Abstract
Methotrexate (MTX) is a well-known chemotherapeutic agent for solid tumor. However, its clinical usage is limited due to low permeability, cellular drug efflux, and non-specific drug delivery. These constraints require dose dumping and result in dose-dependent toxic effects. In the current study, MTX-loaded PEGylated gold nanoparticles (PEG-MTX-AuNPs) exposed to acidic pH (tumor cell) trigger a significant drug release profile. MTX was conjugated to citrate functionalized AuNP and stabilized by thiolated methyl polyethylene glycol (mPEG-SH). PEG-MTX-AuNP was cationic with 39.5 ± 1.2 mV zeta potential and a particle size of 39 nm, as revealed by DLS and TEM. AuNP size ranging from 40 to 50 nm was favorable for maximum endosomal uptake and fast drug release. MTX-AuNP showed 39.4% (22.28 µg/ml) drug loading efficiency. Spectroscopic examinations confirmed MTX chemisorption onto AuNPs via carboxyl (–COOH) and gold dative bond. In vitro release study indicated a 6.5-fold increased MTX release in the acidic environment (pH 4.5) compared to physiological pH. The terminal mPEG provided stability in a biological medium, refrained from protein deactivation, and exhibited significant hemocompatibility (less than 5% hemolysis up to 10 μM concentration). In vitro cytotoxicity against human rhabdomyosarcoma (RD) cells indicated significant (p < 0.0001) anticancer activity of PEG-MTX-AuNPs with low median growth inhibition concentration (GI50 = 3.61 ± 0.94 μM) compared to MTX (13.12 ± 0.98 μM) and AuNPs (5.24 ± 0.98 μM) alone. In conclusion, PEG-MTX-AuNPs offer higher stability in ionic and biological media, hemocompatibility, and acidic pH-dependent MTX release, and mediate enhanced in vitro anticancer activity that makes them a potential candidate for skeletal muscle sarcomas.
Graphic abstract
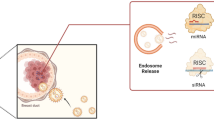
Methotrexate (MTX) is a weak dicarboxylic acid which become ionized at pH 7.4 (PBS) and unionized at endosomal pH 4.5. In ionized state MTX exchanges the negatively charged citrate ions and chemisorbed on AuNPs surface to stabilize and cap AuNPs. While in acidic media MTX become unionized and released in media. The enhanced intracellular release improves therapeutic outcome of MTX. The chemisorption of MTX was further confirmed with FTIR, supported by XRD and UV results.

Methotrexate binding mechanism to AuNP surface






Similar content being viewed by others
References
Ajmal M, Yunus U, Matin A, Haq NU (2015) Synthesis, characterization and in vitro evaluation of methotrexate conjugated fluorescent carbon nanoparticles as drug delivery system for human lung cancer targeting. J Photochem Photobiol B 153:111–120
Ao LM, Gao F, Pan BF, Cui DX, Gu HC (2006) Interaction between gold nanoparticles and bovine serum albumin or sheep antirabbit immunoglobulin G. Chin J Chem 24(2):253–256
Arakelova E, Grigoryan S, Arsenyan F, Babayan N, Grigoryan R, Sarkisyan N (2014) In vitro and in vivo anticancer activity of nanosize zinc oxide composites of doxorubicin. Int J Med Heal Pharm Biomed Eng 8:33–38
Aseichev A, Azizova O, Beckman E, Skotnikova O, Dudnik L, Shcheglovitova O, Sergienko V (2014) Effects of gold nanoparticles on erythrocyte hemolysis. Bull Exp Biol Med 156(4):495–498
Barabadi H, Vahidi H, Mahjoub MA, Kosar Z, Kamali KD, Ponmurugan K, Hosseini O, Rashedi M, Saravanan M (2019) Emerging antineoplastic gold nanomaterials for cervical cancer therapeutics: a systematic review. J Clust Sci 15:1–12
Barabadi H, Vahidi H, Kamali KD, Hosseini O, Mahjoub MA, Rashedi M, Shoushtari FJ, Saravanan M (2020) Emerging theranostic gold nanomaterials to combat lung cancer: a systematic review. J Clust Sci 31(2):323–330
Beidokhti HRN, Ghaffarzadegan R, Mirzakhanlouei S, Ghazizadeh L, Dorkoosh FA (2017) Preparation, characterization, and optimization of folic acid-chitosan-methotrexate core-shell nanoparticles by box-behnken design for tumor-targeted drug delivery. AAPS PharmSciTech 18(1):115–129
Bertino JR (2009) Cancer research: from folate antagonism to molecular targets. Best Pract Res Clin Haematol 22(4):577–582
Boomi P, Poorani GP, Palanisamy S, Selvam S, Ramanathan G, Ravikumar S, Barabadi H, Prabu HG, Jeyakanthan J, Saravanan M (2019) Evaluation of antibacterial and anticancer potential of polyaniline-bimetal nanocomposites synthesized from chemical reduction method. J Clust Sci 30(3):715–726
Caputo F, Clogston J, Calzolai L, Rösslein M, Prina-Mello A (2019) Measuring particle size distribution of nanoparticle enabled medicinal products, the joint view of EUNCL and NCI-NCL. A step by step approach combining orthogonal measurements with increasing complexity. J Controlled Release 299:31–43
Chen Y-H, Tsai C-Y, Huang P-Y, Chang M-Y, Cheng P-C, Chou C-H, Chen D-H, Wang C-R, Shiau A-L, Wu C-L (2007) Methotrexate conjugated to gold nanoparticles inhibits tumor growth in a syngeneic lung tumor model. Mol Pharm 4(5):713–722
Deol S, Weerasuriya N, Shon Y-S (2015) Stability, cytotoxicity and cell uptake of water-soluble dendron–conjugated gold nanoparticles with 3, 12 and 17 nm cores. J Mater Chem B 3(29):6071–6080
Dong YC, Hajfathalian M, Maidment PS, Hsu JC, Naha PC, Si-Mohamed S, Breuilly M, Kim J, Chhour P, Douek P (2019) Effect of gold nanoparticle size on their properties as contrast agents for computed tomography. Sci Rep 9(1):1–13
Du S, Kendall K, Toloueinia P, Mehrabadi Y, Gupta G, Newton J (2012) Aggregation and adhesion of gold nanoparticles in phosphate buffered saline. J Nanopart Res 14(3):758
Durgadas C, Sharma C, Paul W, Rekha M, Sreenivasan K (2012) Aggregation of gold nanoparticles followed by methotrexate release enables Raman imaging of drug delivery into cancer cells. J Nanopart Res 14(9):1127
Elahi N, Kamali M, Baghersad MH (2018) Recent biomedical applications of gold nanoparticles: a review. Talanta 184:537–556
Fan J, Hung W, Li W, Yeh J (2009) Biocompatibility study of gold nanoparticles to human cells. 13th international conference on biomedical engineering. Springer, pp 870–873
Fuliaş A, Popoiu C, Vlase G, Vlase T, Oneţiu D, Săvoiu G, Simu G, Pătruţescu C, Ilia G, Ledeţi I (2014) Thermoanalytical and spectroscopic study on methotrexate–active substance and tablet. Dig J Nanomater Biostruct 9(1):93–98
Gao W, Chan JM, Farokhzad OC (2010) pH-responsive nanoparticles for drug delivery. Mol Pharm 7(6):1913–1920
Gao J, Huang X, Liu H, Zan F, Ren J (2012) Colloidal stability of gold nanoparticles modified with thiol compounds: bioconjugation and application in cancer cell imaging. Langmuir 28(9):4464–4471
Ghorbani M, Zarei M, Mahmoodzadeh F, Ghorbani M (2020) Targeted delivery of methotrexate using a new PEGylated magnetic/gold nanoplatform covered with pH-responsive shell. Int J Polym Mater Polym Biomater 70:1–10
Guerrini L, Alvarez-Puebla R, Pazos-Perez N (2018) Surface modifications of nanoparticles for stability in biological fluids. Materials 11(7):1154
Held J, Mosheimer-Feistritzer B, Gruber J, Mur E, Weiss G (2018) Methotrexate therapy impacts on red cell distribution width and its predictive value for cardiovascular events in patients with rheumatoid arthritis. BMC Rheumatol 2(1):6
Holmboe L, Andersen AM, Mørkrid L, Slørdal L, Hall KS (2012) High dose methotrexate chemotherapy: pharmacokinetics, folate and toxicity in osteosarcoma patients. Br J Clin Pharmacol 73(1):106–114
Jia H, Gao X, Chen Z, Liu G, Zhang X, Yan H, Zhou H, Zheng L (2012) The high yield synthesis and characterization of gold nanoparticles with superior stability and their catalytic activity. CrystEngComm 14(22):7600–7606
Jokerst JV, Lobovkina T, Zare RN, Gambhir SS (2011) Nanoparticle PEGylation for imaging and therapy. Nanomedicine 6(4):715–728
Khatua A, Priyadarshini E, Rajamani P, Patel A, Kumar J, Naik A, Saravanan M, Barabadi H, Prasad A, Paul B (2020) Phytosynthesis, characterization and fungicidal potential of emerging gold nanoparticles using Pongamia pinnata leave extract: a novel approach in nanoparticle synthesis. J Clust Sci 31(1):125–131
Kimling J, Maier M, Okenve B, Kotaidis V, Ballot H, Plech A (2006) Turkevich method for gold nanoparticle synthesis revisited. J Phys Chem B 110(32):15700–15707
Krishnamurthy S, Esterle A, Sharma NC, Sahi SV (2014) Yucca-derived synthesis of gold nanomaterial and their catalytic potential. Nanoscale Res Lett 9(1):627–635
Kumari SDC, Tharani C, Narayanan N, Kumar CS (2013) Formulation and characterization of methotrexate loaded sodium alginate chitosan Nanoparticles. Indian J Res Pharm Biotechnol 1(6):915
Lin L, Xu W, Liang H, He L, Liu S, Li Y, Li B, Chen Y (2015) Construction of pH-sensitive lysozyme/pectin nanogel for tumor methotrexate delivery. Colloids Surf B 126:459–466
Liu X, Atwater M, Wang J, Huo Q (2007) Extinction coefficient of gold nanoparticles with different sizes and different capping ligands. Colloids Surf B 58(1):3–7
Madhusudhan A, Reddy GB, Venkatesham M, Veerabhadram G, Kumar DA, Natarajan S, Yang M-Y, Hu A, Singh SS (2014) Efficient pH dependent drug delivery to target cancer cells by gold nanoparticles capped with carboxymethyl chitosan. Int J Mol Sci 15(5):8216–8234
Manson J, Kumar D, Meenan BJ, Dixon D (2011) Polyethylene glycol functionalized gold nanoparticles: the influence of capping density on stability in various media. Gold Bull 44(2):99–105
Moore TL, Rodriguez-Lorenzo L, Hirsch V, Balog S, Urban D, Jud C, Rothen-Rutishauser B, Lattuada M, Petri-Fink A (2015) Nanoparticle colloidal stability in cell culture media and impact on cellular interactions. Chem Soc Rev 44(17):6287–6305
Muhammad Z, Raza A, Ghafoor S, Naeem A, Naz SS, Riaz S, Ahmed W, Rana NF (2016) PEG capped methotrexate silver nanoparticles for efficient anticancer activity and biocompatibility. Eur J Pharm Sci 91:251–255
Muthukumar T, Prabhavathi S, Chamundeeswari M, Sastry TP (2014) Bio-modified carbon nanoparticles loaded with methotrexate possible carrier for anticancer drug delivery. Mater Sci Eng C 36:14–19
Nadhman A, Nazir S, Khan MI, Ayub A, Muhammad B, Khan M, Shams DF, Yasinzai M (2015) Visible-light-responsive ZnCuO nanoparticles: benign photodynamic killers of infectious protozoans. Int J Nanomed 10:6891
Rahman S (2016) Size and concentration analysis of gold nanoparticles with ultraviolet-visible spectroscopy. Undergrad J Math Model 7(1):2
Rahme K, Nolan MT, Doody T, McGlacken GP, Morris MA, O’Driscoll C, Holmes JD (2013) Highly stable PEGylated gold nanoparticles in water: applications in biology and catalysis. RSC Adv 3(43):21016–21024. https://doi.org/10.1039/c3ra41873a
Saravanan M, Barabadi H, Ramachandran B, Venkatraman G, Ponmurugan K (2019) Emerging plant-based anti-cancer green nanomaterials in present scenario. In: Barcelo D, Petrovic B (eds) Comprehensive analytical chemistry. Elsevier, Amsterdam, pp 291–318
Saravanan M, Vahidi H, Cruz DM, Vernet-Crua A, Mostafavi E, Stelmach R, Webster TJ, Mahjoub MA, Rashedi M, Barabadi H (2020) Emerging antineoplastic biogenic gold nanomaterials for breast cancer therapeutics: a systematic review. Int J Nanomed 15:3577
Sengani M, Grumezescu AM, Rajeswari VD (2017) Recent trends and methodologies in gold nanoparticle synthesis–a prospective review on drug delivery aspect. OpenNano 2:37–46
Stapf M, Teichgräber U, Hilger I (2017) Methotrexate-coupled nanoparticles and magnetic nanochemothermia for the relapse-free treatment of T24 bladder tumors. Int J Nanomed 12:2793
Strober W (2015) Trypan blue exclusion test of cell viability. Curr Protoc Immunol. https://doi.org/10.1002/0471142735.ima03bs111
Tran NTT, Wang T-H, Lin C-Y, Tai Y (2013) Synthesis of methotrexate-conjugated gold nanoparticles with enhanced cancer therapeutic effect. Biochem Eng J 78:175–180
Woolley PV III, Sacher RA, Priego VM, Schanfield MS, Bonnem EM (1983) Methotrexate-induced immune haemolytic anaemia. Br J Haematol 54(4):543–552
Xie J, Fan Z, Li Y, Zhang Y, Yu F, Su G, Xie L, Hou Z (2018) Design of pH-sensitive methotrexate prodrug-targeted curcumin nanoparticles for efficient dual-drug delivery and combination cancer therapy. Int J Nanomed 13:1381
Acknowledgements
We are indebted to the NILOP Nanotheragnostic Research Laboratories of Pakistan Atomic Energy Commission for technical support. We are also thankful to Prof. Dr. N.M. Butt, Chairman, Preston Institute of Nano Science and Technology, for the provision of analytical support in UV-Spectrum analysis.
Funding
This work is funded by PAK-NORWAY Institutional Cooperation Program, PK3004.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rahman, M., Khan, J.A., Kanwal, U. et al. Methotrexate-loaded PEGylated gold nanoparticles as hemocompatible and pH-responsive anticancer drug nanoconjugate. J Nanopart Res 23, 195 (2021). https://doi.org/10.1007/s11051-021-05296-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-021-05296-0