Abstract
Propose
To examine the association between trial sponsorship sources, self-reported conflicts of interest (COI), and study and author characteristics in central nervous system (CNS) oncology clinical trials (CT).
Methods
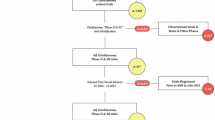
MEDLINE search was performed for original CT on “Central Nervous System Neoplasms“[Mesh]. The investigators assessed for relationships between funding source (industry, academic or cooperative, none, not described), COI (presented, none, or not reported), CT, and author characteristics.
Results
From 2010 to 2015, 319 CT were considered eligible. The majority of the studies involved primary gliomas (55.2%) and were Phase II CT (59.2%). Drug therapy was investigated in 83.0% of the CT. The remaining studies investigated surgery or radiotherapy. A minority of papers were published in journals with impact factor (IF) higher than > 10 (16%) or in regions other than North America and Europe (20.4%). Overall, 83.1% of studies disclosed funding sources: 32.6% from industry alone, 33.9% from an academic or cooperative group, and 10.7% from a mixed funding model. COI data was reported by 85.9% of trials, of which 56.2% reported no COI and 43.8% reported a related COI. Significant predictors for sponsorship (industry and/or academia) on univariate analysis were study design, type of intervention, journal impact factor, study conclusion, transparency of COI and presence of COI. On multivariate analysis, type of intervention, (P < 0.001), journal impact factor (IF) (P = 0.003), presence of COI (P < 0.001) and study conclusion (P = 0.003) remained significant predictors of sponsorship. For predicting COI, significant variables on univariate analysis were disease type, type of intervention, journal IF, funding source, and intervention arm being related to sponsor. On multivariate analysis, disease type (P = 0.003), journal IF (P < 0.001), type of intervention (P = 0.001), and funding source (P = 0.008) remained significant.
Conclusions
The majority of CNS CT reported some external funding sources and non-related COI. We identified that drug trials, higher IF, presence of COI, and a neutral or negative study conclusion are associated with external funding. Likewise drug trials, higher IF, and glioma trials are associated with presence of COI.
Similar content being viewed by others
References
Sun GH, Steinberg JD, Jagsi R (2012) The calculus of national medical research policye United States versus Asia. New Engl J Med 367(8):687–690
Moses H III, Dorsey ER (2012) Biomedical research in an age of austerity. JAMA 308(22):2341–2342
Dorsey ER, de Roulet J, Thompson JP, Reminick JI, Thai A, White-Stellato Z, Beck CA, George BP, Moses H (2010) Funding of US biomedical research, 2003–2008. JAMA 303(2):137–143
Jagsi R, Sheets N, Jankovic A, Motomura AR, Amarnath S, Ubel PA (2009) Frequency, nature, effects, and correlates of conflicts of interest in published clinical cancer research. Cancer 115(12):2783–2791
Booth CM, Cescon DW, Wang L, Tannock IF, Krzyzanowska MK (2008) Evolution of the randomized controlled trial in oncology over three decades. J Clin Oncol 26(33):5458–5464
Peppercorn J, Blood E, Winer E, Partridge A (2007) Association between pharmaceutical involvement and outcomes in breast cancer clinical trials. Cancer 109(7):1239–1246
Moraes FY, Leite ET, Hamstra DA, Feng FY, Arruda FF, Gadia R, Abreu CE, Marta GN, Hanna SA, Silva JL, Carvalho HA (2018) Self-reported conflicts of interest and trial sponsorship of clinical trials in prostate cancer involving radiotherapy. Am J Clin Oncol 41(1):6–12
Djubegovic B, Lacevic M, Cantor A et al (2000) The uncertainty principle and industry-sponsored research. Lancet 356(9230):635–638
Moraes FY, Mendez LC, Taunk NK, Raman S, Suh JH, Souhami L, Slotman BJ, Weltman E, Spratt DE, Berlin A, Marta GN (2018) Funding source, conflict of interest and positive conclusions in neuro-oncology clinical trials. J Neuro-oncol 136:585–593
National Cancer Institute (2014). NCI funded research portfolio. https://fundedresearch.cancer.gov/nciportfolio. Accessed 25 June 2017
Moraes FY, Bonifacio LA, Marta GN, Hanna SA, Atallah ÁN, Moraes VY, Silva JL, Carvalho HA (2015) Hierarchy of evidence referring to the central nervous system in a high-impact radiation oncology journal: a 10-year assessment. Descriptive critical appraisal study. Sao Paulo Med J 133(4):307–313
Bidros DS, Vogelbaum MA (2009) Novel drug delivery strategies in neuro-oncology. Neurotherapeutics 6(3):539–546
Davidson RA (1986) Source of funding and outcome of clinical trials. J Gener Intern Med 1(3):155–158
Yaphe J, Edman R, Knishkowy B, Herman J (2001) The association between funding by commercial interests and study outcome in randomized controlled drug trials. Fam Pract 18(6):565–568
Kjaergard L, Als-Nielsen B (2002) Association between competing interests and authors conclusions: epidemiological study of randomised clinical trials published in the BMJ. BMJ 325(7358):249
Hirsch BR, Califf RM, Cheng SK, Tasneem A, Horton J, Chiswell K, Schulman KA, Dilts DM, Abernethy AP (2013) Characteristics of oncology clinical trials: insights from a systematic analysis of ClinicalTrials. gov. JAMA Intern Med 173(11):972–979
Cihoric N, Tsikkinis A, Minniti G, Lagerwaard FJ, Herrlinger U, Mathier E, Soldatovic I, Jeremic B, Ghadjar P, Elicin O, Lössl K (2017) Current status and perspectives of interventional clinical trials for glioblastoma-analysis of ClinicalTrials. gov. Radiat Oncol 12(1):1
Lloyd S, Buscariollo D, Gross CP, Makarov DV, Yu JB, Aneja S (2012) The funding of phase III clinical trials examining radiation compared with that of other modalities. Int J Radiat Oncol Biol Phys 84(3):S44–S45
Schott G, Pachl H, Limbach U, Gundert-Remy U, Ludwig WD, Lieb K (2010) The financing of drug trials by pharmaceutical companies and its consequences: part 1: a qualitative, systematic review of the literature on possible influences on the findings, protocols, and quality of drug trials. Dtsch Aerzteblatt Int 107(16):279
Schott G, Pachl H, Limbach U, Gundert-Remy U, Lieb K, Ludwig WD (2010) The financing of drug trials by pharmaceutical companies and its consequences: part 2: a qualitative, systematic review of the literature on possible influences on authorship, access to trial data, and trial registration and publication. Dtsch Aerzteblatt Int 107(17):295
Easterbrook PJ, Gopalan R, Berlin JA, Matthews DR (1991) Publication bias in clinical research. The Lancet 337(8746):867–872
McCoy MS, Emanuel EJ (2017) Why there are no “potential” conflicts of interest. JAMA 317(17):1721–1722
Fineberg HV (2017) Conflict of interest: why does it matter? JAMA 317(17):1717–1718
Riechelmann RP, Wang L, O’Carroll A, Krzyzanowska MK (2007) Disclosure of conflicts of interest by authors of clinical trials and editorials in oncology. J Clin Oncol 25(29):4642–4647
Agrawal S, Brennan N, Budetti P (2013) The Sunshine Act—effects on physicians. New Engl J Med 368(22):2054–2057
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors have no conflicts of interests to report.
Human and animal participants
This article does not contain any studies with human participants or animals performed by any of the authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Raman, S., Moraes, F.Y., Mendez, L.C. et al. The relationship of study and authorship characteristics on trial sponsorship and self-reported conflicts of interest among neuro-oncology clinical trials. J Neurooncol 139, 195–203 (2018). https://doi.org/10.1007/s11060-018-2860-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-018-2860-2