Abstract
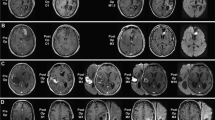
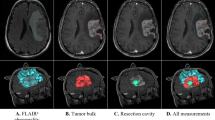
Carmustine wafers are approved for localized treatment of malignant glioma. In this study, overall changes in computed tomography (CT) and magnetic resonance (MR) images of malignant glioma patients treated with carmustine wafer implantation were evaluated. The subjects were 25 patients undergoing craniotomy for malignant glioma resection and carmustine wafer implantation. Changes in the appearance of wafers, the resection cavity, and the adjacent parenchyma on CT and MR imaging were evaluated retrospectively. On CT, the wafers changed from an initially high-dense to an iso-dense appearance. All MR studies showed a low-intense wafer within 2 days. The wafers changed to a high- or iso-intense appearance on fluid attenuated inversion recovery and T1-weighted imaging, whereas they changed to an iso- to low-intense appearance on T2-weighted imaging. Gas in the cavity increased gradually after surgery, achieved a peak at 1 week postoperatively, and then disappeared in 1–3 months. Increased volume of the resection cavity was observed in 48 % of patients. Regarding changes in the adjacent parenchyma, obvious contrast enhancement at the wall of the resection cavity was seen in 91 % of cases at 1 month, but this disappeared gradually. Edema around the resection cavity was increased in 7 patients (28 %), of whom only two experienced symptoms due to edema. We conclude that these radiological changes after carmustine wafer implantation should be carefully followed up, because these changes can easily be mistaken for infectious disease or recurrent tumors.




Similar content being viewed by others
References
Dorner L, Ulmer S, Rohr A, Mehdorn HM, Nabavi A (2011) Space-occupying cyst development in the resection cavity of malignant gliomas following Gliadel(R) implantation–incidence, therapeutic strategies, and outcome. J Clin Neurosci 18(3):347–351. doi:10.1016/j.jocn.2010.05.036
Engelhard HH (2000) Tumor bed cyst formation after BCNU wafer implantation: report of two cases. Surg Neurol 53(3):220–224
McGirt MJ, Villavicencio AT, Bulsara KR, Friedman HS, Friedman AH (2002) Management of tumor bed cysts after chemotherapeutic wafer implantation. Report of four cases. J Neurosurg 96(5):941–945. doi:10.3171/jns.2002.96.5.0941
Sabel M, Giese A (2008) Safety profile of carmustine wafers in malignant glioma: a review of controlled trials and a decade of clinical experience. Curr Med Res Opin 24(11):3239–3257. doi:10.1185/03007990802508180
Ulmer S, Spalek K, Nabavi A, Schultka S, Mehdorn HM, Kesari S, Dorner L (2012) Temporal changes in magnetic resonance imaging characteristics of Gliadel wafers and of the adjacent brain parenchyma. Neuro-oncology 14(4):482–490. doi:10.1093/neuonc/nos003
Prager JM, Grenier Y, Cozzens JW, Chiowanich P, Gorey MT, Meyer JR (2000) Serial CT and MR imaging of carmustine wafers. Am J Neuroradiol 21(1):119–123
Colen RR, Zinn PO, Hazany S, Do-Dai D, Wu JK, Yao K, Zhu JJ (2011) Magnetic resonance imaging appearance and changes on intracavitary Gliadel wafer placement: a pilot study. World J Radiol 3(11):266–272. doi:10.4329/wjr.v3.i11.266
Hammoud DA, Belden CJ, Ho AC, Dal Pan GJ, Herskovits EH, Hilt DC, Brem H, Pomper MG (2003) The surgical bed after BCNU polymer wafer placement for recurrent glioma: serial assessment on CT and MR imaging. Am J Roentgenol 180(5):1469–1475. doi:10.2214/ajr.180.5.1801469
Fleming AB, Saltzman WM (2002) Pharmacokinetics of the carmustine implant. Clin Pharmacokinet 41(6):403–419. doi:10.2165/00003088-200241060-00002
Fink J, Born D, Chamberlain MC (2011) Pseudoprogression: relevance with respect to treatment of high-grade gliomas. Curr Treat Options Oncol 12(3):240–252. doi:10.1007/s11864-011-0157-1
Shah R, Vattoth S, Jacob R, Manzil FF, O’Malley JP, Borghei P, Patel BN, Cure JK (2012) Radiation necrosis in the brain: imaging features and differentiation from tumor recurrence. Radiographics 32(5):1343–1359. doi:10.1148/rg.325125002
Aoki T, Nishikawa R, Sugiyama K, Nonoguchi N, Kawabata N, Mishima K, Adachi J, Kurisu K, Yamasaki F, Tominaga T, Kumabe T, Ueki K, Higuchi F, Yamamoto T, Ishikawa E, Takeshima H, Yamashita S, Arita K, Hirano H, Yamada S, Matsutani M, group NPCs (2014) A multicenter phase I/II study of the BCNU implant (Gliadel((R)) Wafer) for Japanese patients with malignant gliomas. Neurol Med Chir 54(4):290–301
Westphal M, Hilt DC, Bortey E, Delavault P, Olivares R, Warnke PC, Whittle IR, Jaaskelainen J, Ram Z (2003) A phase 3 trial of local chemotherapy with biodegradable carmustine (BCNU) wafers (Gliadel wafers) in patients with primary malignant glioma. Neuro-oncology 5(2):79–88. doi:10.1215/s1522-8517-02-00023-6
Della Puppa A, Rossetto M, Ciccarino P, Del Moro G, Rotilio A, Manara R, Paola Gardiman M, Denaro L, d’Avella D, Scienza R (2010) The first 3 months after BCNU wafers implantation in high-grade glioma patients: clinical and radiological considerations on a clinical series. Acta Neurochir 152(11):1923–1931. doi:10.1007/s00701-010-0759-6
Bock HC, Puchner MJ, Lohmann F, Schutze M, Koll S, Ketter R, Buchalla R, Rainov N, Kantelhardt SR, Rohde V, Giese A (2010) First-line treatment of malignant glioma with carmustine implants followed by concomitant radiochemotherapy: a multicenter experience. Neurosurg Rev 33(4):441–449. doi:10.1007/s10143-010-0280-7
McGirt MJ, Than KD, Weingart JD, Chaichana KL, Attenello FJ, Olivi A, Laterra J, Kleinberg LR, Grossman SA, Brem H, Quinones-Hinojosa A (2009) Gliadel (BCNU) wafer plus concomitant temozolomide therapy after primary resection of glioblastoma multiforme. J Neurosurg 110(3):583–588. doi:10.3171/2008.5.17557
Compliance with Ethical Standards
The authors declare that they have no conflict of interest. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ohue, S., Kohno, S., Inoue, A. et al. Evaluation of serial changes on computed tomography and magnetic resonance imaging after implantation of carmustine wafers in patients with malignant gliomas for differential diagnosis of tumor recurrence. J Neurooncol 126, 119–126 (2016). https://doi.org/10.1007/s11060-015-1941-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-015-1941-8