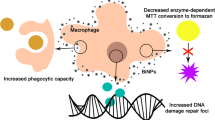
Abstract
Citrate-coated silver nanoparticles (citrate-AgNPs) are among the most commonly used nanomaterials, widely present in industrial and biomedical products. In this study, the cytotoxicity of 30-nm citrate-AgNPs on the macrophage cell line RAW 264.7 was evaluated, using a battery of cytotoxicity endpoints (viability, oxidative stress, and cytostaticity/clastogenicity), at 24 and 48 h of exposure. Citrate-AgNPs decreased cell proliferation and viability only at 75 μg/mL, suggesting a low sensitivity of RAW cells to lower doses of these AgNPs. After 24 h of exposure, ROS content decreased in cells exposed to 60 μg/mL AgNPs (IC20 value), corroborating the high tolerance of these cells to citrate-AgNPs. However, these cells suffered an impairment of the cell cycle, shown by an increase at the sub-G1 phase. This increase of the sub-G1 population was correlated with an increase of DNA fragmentation, suggesting an increase of apoptosis. Thus, our data are important to understand the effects of low concentrations (IC20) of citrate-AgNPs on in vitro vital macrophage functions.





Similar content being viewed by others
References
Abdelhalim MAK, Jarrar BM (2011) Renal tissue alterations were size-dependent with smaller ones induced more effects and related with time exposure of gold nanoparticles. Lipids Health Dis 10(1):1
Aldossari AA, Shannahan JH, Podila R (2015) Scavenger receptor B1 facilitates macrophage uptake of silver nanoparticles and cellular activation. J Nanopart Res 17(7):1–14
Arora S, Jain J, Rajwade JM, Paknikar KM (2008) Cellular responses induced by silver nanoparticles: in vitro studies. Toxicol Lett 179(2):93–100
AshaRani P, Low Kah Mun G, Hande M, Valiyaveettil S (2009) Cytotoxicity and genotoxicity of silver nanoparticles in human cells. ACS Nano 3(2):279–290
Avalos A, Haza AI, Mateo D, Morales P (2014) Cytotoxicity and ROS production of manufactured silver nanoparticles of different sizes in hepatoma and leukemia cells. J Appl Toxicol 34(4):413–423
Bancos S, Stevens DL, Tyner KM (2015) Effect of silica and gold nanoparticles on macrophage proliferation, activation markers, cytokine production, and phagocytosis in vitro. Int J Nanomedicine 10:183–206
Bastos V, de Oliveira JF, Brown D, Johnston H, Malheiro E, Daniel-da-Silva AL, Duarte IF, Santos C, Oliveira H (2016a) The influence of citrate or PEG coating on silver nanoparticle toxicity to a human keratinocyte cell line. Toxicol Lett 249:29–41
Bastos V, Ferreira-de-Oliveira JM, Carrola J, Daniel-da-Silva AL, Duarte IF, Santos C, Oliveira H (2016b) Coating independent cytotoxicity of citrate- and PEG-coated silver nanoparticles on a human hepatoma cell line. J Environ Sci 51:191–201
Behra R, Sigg L, Clift M, Herzog F, Minghetti M, Johnston B, Petri-Fink A, Rothen-Rutishauser B (2013) Bioavailability of silver nanoparticles and ions: from a chemical and biochemical perspective. J R Soc Interface 10(87):20130396
Benn T, Westerhoff P (2008) Nanoparticle silver released into water from commercially available sock fabrics. Environ Sci Technol 42(11):4133–4139
Browning L, Lee K, Nallathamby P, Xu X-HN (2013) Silver nanoparticles incite size- and dose-dependent developmental phenotypes and nanotoxicity in zebrafish embryos. Chem Res Toxicol 26(10):1503–1513
Diaz-Diestra D, Beltran-Huarac J, Bracho-Rincon DP, González-Feliciano JA, González CI, Weiner BR, Morell G (2015) Biocompatible ZnS: Mn quantum dots for reactive oxygen generation and detection in aqueous media. Journal of Nanopart Res 17(12):461
Eckhardt S, Brunetto P, Gagnon J, Priebe M, Giese B, Fromm K (2013) Nanobio silver: its interactions with peptides and bacteria, and its uses in medicine. Chem Rev 113(7):4708–4754
Ferreira de Oliveira JM, Costa M, Pedrosa T, Pinto P, Remédios C, Oliveira H, Pimentel F, Almeida L, Santos C (2014) Sulforaphane induces oxidative stress and death by p53-independent mechanism: implication of impaired glutathione recycling. PLoS One 9(3):e92980
Gliga A, Skoglund S, Odnevall Wallinder I, Fadeel B, Karlsson H (2014) Size-dependent cytotoxicity of silver nanoparticles in human lung cells: the role of cellular uptake, agglomeration and Ag release. Part Fibre Toxicol 11(1):1
Grosse S, Evje L, Syversen T (2013) Silver nanoparticle-induced cytotoxicity in rat brain endothelial cell culture. Toxicol in Vitro 27(1):305–313
Haase A, Tentschert J, Jungnickel H, Graf P, Mantion A, Draude F, Plendl J, Goetz ME, Galla S, Masic A, Thuenemann AF, Taubert A, Arlinghaus HF, Luch A (2011) Toxicity of silver nanoparticles in human macrophages: uptake, intracellular distribution and cellular responses. J Phys Conf Ser 304(1):012030 IOP Publishing
Hamilton RF, Buckingham S, Holian A (2014) The effect of size on Ag nanosphere toxicity in macrophage cell models and lung epithelial cell lines is dependent on particle dissolution. Int J Mol Sci 15(4):6815–6830
Hussain SM, Hess KL, Gearhart JM, Geiss KT, Schlager JJ (2005) In vitro toxicity of nanoparticles in BRL 3A rat liver cells. Toxicol in Vitro 19(7):975–983
Kajstura M, Halicka HD, Pryjma J, Darzynkiewicz Z (2007) Discontinuous fragmentation of nuclear DNA during apoptosis revealed by discrete "sub-G1" peaks on DNA content histograms. Cytometry A 71(3):125–131
Kaur J, Tikoo K (2013) Evaluating cell specific cytotoxicity of differentially charged silver nanoparticles. Food Chem Toxicol 51:1–14
Li Y, Zhang Y, Yan B (2014) Nanotoxicity overview: nano-threat to susceptible populations. Int J Mol Sci 15(3):3671–3697
Liu WT (2006) Nanoparticles and their biological and environmental applications. J Biosci Bioeng 102(1):1–7
Misra R, Easton M (1999) Comment on analyzing flow cytometric data for comparison of mean values of the coefficient of variation of the G1 peak. Cytometry 36(2):112–116
Mukherjee D, Royce SG, Sarkar S, Thorley A, Schwander S, Ryan MP, Porter AE, Chung KF, Tetley TD, Zhang J, Georgopoulos PG (2014) Modeling in vitro cellular responses to silver nanoparticles. J Toxicol 2014
Munusamy P, Wang C, Engelhard MH, Baer DR, Smith JN, Liu C, Kodali V, Thrall BD, Chen S, Porter AE, Ryan MP (2015) Comparison of 20 nm silver nanoparticles synthesized with and without a gold core: structure, dissolution in cell culture media, and biological impact on macrophages. Biointerphases 10(3):031003
Nowack B, Bucheli T (2007) Occurrence, behavior and effects of nanoparticles in the environment. Environ Pollut 150(1):5–22
Nowack B, Krug H, Height M (2011) 120 years of nanosilver history: implications for policy makers. Environ Sci Technol 45(4):1177–1183
Oberdorster G, Maynard A, Donaldson K, Castranova V, Fitzpatrick J, Ausman K, Carter J, Karn B, Kreyling W, Lai D et al (2005) Principles for characterizing the potential human health effects from exposure to nanomaterials: elements of a screening strategy. Part Fibre Toxicol 2:8
Oliveira H, Monteiro C, Pinho F, Pinho S, Ferreira de Oliveira JM, Santos C (2014) Cadmium-induced genotoxicity in human osteoblast-like cells. Mutat Res Genet Toxicol Environ Mutagen 775-776:38–47
Orlowski P, Krzyzowska M, Zdanowski R, Winnicka A, Nowakowska J, Stankiewicz W, Tomaszewska E, Celichowski G, Grobelny J (2013) Assessment of in vitro cellular responses of monocytes and keratinocytes to tannic acid modified silver nanoparticles. Toxicol in Vitro 27(6):1798–1808
Orr GA, Chrisler WB, Cassens KJ, Tan R, Tarasevich BJ, Markillie LM, Zangar RC, Thrall BD (2011) Cellular recognition and trafficking of amorphous silica nanoparticles by macrophage scavenger receptor A. Nanotoxicology 5(3):296–311
Park E-JJ, Yi J, Kim Y, Choi K, Park K (2010) Silver nanoparticles induce cytotoxicity by a Trojan-horse type mechanism. Toxicol InVitro 24(3):872–878
Park J, Lim D-HH, Lim H-JJ, Kwon T, Choi J-SS, Jeong S, Choi I-HH, Cheon J (2011) Size dependent macrophage responses and toxicological effects of Ag nanoparticles. Chem Commun (Camb) 47(15):4382–4384
Pratsinis A, Hervella P, Leroux JC, Pratsinis SE, Sotiriou GA (2013) Toxicity of silver nanoparticles in macrophages. Small 9(15):2576–2584
Salata OV (2004) Applications of nanoparticles in biology and medicine. J Nanobiotechnology 2(1):3
Sasidharan A, Chandran P, Menon D, Raman S, Nair S, Koyakutty M (2011) Rapid dissolution of ZnO nanocrystals in acidic cancer microenvironment leading to preferential apoptosis. Nano 3(9):3657–3669
Shannahan JH, Sowrirajan H (2015) Impact of silver and iron nanoparticle exposure on cholesterol uptake by macrophages. J Nanomater 501:127235
Sharma V, Yngard R, Lin Y (2009) Silver nanoparticles: green synthesis and their antimicrobial activities. Adv Colloid Interf Sci 145(1):83–96
Smita S, Gupta S, Bartonova A, Dusinska M, Gutleb A, Rahman Q (2012) Nanoparticles in the environment: assessment using the causal diagram approach. Environ Health 11(Suppl 1):S13
Twentyman P, Luscombe M (1987) A study of some variables in a tetrazolium dye (MTT) based assay for cell growth and chemosensitivity. Br J Cancer 56(3):279–285
Varner K, Sanford J, El-Badawy A, Feldhake D, Venkatapathy R (2010) State of the science literature review: everything nanosilver and more. US Environmental Protection Agency, Washington, DC, EPA/600/R-10/084
Wang H, Wu L, Reinhard BMM (2012) Scavenger receptor mediated endocytosis of silver nanoparticles into J774A.1 macrophages is heterogeneous. ACS Nano 6(8):7122–7132
Wang X, Ji Z, Chang C, Zhang H, Wang M, Liao Y-P, Lin S, Meng H, Li R, Sun B et al (2014) Use of coated silver nanoparticles to understand the relationship of particle dissolution and bioavailability to cell and lung toxicological potential. Small 10(2):385–398
Zhang T, Wang L, Chen Q, Chen C (2014) Cytotoxic potential of silver nanoparticles. Yonsei Med J 55(2):283–291
Zhang H, Wang X, Wang M, Li L, Chang CH, Ji Z, Xia T, Nel AE (2015) Mammalian cells exhibit a range of sensitivities to silver nanoparticles that are partially explicable by variations in antioxidant defense and Metallothionein expression. Small 11(31):3797–3805
Acknowledgments
This work has been funded by the European Regional Development Fund (FEDER) through the Competitive Factors Thematic Operational Programme (COMPETE) and by National Funds through the Foundation for Science and Technology (FCT), under the projects CICECO—FCOMP-01-0124-FEDER-037271 (Refª. FCT PEst-C/CTM/LA0011/2013) and FCOMP-01-0124-FEDER-021456 (Refª. FCT PTDC/SAU-TOX/120953/2010). The grants awarded by FCT to V.B. (SFRH/BD/81792/2011) and H.O. (SFRH/BPD/111736/2015) are also acknowledged. I.F.D acknowledges FCT/MCTES for a research contract under the Program “Investigador FCT” 2014.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflicting interests
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Bastos, V., Duarte, I.F., Santos, C. et al. A study of the effects of citrate-coated silver nanoparticles on RAW 264.7 cells using a toolbox of cytotoxic endpoints. J Nanopart Res 19, 163 (2017). https://doi.org/10.1007/s11051-017-3855-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-017-3855-1