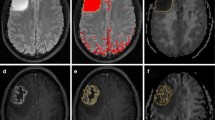
Abstract
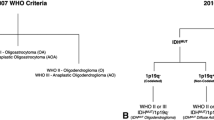
The purpose of this study is to assess the diagnostic performance of diffusion kurtosis imaging (DKI) for in vivo molecular profiling of human glioma. Normalized mean kurtosis (MKn) and mean diffusivity (MDn) metrics from DKI were assessed in 50 patients with histopathologically confirmed glioma. The results were compared in regard to the WHO-based histological findings and molecular characteristics leading to integrated diagnosis (Haarlem Consensus): isocitrate-dehydrogenase (IDH1/2) mutation status, alpha-thalassemia/mental retardation syndrome X-linked (ATRX) expression, chromosome 1p/19q loss of heterozygosity (LOH), and O6-methylguanine DNA methyltransferase (MGMT) promoter methylation status. MKn was significantly lower in tumors with IDH1/2 mutation (0.43 ± 0.09) and ATRX loss of expression (0.41 ± 0.11) than in those with IDH1/2 wild type (0.57 ± 0.09, p < 0.001) and ATRX maintained expression (0.51 ± 0.10, p = 0.004), respectively. Regarding the integrated molecular diagnosis, MKn was significantly higher in primary glioblastoma (0.57 ± 0.10) than in astrocytoma (0.39 ± 0.11, p < 0.001) and oligodendroglioma (0.47 ± 0.05, p = 0.003). MK may be used to provide insight into the human glioma molecular profile regarding IDH1/2 mutation status and ATRX expression. Considering the diagnostic and prognostic significance of these molecular markers, MK appears to be a promising in vivo biomarker for glioma. The diagnostic performance of MK seems to fit more with the integrated molecular approach than the conventional histological findings of the current WHO 2007 classification.


Similar content being viewed by others
References
Louis DN, Ohgaki H, Wiestler OD et al (2007) The 2007 WHO classification of tumours of the central nervous system. Acta Neuropathol 114(2):97–109. doi:10.1007/s00401-007-0243-4
van Cauter S, Veraart J, Sijbers J et al (2012) Gliomas: diffusion kurtosis MR imaging in grading. Radiology 263(2):492–501. doi:10.1148/radiol.12110927
Scott JN, Brasher PMA, Sevick RJ et al (2002) How often are nonenhancing supratentorial gliomas malignant? A population study. Neurology 59(6):947–949
Watanabe M, Tanaka R, Takeda N (1992) Magnetic resonance imaging and histopathology of cerebral gliomas. Neuroradiology 34(6):463–469
Dean BL, Drayer BP, Bird CR et al (1990) Gliomas: classification with MR imaging. Radiology 174(2):411–415. doi:10.1148/radiology.174.2.2153310
van Cauter S, Keyzer F de, Sima DM et al (2014) Integrating diffusion kurtosis imaging, dynamic susceptibility-weighted contrast-enhanced MRI, and short echo time chemical shift imaging for grading gliomas. Neuro Oncol 16(7):1010–1021. doi:10.1093/neuonc/not304
Kulkarni AV, Guha A, Lozano A et al (1998) Incidence of silent hemorrhage and delayed deterioration after stereotactic brain biopsy. J Neurosurg 89(1):31–35. doi:10.3171/jns.1998.89.1.0031
Kärger J (1985) NMR self-diffusion studies in heterogeneous systems. Adv Colloid Interface Sci 23:129–148. doi:10.1016/0001-8686(85)80018-X
Jensen JH, Helpern JA, Ramani A et al (2005) Diffusional kurtosis imaging: the quantification of non-gaussian water diffusion by means of magnetic resonance imaging. Magn Reson Med 53(6):1432–1440. doi:10.1002/mrm.20508
Jensen JH, Helpern JA (2010) MRI quantification of non-Gaussian water diffusion by kurtosis analysis. NMR biomed 23(7): 698–710. doi:10.1002/nbm.1518
Lu H, Jensen JH, Ramani A et al. (2006) Three-dimensional characterization of non-Gaussian water diffusion in humans using diffusion kurtosis imaging. NMR biomed 19(2): 236–247. doi:10.1002/nbm.1020
Poot DHJ, den Dekker AJ, Achten E et al (2010) Optimal experimental design for diffusion kurtosis imaging. IEEE Trans Med Imaging 29(3):819–829. doi:10.1109/TMI.2009.2037915
Raab P, Hattingen E, Franz K et al (2010) Cerebral gliomas: diffusional kurtosis imaging analysis of microstructural differences. Radiology 254(3):876–881. doi:10.1148/radiol.09090819
Jiang R, Jiang J, Zhao L et al (2015) Diffusion kurtosis imaging can efficiently assess the glioma grade and cellular proliferation. Oncotarget 6(39):42380–42393
Tan Y, Zhang H, Zhao R-F et al (2016) Comparison of the values of MRI diffusion kurtosis imaging and diffusion tensor imaging in cerebral astrocytoma grading and their association with aquaporin-4. Neurol India 64(2):265. doi:10.4103/0028-3886.177621
Louis DN, Perry A, Burger P et al (2014) International Society Of Neuropathology—Haarlem consensus guidelines for nervous system tumor classification and grading. Brain Pathol 24(5):429–435. doi:10.1111/bpa.12171
Reuss DE, Mamatjan Y, Schrimpf D et al (2015) IDH mutant diffuse and anaplastic astrocytomas have similar age at presentation and little difference in survival: a grading problem for WHO. Acta Neuropathol 129(6):867–873. doi:10.1007/s00401-015-1438-8
Reuss DE, Sahm F, Schrimpf D et al (2015) ATRX and IDH1-R132H immunohistochemistry with subsequent copy number analysis and IDH sequencing as a basis for an “integrated” diagnostic approach for adult astrocytoma, oligodendroglioma and glioblastoma. Acta Neuropathol 129(1):133–146. doi:10.1007/s00401-014-1370-3
Gerson SL (2004) MGMT: its role in cancer aetiology and cancer therapeutics. Nat Rev Cancer 4(4):296–307. doi:10.1038/nrc1319
Sahm F, Reuss D, Koelsche C et al (2014) Farewell to oligoastrocytoma: in situ molecular genetics favor classification as either oligodendroglioma or astrocytoma. Acta Neuropathol 128(4):551–559. doi:10.1007/s00401-014-1326-7
Capper D, Weissert S, Balss J et al (2010) Characterization of R132H mutation-specific IDH1 antibody binding in brain tumors. Brain Pathol 20(1):245–254. doi:10.1111/j.1750-3639.2009.00352.x
Schittenhelm J, Mittelbronn M, Meyermann R et al (2011) Confirmation of R132H mutation of isocitrate dehydrogenase 1 as an independent prognostic factor in anaplastic astrocytoma. Acta Neuropathol 122(5):651–652. doi:10.1007/s00401-011-0885-0
Hartmann C, Meyer J, Balss J et al (2009) Type and frequency of IDH1 and IDH2 mutations are related to astrocytic and oligodendroglial differentiation and age: a study of 1010 diffuse gliomas. Acta Neuropathol 118(4):469–474. doi:10.1007/s00401-009-0561-9
Thon N, Eigenbrod S, Grasbon-Frodl EM et al (2009) Novel molecular stereotactic biopsy procedures reveal intratumoral homogeneity of loss of heterozygosity of 1p/19q and TP53 mutations in World Health Organization grade II gliomas. J Neuropathol Exp Neurol 68(11):1219–1228. doi:10.1097/NEN.0b013e3181bee1f1
Hegi ME, Diserens A-C, Gorlia T et al (2005) MGMT gene silencing and benefit from temozolomide in glioblastoma. N Engl J Med 352(10):997–1003. doi:10.1056/NEJMoa043331
Tozer DJ, Jäger HR, Danchaivijitr N et al (2007) Apparent diffusion coefficient histograms may predict low-grade glioma subtype. NMR Biomed 20(1):49–57. doi:10.1002/nbm.1091
Kang Y, Choi SH, Kim Y-J et al (2011) Gliomas: histogram analysis of apparent diffusion coefficient maps with standard- or high-b-value diffusion-weighted MR imaging—correlation with tumor grade. Radiology 261(3):882–890. doi:10.1148/radiol.11110686
Falangola MF, Jensen JH, Babb JS et al (2008) Age-related non-Gaussian diffusion patterns in the prefrontal brain. J Magn Reson Imaging 28(6):1345–1350. doi:10.1002/jmri.21604
Coutu JP, Chen JJ, Rosas HD et al (2014) Non-Gaussian water diffusion in aging white matter. Neurobiol Aging 35(6):1412–1421. doi:10.1016/j.neurobiolaging.2013.12.001
Kleihues P, Soylemezoglu F, Schauble B et al (1995) Histopathology, classification, and grading of gliomas. Glia 15(3):211–221. doi:10.1002/glia.440150303
Soeda A, Hara A, Kunisada T et al. (2015) The evidence of glioblastoma heterogeneity. Sci Rep 5: 7979. doi:10.1038/srep07979
DeAngelis LM (2001) Brain tumors. N Engl J Med 344(2):114–123. doi:10.1056/NEJM200101113440207
Cha S (2006) Update on Brain Tumor Imaging: From Anatomy to Physiology. AJNR Am J Neuroradiol 27(3):475–487
Popov S, Jury A, Laxton R et al (2013) IDH1-associated primary glioblastoma in young adults displays differential patterns of tumour and vascular morphology. PLoS One 8(2):e56328. doi:10.1371/journal.pone.0056328
Lee S, Choi SH, Ryoo I et al (2015) Evaluation of the microenvironmental heterogeneity in high-grade gliomas with IDH1/2 gene mutation using histogram analysis of diffusion-weighted imaging and dynamic-susceptibility contrast perfusion imaging. J Neurooncol 121(1):141–150. doi:10.1007/s11060-014-1614-z
Kim SH, Kim H, Kim TS (2005) Clinical, histological, and immunohistochemical features predicting 1p/19q loss of heterozygosity in oligodendroglial tumors. Acta Neuropathol 110(1):27–38. doi:10.1007/s00401-005-1020-x
Acknowledgements
We thank Robert Grimm from Siemens (Erlangen, Germany) for support in image post-processing.
Author information
Authors and Affiliations
Corresponding author
Additional information
The original version of this article was revised: The second author’s family name was incorrect. The name has been updated in this version.
An erratum to this article is available at http://dx.doi.org/10.1007/s11060-016-2281-z.
Rights and permissions
About this article
Cite this article
Hempel, JM., Bisdas, S., Schittenhelm, J. et al. In vivo molecular profiling of human glioma using diffusion kurtosis imaging. J Neurooncol 131, 93–101 (2017). https://doi.org/10.1007/s11060-016-2272-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-016-2272-0