Abstract
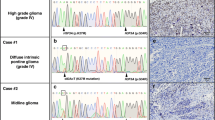
Pediatric glioblastomas recently have been exon sequenced with evidence that approximately 30 % of cases harbour mutations of the histone H3.3 gene. Although studies to determinate their role in risk stratification are on-going, it remains to be determined whether H3.3 mutations could be found in other tumors such as pediatric primitive neuroectodermal tumors of the central nervous system (CNS-PNETs) and whether the presence of H3.3 mutations in glioblastomas could be used as diagnostic tool in their differential diagnosis with CNS-PNETs. We performed a large mutational pyrosequencing-based screening on 123 pediatric glioblastomas and 33 CNS-PNET. The analysis revealed that 39/123 (31.7 %) glioblastomas carry H3.3 mutations. The K27M (AAG → ATG, lysine → methionine) mutation was found in 33 glioblastomas (26 %); the G34R (GGG → AGG, glycine → arginine) was observed in 6 glioblastomas (5.5 %). However, we also identified 4 of 33 cases (11 %) of CNS-PNETs harbouring a H3.3 G34R mutation. Multiplex ligation-dependent probe amplification analysis revealed PDGFR-alpha amplification and EGFR gain in two cases and N-Myc amplification in one case of H3.3 G34R mutated CNS-PNET. None of H3.3 mutated tumors presented a CDKN2A loss. In conclusion, because pediatric patients with glioblastoma and CNS-PNET are treated according to different therapeutic protocols, these findings may raise further concerns about the reliability of the histological diagnosis in the case of an undifferentiated brain tumor harbouring G34R H3.3 mutation. In this view, additional studies are needed to determine whether H3.3 G34 mutated CNS-PNET/glioblastomas may represent a defined tumor subtype.

Similar content being viewed by others
References
McLendon RE, Judkins AR, Eberhart CG, Fuller GN, Sarkar C, Ng HK (2007) Central nervous system primitive neuroectodermal tumours. In: Louis DN, Ohgaki H, Wiestler OD, Cavenee WK (eds) WHO classification of tumours of the central nervous system. IARC, Lyon, pp 141–146
Burger PC (2006) Supratentorial primitive neuroectodermal tumor (sPNET). Brain Pathol 16:86
Pomeroy SL, Tamayo P, Gaasenbeek M et al (2002) Prediction of central nervous system embryonal tumour outcome based on gene expression. Nature 415:436–442
Rheinbay E, Louis DN, Bernstein BE, Suvà ML (2012) A tell-tail sign of chromatin: histone mutations drive pediatric glioblastoma. Cancer Cell 21:329–331
Schwartzentruber J, Korshunov A, Liu XY (2012) Driver mutations in histone H3.3 and chromatin remodelling genes in paediatric glioblastoma. Nature 482:226–231
Wu A, Broniscer TA, McEachron C et al (2012) Somatic histone H3 alterations in pediatric diffuse intrinsic pontine gliomas and non-brainstem glioblastomas. Nat Genet 44:251–253
Talbert PB, Henikoff S (2010) Histone variants–ancient wrap artists of the epigenome. Nat Rev Mol Cell Biol 11:264–275
Simon JA, Kingston RE (2009) Mechanisms of polycomb gene silencing: knowns and unknowns. Nat Rev Mol Cell Biol 10:697–708
Zhou VW, Goren A, Bernstein BE (2011) Charting histone modifications and the functional organization of mammalian genomes. Nat Rev Genet 12:7–18
Khuong-Quang DA, Buczkowicz P, Rakopoulos P et al (2012) K27M mutation in histone H3.3 defines clinically and biologically distinct subgroups of pediatric diffuse intrinsic pontine gliomas. Acta Neuropathol 124:439–447
Hömig-Hölzel C, Savola S (2012) Multiplex ligation-dependent probe amplification (MLPA) in tumor diagnostics and prognostics. Diagn Mol Pathol 21:189–206
Wang Y, Cottman M, Schiffman JD (2012) Molecular inversion probes: a novel microarray technology and its application in cancer research. Cancer Genet 205:341–355
Kraus JA, Felsberg J, Tonn JC, Reifenberger G, Pietsch T (2002) Molecular genetic analysis of the TP53, PTEN, CDKN2A, EGFR, CDK4 and MDM2 tumour-associated genes in supratentorial primitive neuroectodermal tumours and glioblastomas of childhood. Neuropathol Appl Neurobiol 28:325–333
Hayden JT, Frühwald MC, Hasselblatt M et al (2009) Frequent IDH1 mutations in supratentorial primitive neuroectodermal tumors (sPNET) of adults but not children. Cell Cycle 8:1806–1807
Behdad A, Perry A (2010) Central nervous system primitive neuroectodermal tumors: a clinicopathologic and genetic study of 33 cases. Brain Pathol 20:441–450
Pfister S, Remke M, Toedt G et al (2007) Supratentorial primitive neuroectodermal tumors of the central nervous system frequently harbor deletions of the CDKN2A locus and other genomic aberrations distinct from medulloblastomas. Genes Chromosomes Cancer 46:839–851
Inda MM, Castresana JS (2007) RASSF1A promoter is highly methylated in primitive neuroectodermal tumors of the central nervous system. Neuropathology 27:341–346
Inda MM, Munoz J, Coullin P et al (2006) High promoter hypermethylation frequency of p14/ARF in supratentorial PNET but not in medulloblastoma. Histopathology 48:579–587
Picard D, Miller S, Hawkins CE et al (2012) Markers of survival and metastatic potential in childhood CNS primitive neuro-ectodermal brain tumours: an integrative genomic analysis. Lancet Oncol 3:838–848
Broniscer A, Gajjar A (2004) Supratentorial high-grade astrocytoma and diffuse brainstem glioma: two challenges for the pediatric oncologist. Oncologist 9:197–206
Perry A, Miller CR, Gujrati M et al (2009) Malignant gliomas with primitive neuroectodermal tumor-like components: a clinicopathologic and genetic study of 53 cases. Brain Pathol 19:81–90
Paugh BS, Qu C, Jones C et al (2010) Molecular genetic profiling of pediatric high-grade gliomas reveals key differences with the adult disease. J Clin Oncol 28(18):3061–3068
Giangaspero F, Eberhart CG, Haapasalo HH et al (2007) Medulloblastoma. In: Louis DN, Ohgaki H, Wiestler OD, Cavenee WK (eds) WHO classification of tumours of the central nervous system. IARC, Lyon, pp 132–140
Gielen GH, Gessi M, Hammes J et al (2013) H3F3A K27M mutation in pediatric CNS tumors: a marker for diffuse high-grade astrocytomas. Am J Clin Pathol, in press
Ligon KL, Alberta JA, Kho AT et al (2004) The oligodendroglial lineage marker OLIG2 is universally expressed in diffuse gliomas. J Neuropathol Exp Neurol 63:499–509
Conflict of interest
All authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Marco Gessi and Gerrit H. Gielen contributed equally to this work.
Andreas Waha and Torsten Pietsch share senior authorship.
Rights and permissions
About this article
Cite this article
Gessi, M., Gielen, G.H., Hammes, J. et al. H3.3 G34R mutations in pediatric primitive neuroectodermal tumors of central nervous system (CNS-PNET) and pediatric glioblastomas: possible diagnostic and therapeutic implications?. J Neurooncol 112, 67–72 (2013). https://doi.org/10.1007/s11060-012-1040-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-012-1040-z