Abstract
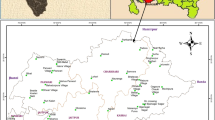
Salinity and sodicity of groundwater are the principal water quality concerns in irrigated areas of arid and semi-arid regions. The hydrochemical characteristics and sodicity of groundwater in the Shirin Sou area, western Iran were investigated in this study by chemical analyses of groundwater samples from 49 wells. Chemical analysis of the groundwater showed that the mean concentration of the cations was in the order: Na+ > Ca2+ > Mg2+ > K+, while that for anions was SO3 2− > Cl− > HCO3 − > NO3 −. The most prevalent water type is Na–SO4 followed by water types Na–Cl and Ca–SO4. The chemical evolution of groundwater is primarily controlled by water–rock interactions: mainly weathering of aluminosilicates, dissolution of sulfate minerals, and cation exchange reactions. Sulfate dissolution and pyrite weathering may both contribute to the SO4 2− load of the groundwater. High Na+ concentrations in groundwater participate in ion-exchange processes, resulting in the displacement of base cations into solution and raised concentrations in groundwater. The principal component analysis (PCA) performed on groundwater identified three principal components controlling variability of groundwater chemistry. Electrical conductivity, Ca2+, Mg2+, Na+, SO4 2−, and Cl− content were associated in the same component (PC1) (salinity), most likely linked to anthropogenic activities.








Similar content being viewed by others
References
Appelo, G. A. J., & Postma, D. (1994). Geochemistry, groundwater and pollution. Rotterdam: A.A. Balkema.
Ayars, J. E., & Tanji, K. K. (1999). Effects of drainage on water quality in arid and semiarid lands. In R. W. Skaggs & J. van Schilfgaarde (Eds.), Agricultural drainage (pp. 831–867). Madison, WI: ASA-CSSA-SSSA.
Baharifar, A., Moinevaziri, H., Bellon, H., & Pique, A. (2004). The crystalline complexes of Hamedan (Sanandaj-Sirjan zone, western Iran): metasedimentary Mezoic sequences affected by Late Cretaceous tectono-metamorphic and plutonic events. Comptes Rendus Geoscience, 336, 1443–1452.
Beltran, J. M. (1999). Irrigation with saline water: benefits and environmental impact. Agricultural Water Management, 40, 183–194.
Dalai, T. K., Krishnaswami, S., & Sarin, M. M. (2002). Barium in the Yamuna River System in the Himalaya: Sources, fluxes, and its behavior during weathering and transport. Geochemistry Geophysics Geosystems, 3, 1076.
Di, H. J., & Cameron, K. C. (2004). Effects of the nitrification inhibitor dicyandiamide on potassium, magnesium and calcium leaching in grazed grassland. Soil Use Management, 20, 2–7.
Dragon, K. (2006). Application of factor analysis to study contamination of a semi-confined aquifer (Wielkopolska Buried Valley aquifer, Poland). Journal of Hydrology, 331, 272–279.
Drever, J. I. (1997). The geochemistry of natural waters (3rd ed.). Upper Saddle River, NJ: Prentice Hall.
Frapporti, G., Hoogendoorn, J. H., & Vriend, S. P. (1995). Detailed hydrochemical studies as a useful extension of national groundwater monitoring networks. Groundwater, 33, 817–828.
Freeze, R. A., & Cherry, J. A. (1979). Groundwater (p. 604). New Jersey: Prentice-Hall.
Garcia, M. G., Hidalgo, M. D., & Blessa, M. A. (2001). Geochemistry of groundwater in the alluvial plain of Tucuman Province, Argentina. Hydrogeology Journal, 9, 597–610.
Gibbs, R. J. (1970). Mechanism controlling world water chemistry. Science, 170, 1088–1090.
Gupta, L. P., & Subramanian, V. (1998). Geochemical factors controlling the chemical nature of water and sediments in the Gomti River, India. Environmental Geology, 36, 102–108.
Hao, X., & Change, C. (2002). Does long-term heavy cattle manure application increase salinity of a clay loam soil in semi-arid southern Alberta? Agriculture, Ecosystems & Environment, 1934, 1–16.
Herczeg, A. (2001). Can major ion chemistry be used estimate groundwater residence time in basaltic aquifer. In R. Cidu (Ed.), Proceeding of the ninth international symposium water–rock interaction (pp. 529–532). Rotterdam: A.A. Balkema.
Hidalgo, M. C., & Cruz-Sanjulian, J. (2001). Groundwater composition, hydrochemical evolution and mass transfer in a regional detrital aquifer (Baza Basin, southern Spain). Applied Geochemistry, 16, 745–758.
Jalali, M. (2005a). Nitrates leaching from agricultural land in Hamedan, western Iran. Agriculture, Ecosystems & Environment, 110, 210–218.
Jalali, M. (2005b). Major ion chemistry in the Bahar area, Hamedan, western Iran. Environmental Geology, 47, 763–772.
Jalali, M. (2006). Chemical characteristics of groundwater in parts of mountainous region, Alvand, Hamedan, Iran. Environmental Geology, 51, 433–446.
Jalali, M. (2007). Salinization of groundwater in arid and semi-arid zones: an example from Tajarak, western Iran. Environmental Geology, 52, 1133–1149.
Jalali, M., & Khanlari, Z. V. (2008). Major ion chemistry of groundwaters in the Damagh area, Hamedan, western Iran. Environmental Geology, 54, 87–93.
Jalali, M., Merikhpour, H., Kaledhonkar, M. J., & Van Der Zee, S. E. A. T. M. (2008). Effects of wastewater irrigation on soil sodicity and nutrient leaching in calcareous soils. Agricultural Water Management, 95, 143–153.
Jalali, M., & Merrikhpour, H. (2008). Effects of poor quality irrigation waters on the nutrient leaching and groundwater quality from sandy soil. Environmental Geology, 53, 1289–1298.
Jalali, M., & Rowell, D. L. (2003). The role of calcite and gypsum in the leaching of potassium in a sandy soil. Experimental Agriculture, 39, 379–394.
Jalali, M., & Rowell, D. L. (2008). Prediction leaching of potassium using the convective-dispersive and the convective log-normal transfer function models. Environmental Geology, 55, 863–874.
Kolahchi, Z., & Jalali, M. (2007). Effect of water quality on the leaching of potassium from sandy soil. Journal of Arid Environments, 68, 624–639.
McLean, W., Jankowski, J., & Lavitt, N. (2000). Groundwater quality and sustainability in an alluvial aquifer, Australia. In O. Sililo, et al. (Eds.), Groundwater, past achievements and future challenges (pp. 567–573). Rotterdam: A.A. Balkema.
Meybeck, M. (1987). Global chemical weathering of surficial rocks estimated from river dissolved loads. American Journal of Science, 287, 401–428.
Miller, N. J., & Miller, J. C. (2000). Statistics and chemometrics for analytical chemistry (4th ed.). Englewood Cliffs, NJ: Pearson Education.
Njitchoua, R., Dever, L., Fontes, J. Ch., & Naah, E. (1997). Geochemistry, origin and recharge mechanisms of groundwaters from the Garoua Sandstone aquifer, northern Cameroom. Journal of Hydrology, 190, 123–140.
Oetting, G. C., Banner, J. L., & Sharp, J. M. (1996). Regional controls on the geochemical evolution of saline groundwaters in the Edward aquifers, Central Texas. Journal of Hydrology, 181, 251–283.
Parkhurst, D. L., & Appelo, C. A. J. (1999). Users guide to PHREEQC (version 2)—A computer program for speciation, batch-reaction, one-dimensional transport, and inverse geochemical calculations: U.S. Geological Survey Water Resources Investigations Report 99-4259.
Richter, B. C., & Kreitler, W. C. (1993). Geochemical techniques for identifying sources of groundwater salinization. New York: CRC Press. ISBN 1-56670-000-0.
Ronen, D., Kanfi, Y., & Magaritz, M. (1983). Sources of nitrates in groundwater of the Coastal Plain of Israel- evolution of ideas. Water Research, 17, 1499–1503.
Rowell, D. L. (1994). Soil science: methods and applications. Harlow: Longman and Scientific Technical.
Saleh, A., Al-Ruwiah, F. M., & Shehata, M. (2001). Ground-water quality of the Nile west bank related to soil characteristics and geological setting. Journal of Arid Environments, 49, 761–784.
Sami, K. (1992). Recharge mechanisms and geochemical processes in a semi-arid sedimentary basin, Eastern Cape, South Africa. Journal of Hydrology, 139, 27–48.
Sepahi, A. (1999). Petrology of the Alvand plutonic complex with special reference on granitoids. Ph.D. thesis, Tarbiat-Moallem University, Tehran, Iran (in Persian).
Sharpley, A. N., & Smith, S. J. (1989). Prediction of soluble phosphorus transport in agricultural runoff. Journal of Environment Quality, 18, 313–316.
Škrbić, B., & Čupić, S. (2004). Trace metal distribution in surface soils of Novi Sad and Bank Sediment of the Danube River. Journal of Environmental Science and Health, Part A, 39, 1547–1558.
Smolders, A. J. P., Hudson-Edwards, K. A., Van der Velde, G., & Roelofs, J. G. M. (2004). Controls on water chemistry of the Pilcomayo River (Bolivia, South-America). Applied Geochemistry, 19, 1745–1758.
Stallard, R. F., & Edmond, J. M. (1987). Geochemistry of the Amazon 3 Weathering chemistry and limits to dissolved inputs. Journal of Geophysical Research, 92, 8293–8302.
Stimson, J., Frape, S., Drimmie, R., & Rudolph, D. (2001). Isotopic and geochemical evidence of regional-scale anisotropy and interconnectivity of an alluvial fan system, Cochabamba Valley, Bolivia. Applied Geochemistry, 16, 1097–1114.
Umar, A., Umar, R., & Ahmad, M. S. (2001). Hydrogeological and hydrochemical framework of regional aquifer system in Kali-Ganga sub-basin, India. Environmental Geology, 40(4–5), 602–611.
Wilcox, L. V. (1955). Classification and use of irrigation waters. Washington, DC: US Dept. Agric. Circ.
Acknowledgments
The author is especially grateful to two anonymous reviewers for critical review, perceptive comments, and editing on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jalali, M. Hydrochemical Characteristics and Sodification of Groundwater in the Shirin Sou, Hamedan, Western Iran. Nat Resour Res 21, 61–73 (2012). https://doi.org/10.1007/s11053-011-9152-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11053-011-9152-2