Abstract
Ultrafine particles (UFP) have been postulated to significantly contribute to the adverse health effects associated with exposure to particulate matter (PM). Due to their extremely small size (aerodynamic diameter <100 nm), UFP are able to deposit deep within the lung after inhalation and evade many mechanisms responsible for the clearance of larger particles. There is a lack of biologically relevant personal exposure metrics for exposure to occupational- and environmental-related micro- and nano-sized PM. The aim of the present study is to assess UFP in induced sputum (IS) and exhaled breath condensate (EBC) as possible biomarkers for assessing lung function impairment. Sputum induction and EBC testing were performed by conventional methods. UFP particles were assessed with the NanoSight LM20 (NanoSight Ltd, London, UK). The subjects included 35 exposed and 25 non-exposed workers. There were no group differences in pulmonary function test results and differential cell counts, but 63.6% of the exposed subjects had a higher percentage of neutrophils (OR3.28 p = 0.03) compared to the non-exposed subjects. The exposed subjects had higher percentages of UFP between 10 and 50 nm (69.45 ± 18.70 vs 60.11 ± 17.52 for the non-exposed group, p = 0.004). No differences were found in the IS samples. Years of exposure correlated positively to UFP content (r = 0.342 p = 0.01) and macrophage content (r = −0.327 p = 0.03). The percentage of small fraction of UFP in EBC, but not IS, is higher in exposed workers, and EBC may be a sensitive biomarker to assess exposure to nanoparticles.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Exposure to dust in the workplace is associated with a variety of pulmonary and systemic illnesses. The reactions that occur within the lungs vary with the size of the dust particle and its biologic activity. Anthropogenic sources, primarily involving the combustion of fossil fuels, account for a significant proportion of nanometer–diameter aerosols in urban areas, and many industrial processes, including welding, smelting, and the use of diesel engines, lead to the production of airborne particles in the nanometer-size range (Harrison et al. 2000). Particulate material (PM) in ambient air pollution is characterized as being coarse (PM10, aerodynamic diameter range 2.5–10 μm), fine (PM2.5, 2.5–0.1 μm), and ultrafine (UFP, nano-sized, <0.1 μm). In occupational hygiene, however, it is common to differentiate manufactured nanoparticles from ultrafine particles (UFP) derived from natural, human, or industrial sources (Debia et al. 2013). Epidemiology has identified correlations between exposure to UFP and increased pulmonary and cardiovascular morbidity and mortality (Strak et al. 2010; Sioutas et al. 2005; Meier et al. 2015). It has been postulated that upon inhalation, UFP and nanoparticles are increasingly toxic compared to larger particles because of their increased reactivity, surface area, particle number mass, deposition potential, and ability to translocate to other organ systems, such as the cardiovascular and/or neuronal system, and elicit adverse effects (Frampton 2001).
Most of the above-cited studies were based on environmental monitoring data, animal models, and controlled human exposures. There is a lack of biologically relevant personal exposure metrics for exposure to occupational- and environmental-related particulate matter. We demonstrated that analysis of particles recovered by induced sputum (IS) can serve as a biological monitoring method in addition to the traditional occupational parameters in several studies (Fireman et al. 1999; Fireman et al. 2004; Stark et al. 2014; Fireman et al. 2014; Fireman et al. 2008). Those studies were performed solely on IS samples and by measuring particle size distribution in the micro range scale (0.5–60 μm). We present here, for what we believe to be the first time, an analysis of UFP particles measured in both IS and exhaled breath condensate (EBC) in order to sample UFP from two anatomical compartments. The UFP that were measured in EBC represent the epithelial lining fluid, and the UFP from IS represents the inner epithelial compartment) (Geiser and Kreyling 2010).
Only few studies have looked at “endogenous” particles in the EBC of smokers (Bredberg et al. 2013) and occupational expose workers (Olin 2012) as a biomarker of inflammatory changes in airway disease that should reflect alterations in the respiratory tract lining fluid, whose main constituents are lipids and proteins. The particles measured in those studies, however, are only in the micro range size (coarse PM10 particles). To date, there have been no reports on individual measurements of UFP content in workers exposed to hazardous particulate matter. The purpose of the current study was to examine the dynamic differences between UFP content in IS vs UFP content in EBC and to determine whether exhaled UFP are correlated with symptomatic, functional, and/or laboratory measures and can serve as useful biomarkers for early detection of impairments in lung function in exposed workers.
Materials and methods
Study population
The study included 60 participants who were divided into two groups according to the presence of exposure. The exposed group included 25 industrial workers who were exposed to occupational dust from industrial sources while executing their work. The non-exposed control group included employees who were not exposed to any kind of dust during their work. Exposure and demographic data were retrieved by a questionnaire followed by a personal interview. The participants were adults over the age of 20 years who came to the Laboratory of Pulmonary and Allergic Diseases, Tel-Aviv Sourasky Medical Center, for a medical assessment related to various respiratory symptoms. They underwent sputum induction, EBC testing, and pulmonary function testing.
Pulmonary function tests
Pulmonary function tests (PFTs) were performed by a Masterlab spirometer (Masterlab, E. Jaeger, Wurzburg, Germany). Measurements were carried out according to standard protocols of the American Thoracic Society (ATS) guidelines (Miller et al. 2005), and the results were expressed as percent predictive value. The best of three consecutive measurements was chosen. Forced expiratory volume in 1 s (FEV1) and forced vital capacity (FVC) were recorded. The FEV1 result and the FEV1/FVC ratio were considered low if they were less than 80% of expected or less than a ratio of 0.75, respectively.
IS collection and processing
IS was obtained as previously described in detail elsewhere (Fireman et al. 1999). After pretreatment with a short-acting β-2 agonist, 3% saline was administered by a nebulizer (U1 Ultrasonic Nebulizer; Omron HealthCare, Henfield, West Sussex, UK) for up to 20 min while the subjects were encouraged to cough and expectorate sputum into a sterile container. Samples were stored at 4 °C and processed within 3 h. All portions with little or no squamous epithelial cells (i.e., a rich non-squamous epithelial cell fraction considered to originate from the lower respiratory tract, hereafter referred to as “plugs”) were collected and treated with dithiothreitol [DTT (Sputolysin); Calbiochem Corp, San Diego, CA, USA]. The cell suspension was filtered through a 52-μm nylon gauze, and the effect of DTT was stopped by diluting the suspension with phosphate-buffered solution to a volume equal to the sputum plus DTT. After centrifugation, the supernatants were resuspended and cytospinned (Shandon Southern Instruments, Sewickley, PA, USA), and the slides were stained with Giemsa.
Sputum was considered eosinophilic if at least 2.7% of the sputum cells were eosinophils and neutrophilic if at least 65% of cells were neutrophils.
EBC collection
EBC was collected by a TurboDECCS device (Medivac, Parma, Italy) according to the manufacturer’s instructions (Goldoni et al. 2013). Briefly, the subjects were asked to breathe into the collecting system for 5 min under conditions of normal tidal volume and without a noseclip. All of the samples were stored at −80 °C until analysis. All EBC collections were performed in a controlled temperature (22–23 °C) and humidity (50%) by a closed air conditioning system.
Ultrafine particle measurement
The particle size number distribution of PM0.1 was assessed in the EBC samples with the NanoSight LM20 system (NanoSight Ltd., Salisbury) using the Nanoparticle Tracking Analysis (NTA) method of visualizing and analyzing particles in liquids that relates the rate of Brownian motion to particle size. The rate of movement is related only to the viscosity of the liquid, the temperature, and the size of the particle and is not influenced by particle density or refractive index. The particles contained in the sample can be visualized by virtue of the light they scatter when illuminated by a laser light source. The technique retrieves samples from a volume of 120 × 80 × 20 microns. Sample concentration is adjusted (107–109 particles/ml) such that a significant number of particles is present in the beam path. Approximately 0.3 ml of EBC was introduced into the viewing unit using a disposable syringe. Three videos of 30-s duration were recorded and analyzed (NTA software version 2.0) for each sample. Total particle count, percent of particles that were in the nano-sized range, and total nano-sized particle counts were recorded.
Statistical analysis
Continuous PFT data, IS deferential cell counts, exposure duration, and age were correlated to exposure by the t test, while categorical data and exposure were assessed by the χ 2 test. Each measurement of PM0.1 in the EBC and in the IS samples were expressed as the average of three consecutive measurements. The frequency of particle ranges (10–50 nm) were combined to one parameter for EBC and IS and associated to exposure status by the t test. A dichotomy cutoff (61%) was used in a logistic regression model to evaluate an association to 10–50 nm particle exposure status. The within-subject effect of particles 10–50 nm in sputum and in EBC was correlated to exposure by the ANOVA test repeated measures. GLM repeated measurements were done using mixed models, and the exposure was used for the between-subject factors and the particles for the within-subject factors. The measurements were done twice in the sputum and EBC samples, respectively.
Results
The demographic and particle characteristics of the studied population are shown in Table 1. The exposed subjects were older than the non-exposed subjects (55.64 ± 14.19 vs 48.08 ± 14.49 years, respectively), had worked for more years (26.36 ± 15.86 × 14.49 vs 12.53 ± 1.8 years, p ≤ 0.01) and were composed mostly of men (20 vs 14 in the non-exposed group p ≤ 0.01).
Percent particles between 10 and 50 nm and percent total particles under 100 nm were higher in the EBC samples for exposed workers and vice versa for IS samples (p < 0.01).
The types of jobs for all study participants are listed in Table 2.
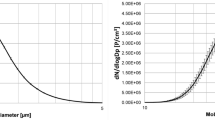
The PFT results were not significantly different between the two groups (Table 3). The differential cell counts in the exposed group had a higher percent of neutrophils (63.03 ± 24.91 vs 52.66 ± 22.83 for the non-exposed group, p = NS) and lower percentage of macrophages 16.21 ± 15.88 vs 27.16 ± 23.04, respectively, p = 0.04). The frequency of UFP in EBC and IS are shown in Fig. 1a, b. The frequency of UFP in IS was almost identical for both groups. In contrast, a different pattern was observed in the EBC results: they were higher in the fraction of 10–50 nm among the exposed subjects compared to the non-exposed ones (69.45 ± 18.70 vs 60.11 ± 17.52, respectively, p = 0.004).
Neutrophilic inflammation is an important biomarker in occupational exposures. In this context, we divided individuals according to a cutoff of 61% neutrophils in the differential cell counts in IS. The results showed that 65.2% of the exposed individuals displayed a neutrophilic pattern compared to only 36.4% of the non-exposed ones (OR 3.28 p = 0.03) (Table 4). Moreover, an increase in percentage of UFP fraction of 10–50 nm had an OR of 1.08 (p = 0.006) (Table 5) to belong to the group of neutrophilic inflammation and vice versa having neutrophilic inflammation predict high risk to accumulate nanoranged particles (fraction of 10–50 nm). The fraction of 10–50 nm in IS did not show a comparable correlation (Fig. 1b). The length of exposure correlated positively to the UFP in EBC (r = 0.342 p = 0.01) and macrophage percentage (r = −0.327 p = 0.03) (Tables 1 and 6).
The gradient of UFP in IS and EBC showed a marked difference between exposed vs non-exposed individuals, being much higher in the latter (59.8 ± 2.68 vs 79.87 ± 1.12 in IS and 72.34 ± 3.19 vs 79.17 ± 1.34, respectively, p = 0.01) (Tables 1 and 7).
Discussion
There are many concerns that nano-sized materials might introduce health risks upon occupational and consumer exposure. Although many of them are produced, handled, and present in fluids during aerosolization of energetic processes, such as vortexing, weighing, sonication, mixing, and blending, no studies have estimated the internal personal-level burden of UFP by means of biological monitoring. To the best of our knowledge, we now present the first study to measure UFP in biological samples in an occupational setup. In the current investigation, we directly measured particulate matter in IS and EBC samples from the airways of exposed individuals compared to non-exposed individuals with the aim of finding a biomarker that can monitor UFP accumulation as a marker of exposure in correlation to functional and inflammatory parameters.
We had previously shown that combined IS and EBC measurements detect underlying inflammation in airways of asymptomatic welders. It emerged that the particle burden (higher % particles >2 mm in diameter), inflammatory cells (higher % neutrophils), and level of oxidative stress (H2O2 in EBC) were a function of the type and the duration of welding (Fireman et al. 2008). Moreover, we demonstrated that biological monitoring by IS indicated that a >92% accumulation of <5-μm particles correlated significantly to a positive beryllium lymphocyte proliferation test result in a group of 63 Israeli and 37 American workers exposed to hazardous dust containing beryllium (OR 3.8, 95% CI 1.2–11.4, p = 0.015) among all participants with a follow-up of 2 years (Fireman et al. 2014).
This is our first report dealing with individual measurements of UFP content in workers exposed to hazardous particulate matter. We tested exposed vs non-exposed individuals referred because of respiratory symptoms. The exposed population was older, had fewer females, and had accumulated more years in the workplace than the non-exposed population. These results are compatible with other publications that showed that patients with a history of high exposure to gases/fumes were less likely to be women and that those with high biological dust exposure were more likely to be older (Rodríguez et al. 2014). The same was shown in exposures to carbon black dust (Neghab et al. 2011).
In our current study, the participants had been exposed to a variety of hazardous particulate matter, and the results revealed that there is a general delay of many years between occupational exposures and the development of pulmonary disease. Our study population represents a unique cohort of workers that were referred for respiratory evaluation after many years of exposure. The initial respiratory symptoms were present, but there was no functional impairment. In fact, there were no differences in pulmonary function parameters between the exposed and the non-exposed individuals.
The findings of the present study emphasize the role of biomarkers involved in inflammatory processes and in the biomonitoring of particulate matter for early detection of injury due to occupational exposure. These findings may be of considerable epidemiological relevance for the assessment of ultrafine particle content.
We had recently evaluated the effect of individual exposure to UFP on functional respiratory parameters and airway inflammation in 52 children aged 6–18 years who had been referred for assessment due to respiratory symptoms. We found that the total and percent of UFP content correlated with wheezing, breath symptom score, and sputum eosinophilia (Benor et al. 2015). The eosinophils we found were not correlated to any of the parameters we studied.
Neutrophils comprise the main type of cells that correlates with occupational exposures, as we previously demonstrated in our study on welders (Fireman et al. 2008) and firefighters (Fireman et al. 2004). The cutoff for abnormality for neutrophils was 61%. This choice was based on a recent unpublished study (2005–2012) on the leukocyte counts of 905 Israeli patients diagnosed as having various pulmonary diseases: We adopted this value at the cutoff value for the current investigation. A very recent study on Indian goldsmiths found that neutrophils in sputum correlated with occupational cadmium exposure, spirometry, and lung cell DNA damage (Moitra et al. 2015). In contrast with our earlier findings and those of others, in the current work, we directly demonstrated that this neutrophilic inflammation is correlated with UFP retrieved from EBC samples but not from UFP recovered from IS samples. Moreover, IL-8, the classical cytokine that induced chemotaxis for neutrophils, was significantly higher in supernatants recovered from IS samples in the exposed workers compared to the non-exposed (data not shown). This raised the interesting question about the patterns of deposition of UFP particles in the airways and their internalization pathway. All of our exposed subjects were actively employed, and the IS and EBC sampling was done within 24 h from their last exposure at the workplace. The exposure was chronic and continued over extended periods of time. In the exposed workers, the small fraction of UFP (10–50 nm) were retained in the epithelial lining fluid and recovered by EBC, while the largest fraction of UFP (50–100 nm) were rapidly translocated to inner compartments. It is known that after particle inhalation and their deposition on the lung epithelium, the retention of particles starts with their wetting by surfactant and the epithelial lining fluid and their subsequent displacement from the air into the aqueous phase regardless of particle shape, surface topography, and surface-free energy (Geiser et al. 2003). Our results support the statement by Moller et al. that the probability of long-term particle retention in the airways is inversely correlated to particle size as was shown in both of our study groups. The amount of retained fraction (10–50 nm), however, was higher in our exposed group. This may be due to the different degrees of airway inflammation among the subjects in both groups. Our exposed workers showed here a clear neutrophilic pattern due to exposure. In this context, we recently showed that the pattern of distribution of UFP that were recovered from the bronchoalveolar lavage of mice exposed to smoke was dependent upon the inflammatory status of the animals and that the retention or translocation of UFP through the alveolar barrier was highly dependent upon inflammatory status (Bar-Shai et al. 2015).
The main limitations of the research are the heterogeneity of the study group and a lack of chemical analysis of particles. We did not analyze metal contents in the present study, but the results of our lab’s earlier analysis of metals in EBC samples by X-ray fluorescence showed that they had not differed between exposed and non-exposed workers (supporting the contention that the main effect of the very small particles is caused by the neutrophilic inflammation and not by the type of metal).
Based on the clear differences that we found between the exposed and non-exposed participants of this study, we are confident that our method may offer new opportunities in the new field of nanomedicine for the diagnosis and therapy of pulmonary airway disease caused by exposure to airborne nano-sized particulate matter.
Conclusions
This study is the first to report a direct dosimetry as well as describe exposure metrics for identifying very small particles recovered from the airways of exposed workers. Nanoparticles may play a crucial role in future surveillance programs in terms of elucidating obscure toxicity resulting from exposure in multiple industries.
References
Bar-Shai A, Alcalay Y, Sagiv A, Rotem M, Feigelson SW, Alon R, Fireman E (2015) Fingerprint of lung fluid ultrafine particles, a novel marker of acute lung inflammation respiration. 90:74–84
Benor S, Alcalay Y, Domany KA, Gut G, Soferman R, Kivity S, Fireman E (2015) Ultrafine particle content in exhaled breath condensate in airways of asthmatic children. J Breath Res 9:026001
Bredberg A, Josefson M, Almstrand AC, Lausmaa J, Sall P, Levinsson A, Larsson P, Olin AC (2013) Comparison of exhaled endogenous particles from smokers and non-smokers using multivariate analysis. Respiration 86:135–142
Debia M, Beaudry C, Weichenthal S, Tardif R, Dufrresne A (2013) Characterization and control of occupational exposure to nanoparticles and ultrafine particles. André Studies and Research Projects/Report R-777, Montréal, IRSST
Enright HA, Bratt JM, Bluhm AP, Kenyon NJ, Louie AY (2013) Tracking retention and transport of ultrafine polystyrene in an asthmatic mouse model using positron emission tomography. Exp Lung Res 39:304–313
Fireman E, Greif J, Schwarz Y, Man A, Ganor E, Ribak Y, Lerman Y (1999) Assessment of hazardous dust exposure by BAL and induced sputum. Chest 115:1720–1728
Fireman EM, Lerman Y, Ganor E, Greif J, Fireman-Shoresh S, Lioy PJ, Banauch GI, Weiden M, Kelly KJ, Prezant DJ (2004) Induced sputum assessment in New York City firefighters exposed to World Trade Center dust. Environ Health Perspect 112:1564–1569
Fireman E, Lerman Y, Stark M, Grinberg N, Primer R, Landau DA, Zilberberg M, Schwartz Y, Jacovovitz R (2008) Detection of occult lung impairment in welders by induced sputum particles and breath oxidation. Am J Indust Med 51:503–511
Fireman E, Lerman Y, Stark M, Pardo A, Schwarz Y, Van Dyke M, Elliot J, Barkes B, Newman L, Maier L (2014) A novel alternative to environmental monitoring to detect workers at risk for beryllium exposure-related health effects. J Occup Environ Hyg 11:809–818
Frampton MW (2001) Systemic and cardiovascular effects of airway injury and inflammation: ultrafine particle exposure in humans. Environ Health Perspect 109(Suppl 4):529–532
Geiser M, Kreyling WG (2010) Deposition and biokinetics of inhaled nanoparticles. Fibre Toxicol 7:2
Geiser M, Matter M, Maye I, Im Hof V, Gehr P, Schurch S (2003) Influence of airspace geometry and surfactant on the retention of man-made vitreous fibers (MMVF 10a). Environ Health Perspect 111:895–901
Goldoni M, Corradi M, Mozzoni P, Folesani G, Alinovi R, Pinelli S, Andreoli R, Pigini D, Tillo R, Filetti A, Garavelli C, Mutti A (2013) Concentration of exhaled breath condensate biomarkers after fractionated collection based on exhaled CO2 signal. J Breath Res 7:171–179
Harrison RM, Shi JP, Shuhua X, Kahn A, Mark D, Kinnersley R et al (2000) Measurement of number, mass and size distribution of particles in the atmosphere. Philos Trans Roy Soc Lond Ser. A—Math. Phys Eng Sci 358(1775):2567–2580
Meier R, Eeftens M, Aguilera I, Phuleria HC, Ineichen A, Davey M, Ragettli MS, Fierz M, Schindler C, Probst-Hensch N, Tsai MY, Künzli N (2015) Ambient ultrafine particle levels at residential and reference sites in urban and rural Switzerland. Environ Sci Technol 49:2709–2715
Miller MR, Hankinson J, Brusasco V, Burgos F, Casaburi R, Coates A, ATS/ERS Task Force (2005) Standardisation of spirometry. Eur Respir J 26:319–338
Moitra S, Chakraborty K, Bhattacharyya A, Sahu S (2015) Impact of occupational cadmium exposure on spirometry, sputum leukocyte count, and lung cell DNA damage among Indian goldsmiths. Am J Ind Med 58:617–624
Moller W, Felten K, Sommerer K, Scheuch G, Meyer G, Meyer P, Haussinger K, Kreyling WG (2008) Deposition, retention, and translocation of ultrafine particles from the central airways and lung periphery. Am J Respir Crit Care Med 177:426–432
Neghab M, Habibi M, Hassanzadeh J (2011) Symptoms of respiratory disease and lung functional impairment associated with occupational inhalation exposure to carbon black dust. J Occup Health 53:432–438
Olin AC (2012) Induced sputum, exhaled nitric oxide, and particles in exhaled air in assessing airway inflammation in occupational exposures. Clin Chest Med 33:771–782
Rodríguez E, Ferrer J, Zock P, Srra I, Anto JM, Battle J, Kromhout H, Vemeulen R, Gonzalea DD, Benet M, Balcells E, Monso E, Gayete A, Aymerich AG (2014) Lifetime occupational exposure to dusts, gases and fumes is associated with bronchitis symptoms and higher diffusion capacity in COPD patients. PLoS One 9:e88426
Stark M, Lerman Y, Kapel A, Pardo A, Swartz Y, Newman L, Maier L, Fireman E (2014) Induced sputum particles in beryllium-exposed dental technicians reflect hygiene and oxidative stress. Arch Occup Environ Health 69:89–99
Sioutas C, Delfino RJ, Singh M (2005) Exposure assessment for atmospheric ultrafine particles (UFPs) and implications in epidemiologic research. Environ Health Perspect 113:947–955
Strak M, Boogaard H, Meliefste K, Oldenwening M, Zuurbier M, Brunekreef B, Hoek G (2010) Respiratory health effects of ultrafine and fine particle exposure in cyclists. Occup Environ Med 67:118–124
Yu Y, Duan J, Li Y, Yu Y, Jin M, Li C, Wang Y, Sun Z (2015) Combined toxicity of amorphous silica nanoparticles and methylmercury to human lung epithelial cells. Ecotoxicol Environ Saf 112:144–152
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Fireman, E., Edelheit, R., Stark, M. et al. Differential pattern of deposition of nanoparticles in the airways of exposed workers. J Nanopart Res 19, 30 (2017). https://doi.org/10.1007/s11051-016-3711-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-016-3711-8