Abstract
Cathepsin B and urokinase plasminogen activator receptor (uPAR) are postulated to play key roles in glioma invasion. Calcineurin is one of the key regulators of mitochondrial-dependent apoptosis, but its mechanism is poorly understood. Hence, we studied subcellular localization of calcineurin after transcriptional downregulation of uPAR and cathepsin B in glioma. In the present study, efficient downregulation of uPAR and cathepsin B increased the translocation of calcineurin A from the mitochondria to the cytosol, decreased pBAD (S136) expression and its interaction with 14-3-3ζ and increased the interaction of BAD with Bcl-xl. Co-depletion of uPAR and cathepsin B induced mitochondrial translocation of BAD, activation of caspase 3 as well as PARP and cytochrome c and SMAC release. These effects were inhibited by FK506 (10 μM), a specific inhibitor of calcineurin. Calcineurin A was co-localized and also co-immunoprecipitated with Bcl-2. This interaction decreased with co-depletion of uPAR and cathepsin B and also with Bcl-2 inhibitor, HA 14-1 (20 μg/ml). Altered localization and interaction of calcineurin A with Bcl-2 was also observed in vivo when uPAR and cathepsin B were downregulated. In conclusion, downregulation of uPAR and cathepsin B induced apoptosis by targeting calcineurin A to BAD via Bcl-2 in glioma.






Similar content being viewed by others
Change history
12 March 2021
A Correction to this paper has been published: https://doi.org/10.1007/s11060-021-03722-w
References
Malla R, Gopinath S, Alapati K, Gondi CS, Gujrati M, Dinh DH, Mohanam S, Rao JS (2010) Downregulation of uPAR and cathepsin B induces apoptosis via regulation of Bcl-2 and Bax and inhibition of the PI3K/Akt pathway in gliomas. PLoS One 5:e13731
Klee CB, Draetta GF, Hubbard MJ (1988) Calcineurin. Adv Enzymol Relat Areas Mol Biol 61:149–200
Stemmer PM, Klee CB (1994) Dual calcium ion regulation of calcineurin by calmodulin and calcineurin B. Biochemistry 33:6859–6866
Hubbard MJ, Klee CB (1989) Functional domain structure of calcineurin A: mapping by limited proteolysis. Biochemistry 28:1868–1874
Kissinger CR, Parge HE, Knighton DR, Lewis CT, Pelletier LA, Tempczyk A, Kalish VJ, Tucker KD, Showalter RE, Moomaw EW (1995) Crystal structures of human calcineurin and the human FKBP12-FK506-calcineurin complex. Nature 378:641–644
Wang X, Culotta VC, Klee CB (1996) Superoxide dismutase protects calcineurin from inactivation. Nature 383:434–437
Erin N, Bronson SK, Billingsley ML (2003) Calcium-dependent interaction of calcineurin with Bcl-2 in neuronal tissue. Neuroscience 117:541–555
Wang HG, Pathan N, Ethell IM, Krajewski S, Yamaguchi Y, Shibasaki F, McKeon F, Bobo T, Franke TF, Reed JC (1999) Ca2+-induced apoptosis through calcineurin dephosphorylation of BAD. Science 284:339–343
Martin GK, Ohlms LA, Franklin DJ, Harris FP, Lonsbury-Martin BL (1990) Distortion product emissions in humans. III. Influence of sensorineural hearing loss. Ann Otol Rhinol Laryngol Suppl 147:30–42
Fruman DA, Mather PE, Burakoff SJ, Bierer BE (1992) Correlation of calcineurin phosphatase activity and programmed cell death in murine T cell hybridomas. Eur J Immunol 22:2513–2517
Zhao Y, Tozawa Y, Iseki R, Mukai M, Iwata M (1995) Calcineurin activation protects T cells from glucocorticoid-induced apoptosis. J Immunol 154:6346–6354
Lobo FM, Zanjani R, Ho N, Chatila TA, Fuleihan RL (1999) Calcium-dependent activation of TNF family gene expression by Ca2+/calmodulin kinase type IV/Gr and calcineurin. J Immunol 162:2057–2063
Toth R, Szegezdi E, Molnar G, Lord JM, Fesus L, Szondy Z (1999) Regulation of cell surface expression of Fas (CD95) ligand and susceptibility to Fas (CD95)-mediated apoptosis in activation-induced T cell death involves calcineurin and protein kinase C, respectively. Eur J Immunol 29:383–393
Shibasaki F, McKeon F (1995) Calcineurin functions in Ca(2+)-activated cell death in mammalian cells. J Cell Biol 131:735–743
Park CH, Kim YS, Kim YH, Choi MY, Yoo JM, Kang SS, Choi WS, Cho GJ (2008) Calcineurin mediates AKT dephosphorylation in the ischemic rat retina. Brain Res 1234:148–157
Asai A, Qiu J, Narita Y, Chi S, Saito N, Shinoura N, Hamada H, Kuchino Y, Kirino T (1999) High level calcineurin activity predisposes neuronal cells to apoptosis. J Biol Chem 274:34450–34458
Gondi CS, Kandhukuri N, Kondraganti S, Gujrati M, Olivero WC, Dinh DH, Rao JS (2006) RNA interference-mediated simultaneous down-regulation of urokinase-type plasminogen activator receptor and cathepsin B induces caspase-8-mediated apoptosis in SNB19 human glioma cells. Mol Cancer Ther 5:3197–3208
Gondi CS, Lakka SS, Dinh DH, Olivero WC, Gujrati M, Rao JS (2007) Intraperitoneal injection of an hpRNA-expressing plasmid targeting uPAR and uPA retards angiogenesis and inhibits intracranial tumor growth in nude mice. Clin Cancer Res 13:4051–4060
Lakka SS, Gondi CS, Yanamandra N, Olivero WC, Dinh DH, Gujrati M, Rao JS (2004) Inhibition of cathepsin B and MMP-9 gene expression in glioblastoma cell line via RNA interference reduces tumor cell invasion, tumor growth and angiogenesis. Oncogene 23:4681–4689
Gopinath S, Malla RR, Gondi CS, Alapati K, Fassett D, Klopfenstein JD, Dinh DH, Gujrati M, Rao JS (2010) Co-depletion of cathepsin B and uPAR induces G0/G1 arrest in glioma via FOXO3a mediated p27 upregulation. PLoS One 5:e11668
Shibasaki F, Kondo E, Akagi T, McKeon F (1997) Suppression of signalling through transcription factor NF-AT by interactions between calcineurin and Bcl-2. Nature 386:728–731
Aguirre-Ghiso JA, Alonso DF, Farias EF, Gomez DE, Kier Joffe EB (1999) Deregulation of the signaling pathways controlling urokinase production. Its relationship with the invasive phenotype. Eur J Biochem 263:295–304
Mohanam S, Sawaya R, McCutcheon I, Ali-Osman F, Boyd D, Rao JS (1993) Modulation of in vitro invasion of human glioblastoma cells by urokinase-type plasminogen activator receptor antibody. Cancer Res 53:4143–4147
Sivaparvathi M, Sawaya R, Wang SW, Rayford A, Yamamoto M, Liotta LA, Nicolson GL, Rao JS (1995) Overexpression and localization of cathepsin B during the progression of human gliomas. Clin Exp Metastasis 13:49–56
Reed JC (1997) Double identity for proteins of the Bcl-2 family. Nature 387:773–776
del Peso L, Gonzalez-Garcia M, Page C, Herrera R, Nunez G (1997) Interleukin-3-induced phosphorylation of BAD through the protein kinase Akt. Science 278:687–689
Scheid MP, Duronio V (1998) Dissociation of cytokine-induced phosphorylation of Bad and activation of PKB/akt: involvement of MEK upstream of Bad phosphorylation. Proc Natl Acad Sci USA 95:7439–7444
Zha J, Harada H, Yang E, Jockel J, Korsmeyer SJ (1996) Serine phosphorylation of death agonist BAD in response to survival factor results in binding to 14-3-3 not BCL-X(L). Cell 87:619–628
Masters SC, Yang H, Datta SR, Greenberg ME, Fu H (2001) 14-3-3 inhibits Bad-induced cell death through interaction with serine-136. Mol Pharmacol 60:1325–1331
Danial NN (2008) BAD: undertaker by night, candyman by day. Oncogene 27 Suppl 1:S53–S70
Chiang CW, Kanies C, Kim KW, Fang WB, Parkhurst C, Xie M, Henry T, Yang E (2003) Protein phosphatase 2A dephosphorylation of phosphoserine 112 plays the gatekeeper role for BAD-mediated apoptosis. Mol Cell Biol 23:6350–6362
Jacobsson S, Jonsson L, Rank F, Rothman U (1976) Studies on healing of Debrisan-treated wounds. Scand J Plast Reconstr Surg 10:97–101
Hortelano S, Lopez-Collazo E, Bosca L (1999) Protective effect of cyclosporin A and FK506 from nitric oxide-dependent apoptosis in activated macrophages. Br J Pharmacol 126:1139–1146
Yardin C, Terro F, Lesort M, Esclaire F, Hugon J (1998) FK506 antagonizes apoptosis and c-jun protein expression in neuronal cultures. Neuroreport 9:2077–2080
Takadera T, Sakamoto Y, Hizume Y, Ohyashiki T (2007) Cyclosporine A- and FK506-induced apoptosis in PC12 cells. Cell Biol Toxicol 23:355–360
Colledge M, Scott JD (1999) AKAPs: from structure to function. Trends Cell Biol 9:216–221
Ayllon V, Cayla X, Garcia A, Roncal F, Fernandez R, Albar JP, Martinez C, Rebollo A (2001) Bcl-2 targets protein phosphatase 1 alpha to Bad. J Immunol 166:7345–7352
Rong YP, Barr P, Yee VC, Distelhorst CW (2009) Targeting Bcl-2 based on the interaction of its BH4 domain with the inositol 1,4,5-trisphosphate receptor. Biochim Biophys Acta 1793:971–978
Gondi CS, Lakka SS, Dinh D, Olivero W, Gujrati M, Rao JS (2004) Downregulation of uPA, uPAR and MMP-9 using small, interfering, hairpin RNA (siRNA) inhibits glioma cell invasion, angiogenesis and tumor growth. Neuron Glia Biol 1:165–176
Acknowledgment
This research was supported by National Institutes of Health, CA116708 (to JSR). Contents of this manuscript are solely the responsibility of the authors and do not necessarily represent the official views of NIH.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
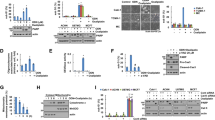
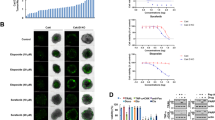
Localization of calcineurin A and BAD in U251 and 4910 cells. A. Co-localization of calcineurin A and MTCO2 in glioma cells. After overnight growth, cells were fixed and incubated with calcineurin A and MTCO2 antibodies followed by incubation with species-specific Alexa Fluor secondary antibodies. B. Expression of BAD in cytosolic and mitochondrial fractions from U251 and 4910 cell lysates after transfection with plasmids expressing full-length uPAR and cathepsin B or scrambled vector for 48 hrs. After transfection, whole cell lysates (WL) were collected and equal amounts of protein were immunoblotted for uPAR, cathepsin B and BAD. Cytosolic (CF) and mitochondrial (MF) fractions were immunoblotted for BAD.
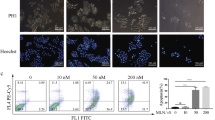
Effect of pCU on co-localization of BAD with 14-3-3 or Bcl-xl and mitochondrial membrane potential in U251 and 4910 cells. A. Immunofluorescence assay was used to detect the co-localization of pBAD (S136) and 14-3-3ζ in pSV- and pCUtreated= U251 and 4910 cells. 72 hrs after treatment, cells were fixed and incubated with pBAD (S136) and 14-3-3ζ antibodies followed by incubation with species-specific Alexa Fluor secondary antibodies. B. Co-localization of BAD and Bcl-xl in pSV- and pCU-treated cells. C. MitoLight dye-based quantification of pCU-induced loss of mitochondrial membrane potential in the presence or absence of FK506 in glioma cells. Flow cytometry analysis with MitoLight dye was carried out in pCU-treated cells in the presence or absence of FK506. The percentage of MitoLight-positive cells (emitting red fluorescence) read from the M1 marker and negative cells from the M2 marker. The position of M1 marker was adjusted according to the results of the negative control (cells without dye).
Interaction of calcineurin A and Bcl-2 in uPAR and cathepsin B-overexpressed glioma cells. A. Expression of Bcl-2 in pCU-treated U251 and 4910 cells. B. Immunoprecipitation of Bcl-2 in uPAR and cathepsin B-overexpressed glioma cells. Total cell lysates from U251 and 4910 cells transfected with plasmids expressing full-length uPAR and cathepsin B were immunoprecipitated with Bcl-2 and then immunoblotted for Bcl-2 and calcineurin A.
About this article
Cite this article
Malla, R.R., Gopinath, S., Gondi, C.S. et al. RETRACTED ARTICLE: uPAR and cathepsin B downregulation induces apoptosis by targeting calcineurin A to BAD via Bcl-2 in glioma. J Neurooncol 107, 69–80 (2012). https://doi.org/10.1007/s11060-011-0727-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-011-0727-x