Abstract
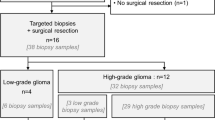
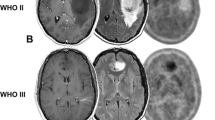
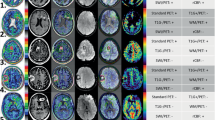
Treatment-related changes can be difficult to differentiate from progressive glioma using MRI with contrast (CE). The purpose of this study is to compare the sensitivity and specificity of 18F-DOPA-PET and MRI in patients with recurrent glioma. Thirteen patients with MRI findings suspicious for recurrent glioma were prospectively enrolled and underwent 18F-DOPA-PET and MRI for neurosurgical planning. Stereotactic biopsies were obtained from regions of concordant and discordant PET and MRI CE, all within regions of T2/FLAIR signal hyperintensity. The sensitivity and specificity of 18F-DOPA-PET and CE were calculated based on histopathologic analysis. Receiver operating characteristic curve analysis revealed optimal tumor to normal (T/N) and SUVmax thresholds. In the 37 specimens obtained, 51% exhibited MRI contrast enhancement (M+) and 78% demonstrated 18F-DOPA-PET avidity (P+). Imaging characteristics included M−P− in 16%, M−P+ in 32%, M+P+ in 46% and M+P− in 5%. Histopathologic review of biopsies revealed grade II components in 16%, grade III in 43%, grade IV in 30% and no tumor in 11%. MRI CE sensitivity for recurrent tumor was 52% and specificity was 50%. PET sensitivity for tumor was 82% and specificity was 50%. A T/N threshold > 2.0 altered sensitivity to 76% and specificity to 100% and SUVmax > 1.36 improved sensitivity and specificity to 94 and 75%, respectively. 18F-DOPA-PET can provide increased sensitivity and specificity compared with MRI CE for visualizing the spatial distribution of recurrent gliomas. Future studies will incorporate 18F-DOPA-PET into re-irradiation target volume delineation for RT planning.


Similar content being viewed by others
References
Siegel RL, Miller KD, Jemal A (2016) Cancer statistics, 2016. CA Cancer J Clin 66:7–30
Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, Belanger K, Brandes AA, Marosi C, Bogdahn U, Curschmann J, Janzer RC, Ludwin SK, Gorlia T, Allgeier A, Lacombe D, Cairncross JG, Eisenhauer E, Mirimanoff RO (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352:987–996
Stupp R, Taillibert S, Kanner AA, Kesari S, Steinberg DM, Toms SA, Taylor LP, Lieberman F, Silvani A, Fink KL, Barnett GH, Zhu JJ, Henson JW, Engelhard HH, Chen TC, Tran DD, Sroubek J, Tran ND, Hottinger AF, Landolfi J, Desai R, Caroli M, Kew Y, Honnorat J, Idbaih A, Kirson ED, Weinberg U, Palti Y, Hegi ME, Ram Z (2015) Maintenance therapy with tumor-treating fields plus temozolomide vs temozolomide alone for glioblastoma: a randomized clinical trial. J Am Med Assoc 314:2535–2543
Youland RS, Schomas DA, Brown PD, Nwachukwu C, Buckner JC, Giannini C, Parney IF, Laack NN (2013) Changes in presentation, treatment, and outcomes of adult low-grade gliomas over the past fifty years. Neuro Oncol 15:1102–1110
Shaw EG, Wang M, Coons SW, Brachman DG, Buckner JC, Stelzer KJ, Barger GR, Brown PD, Gilbert MR, Mehta MP (2012) Randomized trial of radiation therapy plus procarbazine, lomustine, and vincristine chemotherapy for supratentorial adult low-grade glioma: initial results of RTOG 9802. J Clin Oncol 30:3065–3070
de Wit MC, de Bruin HG, Eijkenboom W, Sillevis Smitt PA, van den Bent MJ (2004) Immediate post-radiotherapy changes in malignant glioma can mimic tumor progression. Neurology 63:535–537
Watling CJ, Lee DH, Macdonald DR, Cairncross JG (1994) Corticosteroid-induced magnetic resonance imaging changes in patients with recurrent malignant glioma. J Clin Oncol 12:1886–1889
Wen PY, Macdonald DR, Reardon DA, Cloughesy TF, Sorensen AG, Galanis E, Degroot J, Wick W, Gilbert MR, Lassman AB, Tsien C, Mikkelsen T, Wong ET, Chamberlain MC, Stupp R, Lamborn KR, Vogelbaum MA, van den Bent MJ, Chang SM (2010) Updated response assessment criteria for high-grade gliomas: response assessment in neuro-oncology working group. J Clin Oncol 28:1963–1972
Chen W, Silverman DH, Delaloye S, Czernin J, Kamdar N, Pope W, Satyamurthy N, Schiepers C, Cloughesy T (2006) 18F-FDOPA PET imaging of brain tumors: comparison study with 18F-FDG PET and evaluation of diagnostic accuracy. J Nucl Med 47:904–911
Dunet V, Pomoni A, Hottinger A, Nicod-Lalonde M, Prior JO (2016) Performance of 18F-FET versus 18F-FDG-PET for the diagnosis and grading of brain tumors: systematic review and meta-analysis. Neuro Oncol 18:426–434
Youland RS, Kitange GJ, Peterson TE, Pafundi DH, Ramiscal JA, Pokorny JL, Giannini C, Laack NN, Parney IF, Lowe VJ, Brinkmann DH, Sarkaria JN (2013) The role of LAT1 in (18)F-DOPA uptake in malignant gliomas. J Neurooncol 111:11–18
Pafundi DH, Laack NN, Youland RS, Parney IF, Lowe VJ, Giannini C, Kemp BJ, Grams MP, Morris JM, Hoover JM, Hu LS, Sarkaria JN, Brinkmann DH (2013) Biopsy validation of 18F-DOPA PET and biodistribution in gliomas for neurosurgical planning and radiotherapy target delineation: results of a prospective pilot study. Neuro Oncol 15:1058–1067
Karunanithi S, Sharma P, Kumar A, Khangembam BC, Bandopadhyaya GP, Kumar R, Goenka A, Gupta DK, Malhotra A, Bal C (2013) Comparative diagnostic accuracy of contrast-enhanced MRI and (18)F-FDOPA PET-CT in recurrent glioma. Eur Radiol 23:2628–2635
Kelly PJ, Daumas-Duport C, Kispert DB, Kall BA, Scheithauer BW, Illig JJ (1987) Imaging-based stereotaxic serial biopsies in untreated intracranial glial neoplasms. J Neurosurg 66:865–874
Hochberg FH, Pruitt A (1980) Assumptions in the radiotherapy of glioblastoma. Neurology 30:907–911
Wallner KE, Galicich JH, Krol G, Arbit E, Malkin MG (1989) Patterns of failure following treatment for glioblastoma multiforme and anaplastic astrocytoma. Int J Radiat Oncol Biol Phys 16:1405–1409
Macdonald DR, Cascino TL, Schold SC Jr, Cairncross JG (1990) Response criteria for phase II studies of supratentorial malignant glioma. J Clin Oncol 8:1277–1280
Bell C, Dowson N, Puttick S, Gal Y, Thomas P, Fay M, Smith J, Rose S (2015) Increasing feasibility and utility of (18)F-FDOPA PET for the management of glioma. Nucl Med Biol 42:788–795
Beuthien-Baumann B, Bredow J, Burchert W, Fuchtner F, Bergmann R, Alheit HD, Reiss G, Hliscs R, Steinmeier R, Franke WG, Johannsen B, Kotzerke J (2003) 3-O-methyl-6-[18F]fluoro-l-DOPA and its evaluation in brain tumour imaging. Eur J Nucl Med Mol Imaging 30:1004–1008
Becherer A, Karanikas G, Szabo M, Zettinig G, Asenbaum S, Marosi C, Henk C, Wunderbaldinger P, Czech T, Wadsak W, Kletter K (2003) Brain tumour imaging with PET: a comparison between [18F]fluorodopa and [11C]methionine. Eur J Nucl Med Mol Imaging 30:1561–1567
Karunanithi S, Sharma P, Kumar A, Gupta DK, Khangembam BC, Ballal S, Kumar R, Bal C (2014) Can (18)F-FDOPA PET/CT predict survival in patients with suspected recurrent glioma? a prospective study. Eur J Radiol 83:219–225
Chiaravalloti A, Fiorentini A, Villani V, Carapella C, Pace A, Di Pietro B, Di Russo C, Palumbo B, Floris R, Schillaci O (2015) Factors affecting (1)(8)F FDOPA standardized uptake value in patients with primary brain tumors after treatment. Nucl Med Biol 42:355–359
Janvier L, Olivier P, Blonski M, Morel O, Vignaud JM, Karcher G, Taillandier L, Verger A (2015) Correlation of SUV-derived indices with tumoral aggressiveness of gliomas in static 18F-FDOPA PET: use in clinical practice. Clin Nucl Med 40:e429–435
Nioche C, Soret M, Gontier E, Lahutte M, Dutertre G, Dulou R, Capelle L, Guillevin R, Foehrenbach H, Buvat I (2013) Evaluation of quantitative criteria for glioma grading with static and dynamic 18F-FDopa PET/CT. Clin Nucl Med 38:81–87
Chen MW, Morsy AA, Liang S, Ng WH (2016) Re-do craniotomy for recurrent grade IV glioblastomas: impact and outcomes from the National Neuroscience Institute Singapore. World Neurosurg 87:439–445
Oppenlander ME, Wolf AB, Snyder LA, Bina R, Wilson JR, Coons SW, Ashby LS, Brachman D, Nakaji P, Porter RW, Smith KA, Spetzler RF, Sanai N (2014) An extent of resection threshold for recurrent glioblastoma and its risk for neurological morbidity. J Neurosurg 120:846–853
Ringel F, Pape H, Sabel M, Krex D, Bock HC, Misch M, Weyerbrock A, Westermaier T, Senft C, Schucht P, Meyer B, Simon M (2016) Clinical benefit from resection of recurrent glioblastomas: results of a multicenter study including 503 patients with recurrent glioblastomas undergoing surgical resection. Neuro Oncol 18:96–104
Fogh SE, Andrews DW, Glass J, Curran W, Glass C, Champ C, Evans JJ, Hyslop T, Pequignot E, Downes B, Comber E, Maltenfort M, Dicker AP, Werner-Wasik M (2010) Hypofractionated stereotactic radiation therapy: an effective therapy for recurrent high-grade gliomas. J Clin Oncol 28:3048–3053
Shepherd SF, Laing RW, Cosgrove VP, Warrington AP, Hines F, Ashley SE, Brada M (1997) Hypofractionated stereotactic radiotherapy in the management of recurrent glioma. Int J Radiat Oncol Biol Phys 37:393–398
Cho KH, Hall WA, Gerbi BJ, Higgins PD, McGuire WA, Clark HB (1999) Single dose versus fractionated stereotactic radiotherapy for recurrent high-grade gliomas. Int J Radiat Oncol Biol Phys 45:1133–1141
Niyazi M, Jansen NL, Rottler M, Ganswindt U, Belka C (2014) Recurrence pattern analysis after re-irradiation with bevacizumab in recurrent malignant glioma patients. Radiat Oncol 9:299
Grosu AL, Weber WA, Riedel E, Jeremic B, Nieder C, Franz M, Gumprecht H, Jaeger R, Schwaiger M, Molls M (2005) l-(methyl-11C) methionine positron emission tomography for target delineation in resected high-grade gliomas before radiotherapy. Int J Radiat Oncol Biol Phys 63:64–74
Arbizu J, Tejada S, Marti-Climent JM, Diez-Valle R, Prieto E, Quincoces G, Vigil C, Idoate MA, Zubieta JL, Penuelas I, Richter JA (2012) Quantitative volumetric analysis of gliomas with sequential MRI and (1)(1)C-methionine PET assessment: patterns of integration in therapy planning. Eur J Nucl Med Mol Imaging 39:771–781
Acknowledgements
This work has been accepted for presentation at the 2017 ASTRO Annual Meeting (September 24–27, 2017) in San Diego, California.
Funding
This work was supported by a generous Grant from Brains Together for a Cure, the National Cancer Institute (CA178200) and by a Mayo Brain Tumor NIH SPORE Grant (CA108961).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No actual or potential conflicts of interest exist pertaining to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
11060_2018_2750_MOESM1_ESM.tif
Supplementary material 1. Supplemental Fig. 1 Receiver operating characteristic analysis for SUVmax and the presence of tumor (A), SUVmax and the presence of high-grade disease (B), T/N ratio and the presence of tumor (C) and T/N ratio and the presence of high-grade disease (D) (TIF 62 KB)
Rights and permissions
About this article
Cite this article
Youland, R.S., Pafundi, D.H., Brinkmann, D.H. et al. Prospective trial evaluating the sensitivity and specificity of 3,4-dihydroxy-6-[18F]-fluoro-l-phenylalanine (18F-DOPA) PET and MRI in patients with recurrent gliomas. J Neurooncol 137, 583–591 (2018). https://doi.org/10.1007/s11060-018-2750-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-018-2750-7