Abstract
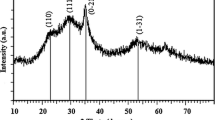
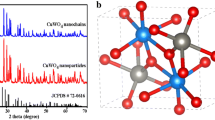
CuS nanoparticles (NPs) of few nanometers in size were prepared by a wet chemical method. The structural, compositional, and optical properties of the NPs were characterized by X-ray diffraction (XRD), scanning electron microscopy, transmission electron microscopy, energy-dispersive X-ray spectroscopy, micro Raman and Fourier transform infrared spectroscopy, N2 adsorption–desorption isotherms, and UV–Vis diffuse reflectance spectroscopy. The XRD pattern proved the presence of hexagonal phase of CuS particles which was further supported by Raman spectrum. The estimated band gap energy of 2.05 eV for the slightly sulfur-rich CuS NPs is relatively larger than that of bulk CuS (1.85 eV), indicating the small size effect. As-prepared NPs showed excellent photocatalytic activity for the degradation of methylene blue (MB) under visible light. The surface-bound OH− ions at the CuS nanostructures help adsorb MB molecules facilitating their degradation process under visible light illumination. The studies presented in this paper suggest that the synthesized CuS NPs are promising, efficient, stable, and visible-light-sensitive photocatalyst for the remediation of wastewater polluted by chemically stable azo dyes such as MB.
Graphical Abstract











Similar content being viewed by others
References
Basu M, Sinha AK, Pradhan M, Sarkar S, Negishi Y, Govind Pal T (2010) Evolution of hierarchical hexagonal stacked plates of CuS from liquid-liquid interface and its photocatalytic application for oxidative degradation of different dyes under indoor lighting. Environ Sci Technol 44:6313–6318
Cao JG, Shen M, Zhou LW (2006) Preparation and electrorheological properties of triethanolamine-modified TiO2. J Solid State Chem 179:1565–1568
Chen YB, Chen L, Wu LM (2008) Water-induced thermolytic formation of homogeneous core–shell CuS microspheres and their shape retention on desulfurization. Cryst Growth Des 8:2736–2740
Corro G, Bañuelos F, Vidal E, Cebada S (2014) Measurements of surface acidity of solid catalysts for free fatty acids esterification in jatropha curcas crude oil for biodiesel production. Fuel 115:625–628
Daghrir R, Drogui P, Robert D (2013) Modified TiO2 for environmental photocatalytic applications: a review. Ind Eng Chem Res 52:3581–3599
Deng C, Ge X, Hu H, Yao L, Han C, Zhao D (2014) Template-free and green sonochemical synthesis of hierarchically structured CuS hollow microspheres displaying excellent fenton-like catalytic activities. Cryst Eng Comm 16:2738–2745
Gorai S, Ganguli D, Chaudhuri S (2005) Synthesis of copper sulfides of varying morphologies and stoichiometries controlled by chelating and nonchelating solvents in a solvothermal process. Cryst Growth Des 5:875–877
Gu W, Wu P (2007) FT-IR and 2D-IR spectroscopic studies on the effect of ions on the phase separation behavior of PVME aqueous solution. Anal Sci 23:823–827
Guo Y, Wang L, Yang L, Zhang J, Jiang L, Ma X (2011) Optical and photocatalytic properties of arginine-stabilized cadmium sulfide. Mater Lett 65:486–489
Gupta VK, Pathania D, Agarwal S, Singh P (2012) Adsorptional photocatalytic degradation of methylene blue onto pectin–CuS nanocomposite under solar light. J Hazard Mater 243:179–186
Han Y, Wang Y, Gao W, Wang Y, Jiao L, Yuan H, Liu S (2011) Synthesis of novel CuS with hierarchical structures and its application in lithium-ion batteries. Powder Technol 212:64–68
Hoffmann MR, Martin ST, Choi W, Bahneman DW (1995) Environmental applications of semiconductor photocatalysis. Chem Rev 95:69–96
Houas A, Lachheb H, Ksibi M, Elaloui E, Guillard C, Herrmann JM (2001) Photocatalytic degradation pathway of methylene blue in water. Appl Catal B 31:145–157
Jeromenok J, Weber J (2013) Restricted Access: on the nature of adsorption/desorption hysteresis in amorphous, microporous polymeric materials. Langmuir 29:12982–12989
Jiang X, Xie Y, Lu J, He W, Zhu L, Qian Y (2000) Preparation and phase transformation of nanocrystalline copper sulfides (Cu9S8, Cu7S4 and CuS) at low temperature. J Mater Chem 10:2193–2196
Kumar P, Gusain M, Nagarajan R (2011) Synthesis of Cu1.8S and CuS from copper-thiourea containing precursors; anionic (Cl−, NO3 −, SO4 2−) influence on the product stoichiometry. Inorg Chem 50:3065–3070
Kumar P, Nagarajan R, Sarangi R (2013) Quantitative X-ray absorption and emission spectroscopies: electronic structure elucidation of Cu2S and CuS. J Mater Chem C 1:2448–2454
Li HL, Zhu YC, Avivi S, Palchik O, Xiong JP, Koltypin Y, Palchik V, Gedanken A (2002) Sonochemical process for the preparation of α-CuSe nanocrystals and flakes. J Mater Chem 12:3723–3727
Linsebigler AL, Lu G, Yates JT (1995) Photocatalysis on TiO2 surfaces: principles, mechanisms, and selected results. Chem Rev 95:735–758
Liufu SC, Chen LD, Yao Q, Huang FQ (2008) In situ assembly of CuxS quantum-dots into thin film: a highly conductive p-type transparent film. J Phys Chem C 112:12085–12088
Mane RS, Lokhande CD (2000) Chemical deposition method for metal chalcogenide thin films. Mater Chem Phys 65:1–31
Meng X, Tian G, Chen Y, Zhai R, Zhou J, Shi Y, Cao X, Zhoua W, Fu H (2013) Hierarchical CuS hollow nanospheres and their structure-enhanced visible light photocatalytic properties. Cryst Eng Commun 15:5144–5149
Munce CG, Parker GK, Holt SA, Hope GA (2007) A Raman spectroelectrochemical investigation of chemical bath deposited CuxS thin films and their modification. Colloids Surf A 295:152–158
Nagaveni K, Sivalingam G, Hegde MS, Madras G (2004) Solar photocatalytic degradation of dyes: high activity of combustion synthesized nano TiO2. Appl Catal B 48:83–93
Ou S, Xie Q, Ma D, Liang J, Hu X, Yu W, Qian Y (2005) A precursor decomposition route to polycrystalline CuS nanorods. Mater Chem Phys 94:460–466
Panigrahi PK, Pathak A (2013) The growth of bismuth sulfide nanorods from spherical-shaped amorphous precursor particles under hydrothermal condition. J Nanopart, Article ID 367812:1–11
Pei LZ, Wang JF, Tao XX, Wang SB, Dong YP, Fan CG, Zhang QF (2011) Synthesis of CuS and Cu1.1Fe1.1S2 crystals and their electrochemical properties. Mater Charact 62:354–359
Qiu X, Miyauchi M, Yu H, Irie H, Hashimoto K (2010) Visible-light-driven Cu(II) − (Sr1−yNay)(Ti1−xMox)O3 photocatalysts based on conduction band control and surface ion modification. J Am Chem Soc 132:15259–15267
Roy P, Srivastava SK (2007) Low-temperature synthesis of CuS nanorods by simple wet chemical method. Mater Lett 61:1693–1697
Safrani T, Jopp J, Golan Y (2013) A comparative study of the structure and optical properties of copper sulfide thin films chemically deposited on various substrates. RSC Adv 3:23066–23074
Setkus A, Galdikas A, Mironas A, Simkieni I, Ankutiene I, Janickis V, Kaciulis S, Mattogno G, Ingo GM (2001) Properties of CuxS thin film based structures: influence on the sensitivity to ammonia at room temperatures. Thin Solid Films 391:275–281
Shen G, Chen D, Tang K, Liu X, Huang L, Qian Y (2003) General synthesis of metal sulfides nanocrystallines via a simple polyol route. J Solid State Chem 173(1):232–235
Shu QW, Li CM, Gao PF, Gaoa MX, Huang CZ (2015) Porous hollow CuS nanospheres with prominent peroxidase-like activity prepared in large scale by a one-pot controllable hydrothermal step. RSC Adv 5:17458–17465
Sing KSW, Everett DH, Haul RAW, Moscou L, Pierotty RA, Rouquerol J, Siemieniewska T (1985) Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity. Pure Appl Chem 57:603–619
Van der Vegt NFA, Kusuma VA, Freeman BD (2010) Basis of solubility versus TC correlations in polymeric gas separation membranes. Macromolecules 43:1473–1479
Wang X, Xu C, Zhang Z (2006) Synthesis of CuS nanorods by one-step reaction. Mater Lett 60(3):345–348
Wang X, Fang Z, Lin X (2009) Copper sulfide nanotubes: facile, large-scale synthesis, and application in photodegradation. J Nanopart Res 11:731–736
Wu C, Zhou G, Mao D, Zhang Z, Wu Y, Wang W, Luo L, Wang L, Yu Y, Hu J, Zhu Z, Zhang Y, Jie J (2013) CTAB assisted synthesis of CuS microcrystals: synthesis, mechanism, and electrical properties. J Mater Sci Technol 29(11):1047–1052
Xiong S, Xi B, Qian Y (2010) CdS hierarchical nanostructures with tunable morphologies: preparation and photocatalytic Properties. J Phys Chem C 114:14029–14035
Xu N, Shi Z, Fan Y, Dong J, Shi J, Hu M (1999) Effects of particle size of TiO2 on photocatalytic degradation of methylene blue in aqueous suspensions. Ind Eng Chem Res 38:373–379
Xu Y, Salim N, Bumby CW, Tilley RD (2009) Synthesis of SnS quantum dots. J Am Chem Soc 131:15990–15991
Zhang W, Wen X, Yang S (2003) Synthesis and Characterization of uniform arrays of copper sulfide nanorods coated with nanolayers of polypyrrole. Langmuir 19:4420–4426
Zhang YC, Du ZN, Li KW, Zhang M, Dionysiou DD (2011) High-performance visible-light-driven SnS2/SnO2 nanocomposite photocatalyst prepared via in situ hydrothermal oxidation of SnS2 nanoparticles. ACS Appl Mater Interfaces 3:1528–1537
Acknowledgments
Authors thank Dr. J.M. Gracia y Jimenez for providing the FT-IR equipment, and the central laboratory of IFUAP-BUAP for the Raman spectroscopy facility. The technical assistance of Rogelio Moran Elvira in SEM measurement and Ma. Luisa Raman Garcia in XRD analysis has been acknowledged. The CuS nanoparticles used in this work was developed for the projects: Centro Mexicano de Innovación en Energía Solar (CeMIE-Sol 207450/P28), Programa de Apoyo a Proyectos de Investigación e Innovación Tecnológica (PAPIIT-UNAM IN 113214, IN 107815), and Consejo Nacional de Ciencia y Tecnología (CONACyT-238869).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pal, M., Mathews, N.R., Sanchez-Mora, E. et al. Synthesis of CuS nanoparticles by a wet chemical route and their photocatalytic activity. J Nanopart Res 17, 301 (2015). https://doi.org/10.1007/s11051-015-3103-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-015-3103-5