Abstract
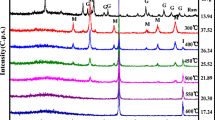
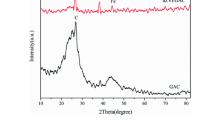
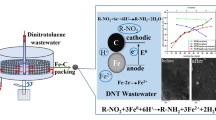
Nanoscale zero-valent iron (nZVI) was continuously prepared by high-gravity reaction precipitation through a novel impinging stream-rotating packed bed (IS-RPB). Reactant solutions of FeSO4 and NaBH4 were conducted into the IS-RPB with flow rates of 60 L/h and rotating speed of 1000 r/min for the preparation of nZVI. As-prepared nZVI obtained by IS-RPB were quasi-spherical morphology and almost uniformly distributed with a particle size of 10–20 nm. The reactivity of nZVI was estimated by the degradation of 100 ml nitrobenzene (NB) with initial concentration of 250 mg/L. The optimum dosage of nZVI obtained by IS-RPB was 4.0 g/L as the NB could be completely removed within 10 min, which reduced 20% compared with nZVI obtained by stirred tank reactor (STR). The reduction of NB and production of aniline (AN) followed pseudo-first-order kinetics, and the pseudo-first-order rate constants were 0.0147 and 0.0034 s−1, respectively. Furthermore, the as-prepared nZVI using IS-RPB reactor in this work can be used within a relatively wide range pH of 1–9.

Graphical abstract







Similar content being viewed by others
References
Agrawal A, Tratnyek PG (1996) Reduction of nitro aromatic compounds by zero-valent iron metal. Environ Sci Technol 30:153–160. doi:10.1021/es950211h
Chaithawiwat K, Vangnai A, McEvoy JM, Pruess B, Krajangpan S, Khan E (2016) Role of oxidative stress in inactivation of Escherichia coli BW25113 by nanoscale zero-valent iron. Sci Total Environ 565:857–862. doi:10.1016/j.scitotenv.2016.02.191
Chen JF, Shao L (2003) Mass production of nanoparticles by high gravity reactive precipitation technology with low cost. China Particuology 1:64–69. doi:10.1016/S1672-2515(07)60110-9
Chen JF, Wang YH, Guo F, Wang XM, Zheng C (2000) Synthesis of nanoparticles with novel technology: high-gravity reactive precipitation. Ind Eng Chem Res 39:948–954. doi:10.1021/ie990549a
Crane RA, Scott TB (2012) Nanoscale zero-valent iron: future prospects for an emerging water treatment technology. J Hazard Mater 211:112–125. doi:10.1016/j.jhazmat.2011.11.073
Dirksen JA, Ring TA (1991) Fundamentals of crystallizations kinetic effects on particle size distributions and morphology. Chem Eng Sci 46:2389–2427. doi:10.1016/0009-2509(91)80035-W
Dong J, Zhao YS, Zhao R, Zhou R (2010) Effects of pH and particle size on kinetics of nitrobenzene reduction by zero-valent iron. J EnvironSci 22:1741–1747. doi:10.1016/s1001-0742(09)60314-4
Fan HL, Zhou SF, Qi GS, Liu YZ (2016) Continuous preparation of Fe3O4 nanoparticles using impinging stream-rotating packed bed reactor and magnetic property thereof. J Alloys Compd 662:497–504. doi:10.1016/j.jallcom.2015.12.025
Feng ZR, Jiao WZ, Liu YZ, Xu CC, Guo L, Yu LS (2015) Treatment of nitrobenzene-containing wastewater by iron-carbon micro-electrolysis. CIESC Journal 66:1150–1155. doi:10.11949/j.issn.0438-1157.20141329
Guo K, Guo F, Feng YD, Chen JF, Zheng C, Gardner NC (2000) Synchronous visual and RTD study on liquid flow in rotating packed-bed contactor. Chem Eng Sci 55:1699–1706. doi:10.1016/s0009-2509(99)00369-3
Hahn H (1997) Gas phase synthesis of nanocrystalline materials. Nanostruct Mater 9:3–12. doi:10.1016/s0965-9773(97)00013-5
Jiao WZ, Liu YZ, Qi GS (2010) A new impinging stream-rotating packed bed reactor for improvement of micromixing iodide and iodate. Chem Eng J 157:168–173. doi:10.1016/j.cej.2009.11.032
Jiao WZ, Feng ZR, LIu YZ, Jiang HH (2016a) Degradation of nitrobenzene-containing wastewater by carbon nanotubes immobilized nanoscale zerovalent iron. J Nanopart Res 18:198. doi:10.1007/s11051-016-3512-0
Jiao WZ, Wang YH, Li XX, Xu CC, Liu YZ, Zhang QL (2016b) Stabilization performance of methanol-diesel emulsified fuel prepared using an impinging stream-rotating packed bed. Renew Energ 85:573–579. doi:10.1016/j.renene.2015.06.068
Karlsson MNA, Deppert K, Wacaser BA, Karlsson LS, Malm JO (2005) Size-controlled nanoparticles by thermal cracking of iron pentacarbonyl. Appl Phys A-Mater 80:1579–1583. doi:10.1007/s00339-004-2987-1
Lefevre E, Bossa N, Wiesner MR, Gunsch CK (2016) A review of the environmental implications of in situ remediation by nanoscale zero valent iron (nZVI): behavior, transport and impacts on microbial communities. Sci Total Environ 565:889–901. doi:10.1016/j.scitotenv.2016.02.003
Li SL, Yan WL, Zhang WX (2009) Solvent-free production of nanoscale zero-valent iron (nZVI) with precision milling. Green Chem 11:1618–1626. doi:10.1039/b913056j
Li YF, Ye HQ, He XD, Han K, Liu H (2012) Synthesis of Ce-doped yttrium aluminum garnet phosphor by impinging streams co-precipitation. J Cent South Univ 19:324–330. doi:10.1007/s11771-012-1008-3
Lin CC, Ho JM, Wu MS (2015) Continuous preparation of Fe3O4 nanoparticles using a rotating packed bed: dependence of size and magnetic property on temperature. Powder Technol 274:441–445. doi:10.1016/j.powtec.2015.01.013
Ling XF et al (2012) Synthesis of nanoscale zero-valent iron/ordered mesoporous carbon for adsorption and synergistic reduction of nitrobenzene. Chemosphere 87:655–660. doi:10.1016/j.chemosphere.2012.02.002
Moraci N, Calabro PS (2010) Heavy metals removal and hydraulic performance in zero-valent iron/pumice permeable reactive barriers. J Environ Manag 91:2336–2341. doi:10.1016/j.jenvman.2010.06.019
Oropeza S, Corea M, Gomez-Yanez C, Cruz-Rivera JJ, Navarro-Clemente ME (2012) Zero-valent iron nanoparticles preparation. Mater Res Bull 47:1478–1485. doi:10.1016/j.materresbull.2012.02.026
Shen ZG, Chen JF, Yun J (2004) Preparation and characterizations of uniform nanosized BaTiO3 crystallites by the high-gravity reactive precipitation method. J Cryst Growth 267:325–335. doi:10.1016/j.jcrysgro.2004.03.018
Shih YH, Tai YT (2010) Reaction of decabrominated diphenyl ether by zerovalent iron nanoplarticles. Chemosphere 78:1200–1206. doi:10.1016/j.chemosphere.2009.12.061
Su LH, Zhen GY, Zhang LJ, Zhao YC, Niu DJ, Chai XL (2015) The use of the core-shell structure of zero-valent iron nanoparticles (NZVI) for long-term removal of sulphide in sludge during anaerobic digestion. Environ Sci Proc Impacts 17:2013–2021. doi:10.1039/c5em00470e
Sun Q et al (2015) Preparation of transparent suspension of lamellar magnesium hydroxide nanocrystals using a high-gravity reactive precipitation combined with surface modification. Ind Eng Chem Res 54:666–671. doi:10.1021/ie504265z
Tratnyek PG, Johnson RL (2006) Nanotechnologies for environmental cleanup. Nano Today 1:44–48. doi:10.1016/s1748-0132(06)70048-2
Wang W, Jin ZH, Li TL, Zhang H, Gao S (2006) Preparation of spherical iron nanoclusters in ethanol-water solution for nitrate removal. Chemosphere 65:1396–1404. doi:10.1016/j.chemosphere.2006.03.075
Wender H, Migowski P, Feil AF, Teixeira SR, Dupont J (2013) Sputtering deposition of nanoparticles onto liquid substrates: recent advances and future trends. Coord Chem Rev 257:2468–2483. doi:10.1016/j.ccr.2013.01.013
Xiang Y, Chu GW, Wen LX, Yang KA, Xiao GT, Chen JF (2010) Experimental investigation of nanoparticles precipitation in a rotating packed bed. Particuology 8:372–378. doi:10.1016/j.partic.2010.05.008
Yang HJ, Chu GW, Zhang JW, Shen ZG, Chen JF (2005) Micromixing efficiency in a rotating packed bed: experiments and simulation. Ind Eng Chem Res 44:7730–7737. doi:10.1021/ie0503646
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was funded by the National Natural Science Foundation of China (U1610106), the Excellent Youth Science and Technology Foundation of Province Shanxi of China (2014021007), and the Program for the Outstanding Innovative Teams of Higher Learning Institutions of Shanxi (201316).
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Jiao, W., Qin, Y., Luo, S. et al. Continuous preparation of nanoscale zero-valent iron using impinging stream-rotating packed bed reactor and their application in reduction of nitrobenzene. J Nanopart Res 19, 52 (2017). https://doi.org/10.1007/s11051-017-3758-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-017-3758-1