Abstract
Background
DNA methylation inhibitors are logical therapeutic candidates for ependymomas originating in the posterior fossa of the brain. Our objective was to test the safety of infusing 5-Azacytidine (5-AZA), a DNA methylation inhibitor, directly into cerebrospinal fluid (CSF) spaces of the fourth ventricle or tumor resection cavity in children with recurrent ependymoma originating in the posterior fossa.
Materials and methods
In patients with recurrent ependymoma whose disease originated in the posterior fossa, a maximal safe subtotal tumor resection was performed. At the conclusion of the tumor resection, a catheter was surgically placed into the fourth ventricle or tumor resection cavity and attached to a ventricular access device. CSF flow from the posterior fossa to the sacrum was confirmed by CINE phase contrast magnetic resonance imaging (MRI) postoperatively. 12 consecutive weekly 10 milligram (mg) infusions of 5-Azacytidine (AZA) were planned. Disease response was monitored with MRI scans and CSF cytology.
Results
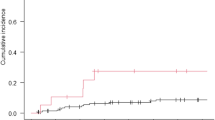
Six patients were enrolled. One patient was withdrawn prior to planned 5-AZA infusions due to surgical complications after tumor resection. The remaining five patients received 8, 12, 12, 12, and 12 infusions, respectively. There were no serious adverse events or new neurological deficits attributed to 5-AZA infusions. All five patients with ependymoma who received 5-AZA infusions had progressive disease. Two of the five patients, however, were noted to have decrease in the size of at least one intraventricular lesion.
Conclusion
5-AZA can be infused into the fourth ventricle or posterior fossa tumor resection cavity without causing neurological toxicity. Future studies with higher doses and/or increased dosing frequency are warranted.


Similar content being viewed by others

References
Merchant TE, Li C, Xiong X, Kun LE, Boop FA, Sanford RA (2009) Conformal radiotherapy after surgery for paediatric ependymoma: a prospective study. Lancet Oncol 10(3):258–266
Bouffet E, Hawkins CE, Ballourah W, Taylor MD, Bartels UK, Schoenhoff N, Tsangaris E, Huang A, Kulkarni A, Mabbot DJ et al (2012) Survival benefit for pediatric patients with recurrent ependymoma treated with reirradiation. Int J Radiat Oncol Biol Phys 83(5):1541–1548
Bouffet E, Tabori U, Huang A, Bartels U (2009) Ependymoma: lessons from the past, prospects for the future. Childs Nerv Syst 25(11):1383–1384
Figarella-Branger D, Civatte M, Bouvier-Labit C, Gouvernet J, Gambarelli D, Gentet JC, Lena G, Choux M, Pellissier JF (2000) Prognostic factors in intracranial ependymomas in children. J Neurosurg 93(4):605–613
Sandberg DI, Crandall KM, Petito CK, Padgett KR, Landrum J, Babino D, He D, Solano J, Gonzalez-Brito M, Kuluz JW (2008) Chemotherapy administration directly into the fourth ventricle in a new piglet model. J Neurosurg Pediatr 1(5):373–380
Sandberg DI, Crandall KM, Koru-Sengul T, Padgett KR, Landrum J, Babino D, Petito CK, Solano J, Gonzalez-Brito M, Kuluz JW (2010) Pharmacokinetic analysis of etoposide distribution after administration directly into the fourth ventricle in a piglet model. J Neuro-Oncology 97(1):25–32
Sandberg DI, Solano J, Petito CK, Mian A, Mou C, Koru-Sengul T, Gonzalez-Brito M, Padgett KR, Luqman A, Buitrago JC et al (2010) Safety and pharmacokinetic analysis of methotrexate administered directly into the fourth ventricle in a piglet model. J Neuro-Oncology 100(3):397–406
Sandberg DI, Peet MM, Johnson MD, Cole P, Koru-Sengul T, Luqman AW (2012) Chemotherapy administration directly into the fourth ventricle in a nonhuman primate model. J Neurosurg Pediatr 9(5):530–541
Sandberg DI, Kerr ML (2016) Ventricular access device placement in the fourth ventricle to treat malignant fourth ventricle brain tumors: technical note. Childs Nerv Syst 32(4):703–707
Sandberg DI, Rytting M, Zaky W, Kerr M, Ketonen L, Kundu U, Moore BD, Yang G, Hou P, Sitton C et al (2015) Methotrexate administration directly into the fourth ventricle in children with malignant fourth ventricular brain tumors: a pilot clinical trial. J Neuro-Oncology 125(1):133–141
Wongtrakoongate P (2015) Epigenetic therapy of cancer stem and progenitor cells by targeting DNA methylation machineries. World J Stem Cells 7(1):137–148
Mack SC, Witt H, Piro RM, Gu L, Zuyderduyn S, Stutz AM, Wang X, Gallo M, Garzia L, Zayne K et al (2014) Epigenomic alterations define lethal CIMP-positive ependymomas of infancy. Nature 506(7489):445–450
McCully CL, Rodgers L, Cruz R, Peer C, Figg WD, Warren K: plasma and cerebrospinal fluid pharmacokinetics of 5-azacytidine following intravenous, intranasal, and intrathecal administration in a non-human primate model. In: International Symposium on Pediatric Neuro-Oncology, Liverpool 2016
Heideman RL, McCully C, Balis FM, Poplack DG (1993) Cerebrospinal fluid pharmacokinetics and toxicology of intraventricular and intrathecal arabinosyl-5-azacytosine (fazarabine, NSC 281272) in the nonhuman primate. Investig New Drugs 11(2–3):135–140
Lansky SB, List MA, Lansky LL, Ritter-Sterr C, Miller DR (1987) The measurement of performance in childhood cancer patients. Cancer 60(7):1651–1656
Patel RP, Sitton CW, Ketonen LM, Hou P, Johnson JM, Romo S, Fletcher S, Shah MN, Kerr M, Zaky W et al (2018) Phase-contrast cerebrospinal fluid flow magnetic resonance imaging in qualitative evaluation of patency of CSF flow pathways prior to infusion of chemotherapeutic and other agents into the fourth ventricle. Childs Nerv Syst 34(3):481–486
Acknowledgements
The authors would like to thank Ian’s Friends Foundation for grant support that helped to fund this study. We would also like to thank Allyson Lack for her assistance with image formatting. Above all, we would like to thank the brave patients who enrolled in this trial and their wonderful families.
Funding
This study was partially funded by grant support from Ian’s Friends Foundation (no grant number) that was awarded to David Sandberg, M.D.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors (Bangning Yu, M.D., Ph.D., Rajan Patel, M.D., Emilie Miesner, PA-C, Jennifer Sabin, PA-C, Sarah Smith, PA-C, Stephen Fletcher, D.O., Manish N. Shah, M.D., and Michael D. Taylor, M.D., Ph.D.) declare that they have no conflicts of interest. Author David Sandberg, M.D. received grant support from Ian’s Friends Foundation to support this study as noted above. Dr. Sandberg also received a speaker honorarium from Aesculap in 2017 for a talk at the International Society for Pediatric Neurosurgery on a topic unrelated to this study. Author John Hagan, Ph.D., has received financial support from GlaxoSmithKline for research that is unrelated to this study. Author Rachael Sirianni, Ph.D., currently receives grant support from Ian’s Friends Foundation and the National Institutes of Health, and she has received grant funding in the past from the Rick Oehme Foundation, the Ben and Catherine Ivy Foundation, the ALS Association, the Barrow Neurological Foundation, the National Science Foundation, and the Department of Defense. Author Rachael Sirianni, Ph.D. owns stock in the company NP Therapeutics, Inc. and works as a consultant for the Ian’s Friends Foundation. None of these relationships for Dr. Sirianni have any connection to the current study.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national search committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants in the study.
Rights and permissions
About this article
Cite this article
Sandberg, D.I., Yu, B., Patel, R. et al. Infusion of 5-Azacytidine (5-AZA) into the fourth ventricle or resection cavity in children with recurrent posterior Fossa Ependymoma: a pilot clinical trial. J Neurooncol 141, 449–457 (2019). https://doi.org/10.1007/s11060-018-03055-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-018-03055-1