Abstract
Purpose
Young adults with isocitrate-dehydrogenase wild-type (IDH-WT) glioblastoma (GBM) represent a rare, understudied population compared to pediatric high-grade glioma, IDH-mutant GBM, or IDH-WT GBM in older patients. We aimed to explore the prognostic impact of epidermal growth factor receptor copy number gain (EGFR CN gain), one of the most common genetic alterations in IDH-WT glioma, in young adults with IDH-WT GBM.
Methods
We performed a retrospective cohort study of patients 18–45 years old with newly diagnosed, IDH-WT GBM whose tumors underwent next-generation sequencing at our institution between 2014 and 2018. The impact of EGFR CN gain on time to tumor progression (TTP) and overall survival (OS) was assessed. A validation cohort of patients 18–45 years old with IDH-WT GBM was analyzed from The Cancer Genome Atlas (TCGA).
Results
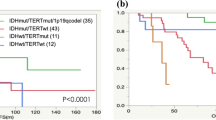
Ten of 28 patients (36%) from our institution had EGFR CN gain, which was associated with shorter TTP (median 6.5 vs. 11.9 months; p = 0.06) and OS (median 16.3 vs. 23.5 months; p = 0.047). The negative prognostic impact of EGFR CN gain on OS persisted in a multivariate model (HR 6.40, 95% CI 1.3–31.0, p = 0.02). In the TCGA cohort (N = 43), EGFR CN gain was associated with shorter TTP and worse OS, although these did not reach statistical significance (TTP, median 11.5 vs. 14.4 months, p = 0.18; OS, median 23.6 vs. 27.8 months; p = 0.18).
Conclusions
EGFR CN gain may be associated with inferior outcomes in young adults with newly diagnosed, IDH-WT GBM, suggesting a potential role for targeting EGFR in this population.


Similar content being viewed by others
References
Ostrom QT, Gittleman H, Liao P et al (2017) CBTRUS Statistical Report: primary brain and other central nervous system tumors diagnosed in the United States in 2010–2014. Neuro Oncol 19(suppl_5):v1–v88. https://doi.org/10.1093/neuonc/nox158
Ohgaki H, Kleihues P (2007) Genetic pathways to primary and secondary glioblastoma. Am J Pathol 170(5):1445–1453. https://doi.org/10.2353/ajpath.2007.070011
Ohgaki H, Dessen P, Jourde B et al (2004) Genetic pathways to glioblastoma. Cancer Res 64(19):6892–6899. https://doi.org/10.1158/0008-5472.CAN-04-1337
Sanson M, Marie Y, Paris S et al (2009) Isocitrate dehydrogenase 1 codon 132 mutation is an important prognostic biomarker in gliomas. J Clin Oncol 27(25):4150–4154. https://doi.org/10.1200/JCO.2009.21.9832
Hamisch C, Ruge M, Kellermann S et al (2017) Impact of treatment on survival of patients with secondary glioblastoma. J Neurooncol 133(2):309–313. https://doi.org/10.1007/s11060-017-2415-y
Popov S, Jury A, Laxton R et al (2013) IDH1-associated primary glioblastoma in young adults displays differential patterns of tumour and vascular morphology. PLoS ONE 8(2):e56328. https://doi.org/10.1371/journal.pone.0056328
Jha P, Suri V, Singh G et al (2011) Characterization of molecular genetic alterations in GBMs highlights a distinctive molecular profile in young adults. Diagnostic Mol Pathol 20(4):225–232. https://doi.org/10.1097/PDM.0b013e31821c30bc
Kiesel S, Pezzutto A, Haas R, Moldenhauer G, Dörken B (1988) Functional evaluation of CD19- and CD22-negative variants of B-lymphoid cell lines. Immunology 64(3):445–450
Vadgaonkar R, Epari S, Chinnaswamy G et al (2018) Distinct demographic profile and molecular markers of primary CNS tumor in 1873 adolescent and young adult patient population. Child’s Nerv Syst 34(8):1489–1495. https://doi.org/10.1007/s00381-018-3785-y
Flavahan WA, Drier Y, Liau BB et al (2016) Insulator dysfunction and oncogene activation in IDH mutant gliomas. Nature 529(7584):110–114. https://doi.org/10.1038/nature16490
Parsons DW, Jones S, Zhang X et al (2008) An integrated genomic analysis of human glioblastoma multiforme. Science (80-) 321(5897):1807–1812. https://doi.org/10.1126/science.1164382
Louis DN, Perry A, Reifenberger G et al (2016) The 2016 World Health Organization classification of tumors of the central nervous system: a summary. Acta Neuropathol 131(6):803–820. https://doi.org/10.1007/s00401-016-1545-1
Figueroa ME, Abdel-Wahab O, Lu C et al (2010) Leukemic IDH1 and IDH2 mutations result in a hypermethylation phenotype, disrupt TET2 function, and impair hematopoietic differentiation. Cancer Cell 18(6):553–567. https://doi.org/10.1016/j.ccr.2010.11.015
Waitkus MS, Diplas BH, Yan H (2016) Isocitrate dehydrogenase mutations in gliomas. Neuro Oncol 18(1):16–26. https://doi.org/10.1093/neuonc/nov136
Jones C, Karajannis MA, Jones DTW et al (2016) Pediatric high-grade glioma: biologically and clinically in need of new thinking. Neuro Oncol 19(2):now101. https://doi.org/10.1093/neuonc/now101
Wick W, van den Bent MJ (2018) First results on the DCVax phase III trial: raising more questions than providing answers. Neuro Oncol 20(10):1283–1284. https://doi.org/10.1093/neuonc/noy125
Ferguson SD, Xiu J, Weathers S-P et al (2016) GBM-associated mutations and altered protein expression are more common in young patients. Oncotarget 7(43):69466–69478. https://doi.org/10.18632/oncotarget.11617
Zhang R, Shi Z, Chen H et al (2016) Biomarker-based prognostic stratification of young adult glioblastoma. Oncotarget 7(4):5030–5041. https://doi.org/10.18632/oncotarget.5456
Brennan CW, Verhaak RGW, McKenna A et al (2013) The somatic genomic landscape of glioblastoma. Cell 155(2):462–477. https://doi.org/10.1016/j.cell.2013.09.034
Binder ZA, Thorne AH, Bakas S et al (2018) Epidermal growth factor receptor extracellular domain mutations in glioblastoma present opportunities for clinical imaging and therapeutic development. Cancer Cell 34(1):163–177.e7. https://doi.org/10.1016/j.ccell.2018.06.006
Thompson JC, Yee SS, Troxel AB et al (2016) Detection of therapeutically targetable driver and resistance mutations in lung cancer patients by next-generation sequencing of cell-free circulating tumor DNA. Clin Cancer Res 22(23):5772–5782. https://doi.org/10.1158/1078-0432.CCR-16-1231
Talevich E, Shain AH, Botton T, Bastian BC (2016) CNVkit: genome-wide copy number detection and visualization from targeted DNA sequencing. PLoS Comput Biol 12(4):e1004873. https://doi.org/10.1371/journal.pcbi.1004873
Liu J, Lichtenberg T, Hoadley KA et al (2018) An integrated TCGA pan-cancer clinical data resource to drive high-quality survival outcome analytics. Cell 173(2):400–416.e11. https://doi.org/10.1016/j.cell.2018.02.052
Chen J-R, Xu H-Z, Yao Y, Qin Z-Y (2015) Prognostic value of epidermal growth factor receptor amplification and EGFRvIII in glioblastoma: meta-analysis. Acta Neurol Scand 132(5):310–322. https://doi.org/10.1111/ane.12401
Smith JS, Tachibana I, Passe SM et al (2001) PTEN mutation, EGFR amplification, and outcome in patients with anaplastic astrocytoma and glioblastoma multiforme. J Natl Cancer Inst 93(16):1246–1256
Shinojima N, Tada K, Shiraishi S et al (2003) Prognostic value of epidermal growth factor receptor in patients with glioblastoma multiforme. Cancer Res 63(20):6962–6970
Brodbelt A, Greenberg D, Winters T et al (2015) Glioblastoma in England: 2007–2011. Eur J Cancer 51(4):533–542. https://doi.org/10.1016/j.ejca.2014.12.014
Curran WJ, Scott CB, Horton J et al (1993) Recursive partitioning analysis of prognostic factors in three Radiation Therapy Oncology Group malignant glioma trials. J Natl Cancer Inst 85(9):704–710
Yan H, Parsons DW, Jin G et al (2009) IDH1 and IDH2 mutations in gliomas. N Engl J Med 360(8):765–773. https://doi.org/10.1056/NEJMoa0808710
Yang P, Zhang W, Wang Y et al (2015) IDH mutation and MGMT promoter methylation in glioblastoma: results of a prospective registry. Oncotarget 6(38):40896–40906. https://doi.org/10.18632/oncotarget.5683
Hartmann C, Hentschel B, Simon M et al (2013) Long-term survival in primary glioblastoma with versus without isocitrate dehydrogenase mutations. Clin Cancer Res 19(18):5146–5157. https://doi.org/10.1158/1078-0432.CCR-13-0017
Molenaar RJ, Verbaan D, Lamba S et al (2014) The combination of IDH1 mutations and MGMT methylation status predicts survival in glioblastoma better than either IDH1 or MGMT alone. Neuro Oncol 16(9):1263–1273. https://doi.org/10.1093/neuonc/nou005
Ohgaki H, Kleihues P (2013) The definition of primary and secondary glioblastoma. Clin Cancer Res 19(4):764–772. https://doi.org/10.1158/1078-0432.CCR-12-3002
Miller JJ, Shih HA, Andronesi OC, Cahill DP (2017) Isocitrate dehydrogenase-mutant glioma: evolving clinical and therapeutic implications. Cancer 123(23):4535–4546. https://doi.org/10.1002/cncr.31039
Berghoff AS, Kiesel B, Widhalm G et al (2017) Correlation of immune phenotype with IDH mutation in diffuse glioma. Neuro Oncol 19(11):1460–1468. https://doi.org/10.1093/neuonc/nox054
Turcan S, Rohle D, Goenka A et al (2012) IDH1 mutation is sufficient to establish the glioma hypermethylator phenotype. Nature 483(7390):479–483. https://doi.org/10.1038/nature10866
Karpel-Massler G, Ishida CT, Bianchetti E et al (2017) Induction of synthetic lethality in IDH1-mutated gliomas through inhibition of Bcl-xL. Nat Commun 8(1):1067. https://doi.org/10.1038/s41467-017-00984-9
Molenaar RJ, Botman D, Smits MA et al (2015) Radioprotection of IDH1-mutated cancer cells by the IDH1-mutant inhibitor AGI-5198. Cancer Res 75(22):4790–4802. https://doi.org/10.1158/0008-5472.CAN-14-3603
Sulkowski PL, Corso CD, Robinson ND et al (2017) 2-Hydroxyglutarate produced by neomorphic IDH mutations suppresses homologous recombination and induces PARP inhibitor sensitivity. Sci Transl Med 9(375):eaal2463. https://doi.org/10.1126/scitranslmed.aal2463
Dang L, Yen K, Attar EC (2016) IDH mutations in cancer and progress toward development of targeted therapeutics. Ann Oncol 27(4):599–608. https://doi.org/10.1093/annonc/mdw013
Leibetseder A, Ackerl M, Flechl B et al (2013) Outcome and molecular characteristics of adolescent and young adult patients with newly diagnosed primary glioblastoma: a study of the Society of Austrian Neurooncology (SANO). Neuro Oncol 15(1):112–121. https://doi.org/10.1093/neuonc/nos283
Kleinschmidt-DeMasters BK, Meltesen L, McGavran L, Lillehei KO (2006) Characterization of glioblastomas in young adults. Brain Pathol 16(4):273–286. https://doi.org/10.1111/j.1750-3639.2006.00029.x
Korshunov A, Sycheva R, Golanov A (2005) The prognostic relevance of molecular alterations in glioblastomas for patients age %3c 50 years. Cancer 104(4):825–832. https://doi.org/10.1002/cncr.21221
Zou P, Xu H, Chen P et al (2013) IDH1/IDH2 mutations define the prognosis and molecular profiles of patients with gliomas: a meta-analysis. PLoS ONE 8(7):e68782. https://doi.org/10.1371/journal.pone.0068782
Lassman AB, Roberts-Rapp L, Sokolova I et al (2019) Comparison of biomarker assays for EGFR: implications for precision medicine in patients with glioblastoma. Clin Cancer Res 25(11):3259–3265. https://doi.org/10.1158/1078-0432.CCR-18-3034
Lassman AB, van den Bent MJ, Gan HK et al (2019) Safety and efficacy of depatuxizumab mafodotin + temozolomide in patients with EGFR -amplified, recurrent glioblastoma: results from an international phase I multicenter trial. Neuro Oncol 21(1):106–114. https://doi.org/10.1093/neuonc/noy091
Zhao J, Chen AX, Gartrell RD et al (2019) Immune and genomic correlates of response to anti-PD-1 immunotherapy in glioblastoma. Nat Med. https://doi.org/10.1038/s41591-019-0349-y
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Research involving human and animal participants
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hoffman, D.I., Abdullah, K.G., McCoskey, M. et al. Negative prognostic impact of epidermal growth factor receptor copy number gain in young adults with isocitrate dehydrogenase wild-type glioblastoma. J Neurooncol 145, 321–328 (2019). https://doi.org/10.1007/s11060-019-03298-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-019-03298-6