18 years of age or older with histologically-documented intracranial Grade 2 meningioma (WHO 2016) after subtotal resection or recurrence (High Risk meningioma) and KPS > 70 were enrolled. Patients with NF2, or concomitant aggressive haematological or solid neoplasm or pregnancy were excluded.
Ethics
The trial was approved by the institutional review board and ethical committee. Each patient signed an approved informed consent before trial enrollment. The protocol was registered in ClinicalTrials.gov (ID: NCT03345940).
Treatment characteristics
Patients were scheduled to undergo CyberKnife multisession radiosurgery (mRS): a dose fraction of 6–8 Gy with a total dose of 24–32 Gy was planned to be delivered in 4 fractions (consecutive days) to an isodose line encompassing 95% of the planning target volume (PTV) to the 80% ± 5% prescription isodose line. The mRS treatments were performed with CyberKnife, as previously described [29]. The target was delineated on high-resolution (1 mm thickness) contrast-enhanced computed tomography (CT) and co-registered high-resolution MR (1 mm thickness), always using T2 and T1-weighted contrast-enhanced sequences, as well as fat-saturated sequences whenever necessary.
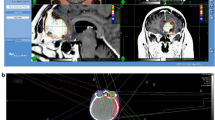
The tumour volume (TV) was identified based on these images as contrast-enhancing areas. No margin was added for the PTV. All anatomical structures were contoured as organs at risk (OARs): brain, brainstem, motor area retinas, eyes, lenses, optic nerves, optic pathways, cochleas, acoustic nerves, and skin. The treatment plans involved an inverse planning method making use of non-isocentric technique (Multiplan Accuray Inc.). A typical treatment plan is depicted in Fig. 1.
Study aim and endpoints
The primary endpoint was tolerance to mRS: the frequency of neurological side effects related to the radiosurgical treatment was evaluated according to Common CTCAE scale ver. 4.0.
The secondary end-points were PFS and local control.
The rate of tumour response was defined on the basis of variation of MR imaging, and evaluated also through advanced MR techniques and volumetric analysis of the lesions as detailed below:
- Complete response (CR):
-
Disappearance of the lesion on MR imaging.
- Partial response (PR):
-
20% decrease in the volumetric size of the lesion on MRI.
- Stable disease (SD):
-
No change in the size of the lesion.
- Progressive disease (PD):
-
Appearance of new lesion/s or tumour volume increase > 20% compared to baseline and confirmed at two consecutive MRs.
The pattern of failure has been defined as follow:
-
In field if the treated tumour grows
-
Marginal if tumour grows beside the original volume (specifically between the PTV boundary and the 50% isodose line
-
Distant in case of a new lesion appearance, distant from the treated PTV.
Pre-treatment evaluation and follow up
Patients were prospectively followed-up after treatment for at least 3 years. Pre and post-treatment evaluations consisted of physical and neurological assessment. Each adverse event (AE) was scored in accordance with the NCI Common Toxicity criteria, version 4.03. Patients were scheduled for follow-up visits and radiological evaluations 4 months after the treatment, every 6 months during the first, second and the third year.
In order to evaluate the tumour control, a T1-weighted volumetric post contrast MRI sequence, with the addition of a T1-weighted volumetric post contrast fat-saturation sequence in case of anterior skull base involvement, were mandatory.
To assess the tumour response, a volumetric analysis was performed by means of co-registration of each follow-up MR on baseline MR. Contouring of the lesion on each post-treatment MR was carried out to ensure that the PTV and the post-irradiation lesions were spatially aligned and comparable.
Data collection
Data collection, based on General Data Protection Regulation (GDPR) guidelines, ensured privacy during the clinical trial processes, and each patient was provided with an identification number. Clinical, neurological and radiological data were recorded on a case report form (CRF) and on an electronic database according to the Good Clinical Practice guidelines (GCP), both at baseline and during the follow-up period at each scheduled session.
Statistical analysis
Descriptive statistics were provided in terms of absolute numbers and percentages for categorical data, means with standard deviations (SDs) and medians with value ranges for continuous data. PFS was analysed using the Kaplan–Meier method in order to obtain survival curve and probabilities at different time points.
Results
The study was activated on the 5th April 2017, and accrual was completed on 7th May 2019. The analysis date for this report was May 2021.
Twenty-four out of 27 screened patients were included, 3 were not eligible. All patients completed the treatment protocol, including twelve males and twelve females. The average age at treatment time was 61 years (range 37–85). All patients but one was alive at the date of the analysis (one death due to Covid-19 related pulmonary complication).
The most common tumour location was convexity (40%), followed by parasagittal (36%), and skull base (24%). Eight patients had residual disease and 16 had recurrence. As regards histology, 24 patients were diagnosed with atypical meningioma, 1 with focal rhabdoid aspect, 3 with chordoid features. In 19 cases the number of mitosis in 10 high power fields (HPF) was > 4, in 5 it was ≤ 4. The mean MIB1 value in the whole cohort was 8%, ranging from 2 to 15%.
The median pre-SRS tumour volume was 2.7 cc (range 0.6–16.4 cc).
The prescription dose was respectively 28 Gy in 4 fractions for 22 patients and 24 Gy in 4 fractions for 2 patients, with a median prescription isodose line of 81% (range 78–84%). The median conformity index was 1.30 (range 1.17–2.95). Patients, tumour and dosimetric features are displayed in Table 1.
One patient developed grade 2 toxicity (brief generalized seizure), according to CTCAE ver. 4.0 4 months from SRS. No severe adverse events occurred (CTCAE ≥ III).
The median follow-up was 28 months.
In 54% of the patients a volumetric reduction (> 20%) of the treated lesions was observed (see Fig. 2). At the last follow-up 8 patients (33.3%) showed stable disease, 8 (33.3%) partial response, and 8 (33.3%) had developed progressive disease. As regards to the latter group, the recurrence was distant from the original treated site in 6 patients (in 4 cases more than 3 cm from the PTV, and in 2 cases between 1 and 2 cm from the PTV). The last two patients experienced both marginal and infield progression. Considering both infield and outfield progressions, PFS at 3 year was 47% (95% confidence interval, CI, 22–69%), and local control at last follow-up was 63% (Fig. 3). Considering only the infield progressions to evaluate the efficacy of the mRS schedule, 3-year PFS was 86% (95% CI 55–96%), and local control at last follow-up was 92% (Fig. 4). The median time to tumour progression was 17 months (range 3–29). Four (50%) of the 8 progressed patients had mRS for residual tumour, 4 (50%) for recurrence.
In A and C are respectively depicted the axial and coronal post gadolinium, pre-radiosurgery images of frontal parasagittal atypical meningioma. In B and D the 24 months post-radiosurgery images show a partial response of the tumor. The treatment plan are presented in Fig. 1
All recurrent patients but 2 (75%) had a number of mitosis in 10 high power fields > 4.
The mean MIB-1 value in the progression group is 7%; while in the stable disease group is 8%.
A new mRS, with the same RS schedule was performed on 6 patients with outfield progressions.
No adverse events occurred in these cases. Three patients underwent surgery.
Discussion
The issue about the timing and the modality to treat recurrent and residual grade 2 meningioma is still under debate. A recent RTOG trial [11, 30] proposed the use of high dose IMRT treatment (54–60 Gy) by using conventional margins (up to 2 cm) to achieve better local control in this kind of patient group suffering from high-risk meningioma. In this study the authors report a 3-year PFS rate of 58.8% after IMRT treatment with the 92.9% of recurrences, within the planning target volume. One patient (1.9%) died because of treatment related toxicity and near 50% of the patients suffered from grade 1–3 toxicity. Considering this, the authors argue the possibility of dose escalation to the whole volume (instead of two different dose levels) or the possibility of IMRT with radiosurgery boost.
In a large experience from the International Radiosurgery Research Foundation (IRRF) the authors report that using single session radiosurgery [23] a 1, 2, and 5-years PFS rate of 84.2%, 67.8%, and 36.4% for atypical meningiomas (median 41.4 months) which are comparable to the IMRT experiences; the adverse events were observed in 12.5% of cases (50 symptomatic).
In this scenario, we started our experience by treating patients with high dose mRS (24–28 Gy in 4 fractions) without margins to achieve both a better local control and a safer re-treatment possibility.
As a fact, the present phase II explorative study suggests that mRS could elicit positive results in the treatment of residual/recurrent atypical meningioma without any major adverse events, including the 6 patients who underwent a retreatment with the same doses because of PD.
The above-mentioned schedule was indeed well tolerated; after a median follow-up period of 28 months no grade III–IV toxicity was observed. This, together with the 3-years PFS of 47% and the 92% control rate of the treated recurrent/residual tumour would induce us to confirm the safety and the efficacy of the SRT schedule. Nevertheless, considering the type of recurrence found in our explorative study (75% outside the PTV), in our opinion the main pitfall remains the correct identification of the TV, as stated in a recent publication [24, 30, 31]. Whether or not including the dural tail or a few mm along the dura or healthy brain in proximity of the TV could be taken in consideration to obtain better local tumour control. Biological PET images could help define the optimal target in the TV identification process. PET may indeed enhance the detection of suspicious diffusion, which may elude the current radiological techniques [32]. In adjunction to doses and radiation delivery techniques, new trials should focus on neuroimaging and genetic definition [32, 33]. A possible genetic- and molecular-based classification of meningiomas may introduce a new way of stratifying risk and provide a surrogate prognostic factor of survival and recurrence [33, 34]. The current study does not support specific prognostic factors but some elements such as the number of mitosis per 10 HPF or the MIB1 rate warrant further investigations.
Another favourable element observed has been the low rate of complication after the re-irradiation of new localisations in out-of-field recurred patients. In 6 patients’ new lesions (distant more than 1 cm from the original PTV) were observed: re-irradiation was performed with the same RT schedule without any adverse events. As in oligometastatic patients’ multiple irradiations seems to offer good local control and theoretically better OS. The concept of not offering a definitive cure but a treatment with low morbidity can be in the specific population an affordable option. Quality of life and neuropsychological tests should be introduced to verify this affirmation.
In conclusion, 28 Gy in a 4 fraction schedule achieves good local control in the TV with a low percentage of infield recurrence/progression. The main problem in treating high-risk meningiomas remains the development of out-of-field recurrences, thereby reducing the rate of global local control [24]. Repeat mRS can be a further option to locally control the disease progression as suggested in oligometastatic patients, in which multiple irradiation seems to prolong the OS with good quality of life. A key element in support of this option is the low rate of observed neurological complications (4.1%, grade 2). More data is needed to confirm the results of this study, and an extension of the protocol has been proposed to the Ethics Committee.
Data availability
The datasets generated during the current study are available from the corresponding author on reasonable request.
References
Ostrom QT, Gittleman H, Liao P, Rouse C, Chen Y, Dowling J, Wolinsky Y, Kruchko C, Barnholtz-Sloan J (2014) CBTRUS statistical report: primary brain and central nervous system tumors diagnosed in the United States in 2007–2011. Neuro Oncol 16(44):iv1-63. https://doi.org/10.1093/neuonc/nou223
Claus EB, Bondy ML, Schildkraut JM, Wiemels JL, Wrensch M, Black PM (2005) Epidemiology of intracranial meningioma. Neurosurgery 57(6):1088–1095. https://doi.org/10.1227/01.NEU.0000188281.91351.B9
Louis DN, Perry A, Reifenberger G, von Deimling A, Figarella-Branger D, Cavenee WK et al (2016) The 2016 world health organization classification of tumors of the central nervous system: a summary. Acta Neuropathol 131(6):803–820
Zhi M, Girvigian MR, Miller MJ, Chen JC, Schumacher AJ, Rahimian J et al (2019) Long-term outcomes of newly diagnosed resected atypical meningiomas and the role of adjuvant radiotherapy. World Neurosurg 122:e1153–e1161. https://doi.org/10.1016/j.wneu.2018.11.006
Rogers L, Barani I, Chamberlain M, Kaley TJ, McDermott M, Raizer J et al (2015) Meningiomas: knowledge base, treatment outcomes, and uncertainties. A RANO review. J Neurosurg 122(1):4–23. https://doi.org/10.3171/2014.7.JNS131644
Goldbrunner R, Stavrinou P, Jenkinson MD, Sahm F, Mawrin C, Weber DC, Preusser M, Minniti G, Lund-Johansen M, Lefranc F, Houdart E, Sallabanda K, Le Rhun E, Nieuwenhuizen D, Tabatabai G, Soffietti R, Weller M (2021) EANO guideline on the diagnosis and management of meningiomas. Neuro Oncol. https://doi.org/10.1093/neuonc/noab150ù
Pereira BJA, de Almeida AN, Paiva WS, Teixeira MJ, Marie SKN (2019) Impact of radiotherapy in atypical meningioma recurrence: literature review. Neurosurg Rev 42(3):631–637
Bagshaw HP, Burt LM, Jensen RL, Suneja G, Palmer CA, Couldwell WT et al (2017) Adjuvant radiotherapy for atypical meningiomas. J Neurosurg 126(6):1822–1828
Durand A, Labrousse F, Jouvet A, Bauchet L, Kalamaridès M, Menei P et al (2009) WHO grade II and III meningiomas: a study of prognostic factors. J Neurooncol 95(3):367–375
Delgado-López PD, Corrales-García EM (2021) Role of adjuvant radiotherapy in atypical (WHO grade II) and anaplastic (WHO grade III) meningiomas: a systematic review. Clin Transl Oncol 23:205–221. https://doi.org/10.1007/s12094-020-02434-3
Rogers CL, Won M, Vogelbaum MA, Perry A, Ashby LS, Modi JM, Alleman AM, Galvin J, Fogh SE, Youssef E, Deb N, Kwok Y, Robinson CG, Shu HK, Fisher BJ, Panet-Raymond V, McMillan WG, de Groot JF, Zhang P, Mehta MP (2020) High-risk meningioma: initial outcomes from NRG oncology/RTOG 0539. Int J Radiat Oncol Biol Phys 106(4):790–799. https://doi.org/10.1016/j.ijrobp.2019.11.028
Graffeo CS, Leeper HE, Perry A, Uhm JH, Lachance DJ, Brown PD et al (2017) Revisiting adjuvant radiotherapy after gross total resection of World Health Organization grade II meningioma. World Neurosurg 103:655–663. https://doi.org/10.1016/j.wneu.2017.04.095
Ryu HS, Moon KS, Lee KH et al (2017) Recurred intracranial meningioma: a retrospective analysis for treatment outcome and prognostic factor. Brain Tumor Res Treat 5(2):54–63. https://doi.org/10.14791/btrt.2017.5.2.54
Lee G, Lamba N, Niemierko A, Kim DW, Chapman PH, Loeffler JS, Curry WT, Martuza RL, Oh KS, Barker FG 2nd, Shih HA (2020) Adjuvant radiotherapy versus surveillance following surgical resection of atypical meningiomas. Int J Radiat Oncol Biol Phys 109(1):252–266. https://doi.org/10.1016/j.ijrobp.2020.08.015
Keric N, Kalasauskas D, Freyschlag CF, Gempt J, Misch M, Poplawski A, Lange N, Ayyad A, Thomé C, Vajkoczy P, Meyer B, Ringel F (2020) Impact of postoperative radiotherapy on recurrence of primary intracranial atypical meningiomas. J Neurooncol 146(2):347–355. https://doi.org/10.1007/s11060-019-03382-x
Reddy AK, Ryoo JS, Denyer S, McGuire LS, Mehta AI (2019) Determining the role of adjuvant radiotherapy in the management of meningioma: a Surveillance, Epidemiology, and End Results analysis. Neurosurg Focus 46(6):E3. https://doi.org/10.3171/2019.3.FOCUS1971
Weber DC, Ares C, Villa S, Peerdeman SM, Renard L, Baumert BG et al (2018) Adjuvant postoperative high-dose radiotherapy for atypical and malignant meningioma: a phase-II parallel non-randomized and observation study (EORTC 22042–26042). Radiother Oncol 128(2):260–265
Kaur G, Sayegh ET, Larson A, Bloch O, Madden M, Sun MZ et al (2014) Adjuvant radiotherapy for atypical and malignant meningiomas: a systematic review. Neuro Oncol 16(5):628–636. https://doi.org/10.1093/neuonc/nou025
Valery CA, Faillot M, Lamproglou I, Golmard JL, Jenny C, Peyre M, Mokhtari K, Mazeron JJ, Cornu P, Kalamarides M (2016) Grade II meningiomas and Gamma Knife radiosurgery: analysis of success and failure to improve treatment paradigm. J Neurosurg 125(Suppl 1):89–96. https://doi.org/10.3171/2016.7.GKS161521
Liu X, Shan B, Wang M, Xu J (2018) World Health Organization Grade II Meningiomas: the role of adjuvant/salvage gamma knife surgery after initial surgery and prognostic factor assessment. World Neurosurg 109:e352–e362. https://doi.org/10.1016/j.wneu.2017.09.178
Kim M, Cho YH, Kim JH, Kim CJ, Roh SW, Kwon DH (2020) Role of gamma knife radiosurgery for recurrent or residual World Health Organization grade II and III intracranial meningiomas. Br J Neurosurg 34(3):239–245. https://doi.org/10.1080/02688697.2020.1726285
Cohen-Inbar O, Lee CC, Sheehan JP (2016) The contemporary role of stereotactic radiosurgery in the treatment of meningiomas. Neurosurg Clin N Am 27(2):215–228
Shepard MJ, Xu Z, Kearns K, Li C, Chatrath A, Sheehan K, Sheehan D, Faramand A, Niranjan A, Kano H, Gurewitz J, Bernstein K, Liscak R, Guseynova K, Grills IS, Parzen JS, Cifarelli CP, Rehman AA, Atik A, Bakhsheshian J, Zada G, Chang E, Giannotta S, Speckter H, Wu HM, Kondziolka D, Golfinos JG, Mathieu D, Lee CC, Warnick RE, Lunsford LD, Sheehan JP (2021) Stereotactic radiosurgery for atypical (World Health Organization II) and anaplastic (World Health Organization III) meningiomas: results from a multicenter. Int Cohort Study Neurosurg 88(5):980–988. https://doi.org/10.1093/neuros/nyaa553
Helis CA, Hughes RT, Cramer CK, Tatter SB, Laxton AW, Bourland JD, Munley MT, Chan MD (2020) Stereotactic radiosurgery for atypical and anaplastic meningiomas. World Neurosurg 144:e53–e61. https://doi.org/10.1016/j.wneu.2020.07.211
Acker G, Meinert F, Conti A, Kufeld M, Jelgersma C, Nguyen P et al (2019) Image-guided robotic radiosurgery for treatment of recurrent grade II and III meningiomas. A Single Center Study. World Neurosurg 131:e96–e107. https://doi.org/10.1016/j.wneu.2019.07.058
Zhang M, Ho AL, D’Astous M, Pendharkar AV, Choi CYH, Thompson PA et al (2016) CyberKnife stereotactic radiosurgery for atypical and malignant meningiomas. World Neurosurg 91:574-581.e1. https://doi.org/10.1016/j.wneu.2016.04.019
Vernimmen FJ, Slabbert JP (2010) Assessment of the a/ß ratios for arteriovenous malformations, meningiomas, acoustic neuromas, and the optic chiasma. Int J Radiat Biol 86(6):486–498. https://doi.org/10.3109/09553001003667982
Pinzi V, Bisogno I, Prada F, Ciusani E, Fariselli L (2018) Radiotherapy of meningioma: a treatment in need of radiobiological research. Int J Radiat Biol 94(7):621–627. https://doi.org/10.1080/09553002.2018.1478157
Pinzi V, Marchetti M, De Martin E, Cuccarini V, Tramacere I, Ghielmetti F, Fumagalli ML, Iezzoni C, Fariselli L (2020) Multisession radiosurgery for intracranial meningioma treatment: study protocol of a single arm, monocenter, prospective trial. Radiat Oncol 15(1):26. https://doi.org/10.1186/s13014-020-1478-7
Rogers L, Zhang P, Vogelbaum MA et al (2018) Intermediate-risk meningioma: initial outcomes from NRG Oncology RTOG 0539. J Neurosurg 129:35–47
Momin AA, Shao J, Soni P, Almeida JP, Suh JH, Murphy ES, Chao ST, Angelov L, Mohammadi AM, Barnett GH, Recinos PF, Kshettry VR (2021) Outcomes of salvage radiation for recurrent World Health Organization grade II meningiomas: a retrospective cohort study. J Neurooncol 152(2):373–382. https://doi.org/10.1007/s11060-021-03711-z
Mahase SS, Roth O’Brien DA, No D, Roytman M, Skafida ME, Lin E, Karakatsanis NA, Osborne JR, Brandmaier A, Pannullo SC, Ramakrishna R, Stieg PE, Knisely JPS, Ivanidze J (2021) [68Ga]-DOTATATE PET/MRI as an adjunct imaging modality for radiation treatment planning of meningiomas. Neurooncol Adv. 3(1):vdab012. https://doi.org/10.1093/noajnl/vdab012
Nassiri F, Wang JZ, Singh O, Karimi S, Dalcourt T, Ijad N, Pirouzmand N, Ng HK, Saladino A, Pollo B, Dimeco F, Yip S, Gao A, Aldape KD, Zadeh G (2021) International Consortium on Meningiomas. Loss of H3K27me3 in meningiomas. Neuro Oncol 23(8):1282–1291. https://doi.org/10.1093/neuonc/noab036
Al-Rashed M, Foshay K, Abedalthagafi M (2020) Recent advances in meningioma immunogenetics. Front Oncol 9:1472. https://doi.org/10.3389/fonc.2019.01472
Acknowledgements
The authors would like to thank Rosalind Hendricks for her help with English editing.
Funding
Funding was provided by Accuray.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Partial funding with an Accuray International grant. Marcello Marchetti and Elena De Martin received speaker honorarium from Accuray. The other authors have no financial interests.
Research involving human and animals rights
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Foundation IRCCS Neurological Institute Carlo Besta ethical committee, reference number 201/2011, and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This article does not contain any studies with animals performed by any of the authors.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Marchetti, M., Pinzi, V., Iezzoni, C. et al. Multisession radiosurgery for grade 2 (WHO), high risk meningiomas. A phase II clinical trial. J Neurooncol 157, 397–403 (2022). https://doi.org/10.1007/s11060-022-03978-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-022-03978-w