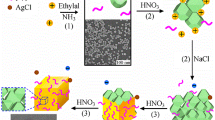
Abstract
Noble metal nano/microstructures have attracted considerable attention because of their unique properties and their various applications. Controlling the shape of noble metal nano/microstructures is a promising strategy to tailor their physical and chemical properties for various applications in fields such as biological labeling and imaging, catalysis, and sensing. Among various specific structures, flower-like and hierarchical silver nano/microstructures have attracted increasing interest because exploration of these novel nano/microstructures with unusual optical properties can provide new perspectives into the rational design of novel materials. It is significantly more challenging to develop facile and effective solution approaches for systematic manipulation of the shape of Ag nano/microstructures. In this article, we revisited the ascorbic acid reduction method to prepare flower-like silver microcrystal with plate petals and hierarchical Ag microcrystal on a large scale and in high purity. Ascorbic acid plays two roles of a reducing agent and a crystal growth regulator. Therefore, the molar ratio of ascorbic acid and silver nitrate is critical to the formation of Ag microcrystal. The controlling of the two different Ag microstructures can be achieved by adjusting the molar ratio of the reactants in aqueous medium at room temperature. The as-prepared Ag microcrystals were characterized by transmission electron microscopy, scanning electron microscopy, and X-ray diffraction. The flower-like Ag microcrystal with plate petals and hierarchical Ag microcrystal with nanoscale sharp tips and gaps could exhibit high catalytic activity and strong surface-enhanced Raman spectroscopy (SERS) activity due to the high surface area and the local electromagnetic field intensity enhancement, respectively. The potential application of the as-prepared Ag microcrystals in catalysis and SERS was investigated, which revealed that these two kinds of Ag microcrystals exhibit high catalytic activities to the NaBH4-catalyzed reduction of 4-nitrophenol and significant SERS effect to 4-aminothiophenol molecular due to their nanoscale sharp tips and gaps. Therefore, the flower-like Ag microcrystal and hierarchical Ag microcrystal investigated here could be promising candidates for single particle catalyst and SERS.





Similar content being viewed by others
References
Cao B, Liu B, Yang J (2013) Facile synthesis of single crystalline gold nanoplates and SERS investigations of 4-aminothiophenol. CrystEngComm 15:5735–5738
Fukuyo T, Imai H (2002) Morphological evolution of silver crystals produced by reduction with ascorbic acid. J Cryst Growth 241:193–199
Gu S, Wang W, Tan F, Gu J, Qiao X, Chen J (2014) Facile route to hierarchical silver microstructures with high catalytic activity for the reduction of p-nitrophenol. Mater Res Bull 49:138–143
Hao E, Schatz GC, Hupp JT (2004) Synthesis and optical properties of anisotropic metal nanoparticles. J Fluoresc 14:331–341
Hao F, Nehl CL, Hafner JH, Nordlander P (2007) Plasmon resonances of a gold nanostar. Nano Lett 7:729–732
Jana NR, Gearheart L, Murphy CJ (2001) Wet chemical synthesis of silver nanorods and nanowires of controllable aspect ratio. Chem Commun 617–618
Jin R, Cao Y, Mirkin CA, Kelly KL, Schatz GC, Zheng JG (2001) Photoinduced conversion of silver nanospheres to nanoprisms. Science 294:1901–1903
Kang L, Xu P, Chen D, Zhang B, Du Y, Han X, Li Q, Wang HL (2013) Amino acid-assisted synthesis of hierarchical silver microspheres for single particle surface-enhanced Raman spectroscopy. J Phys Chem C 117:10007–10012
Kim H, Kosuda KM, Van Duyne RP, Stair PC (2010) Resonance Raman and surface- and tip-enhanced Raman spectroscopy methods to study solid catalysts and heterogeneous catalytic reactions. Chem Soc Rev 39:4820–4844
Liang H, Li Z, Wang W, Wu Y, Xu H (2009) Highly surface-roughened “flower-like” silver nanoparticles for extremely sensitive substrates of surface-enhanced Raman scattering. Adv Mater 21:4614–4618
Lim B, Jiang M, Camargo PHC, Cho EC, Tao J, Lu X, Zhu Y, Xia Y (2009) Pd-Pt bimetallic nanodendrites with high activity for oxygen reduction. Science 324:1302–1305
Lu L, Kobayashi A, Kikkawa Y, Tawa K, Ozaki Y (2006) Oriented attachment-based assembly of dendritic silver nanostructures at room temperature. J Phys Chem B 110:23234–23241
Nikoobakht B, El-Sayed MA (2003) Preparation and growth mechanism of gold nanorods (NRs) using seed-mediated growth method. Chem Mater 15:1957–1962
Qiu T, Zhou Y, Li J, Zhang W, Lang X, Cui T, Chu PK (2009) Hot spots in highly Raman-enhancing silver nano-dendrites. J Phys D Appl Phys 42:175403
Rashid MH, Mandal TK (2007) Synthesis and catalytic application of nanostructured silver dendrites. J Phys Chem C 111:16750–16760
Rodríguez-Lorenzo L, de la Rica R, Álvarez-Puebla RA, Liz-Marzán LM, Stevens MM (2012) Plasmonic nanosensors with inverse sensitivity by means of enzyme-guided crystal growth. Nat Mater 11:604–607
Sajanlal PR, Sreeprasad TS, Samal AK, Pradeep T (2011) Anisotropic nanomaterials: structure, growth, assembly, and functions. Nano Rev 2:5883
Sau TK, Rogach AL (2010) Nonspherical noble metal nanoparticles: colloid-chemical synthesis and morphology control. Adv Mater 22:1781–1804
Sau TK, Rogach AL, Jackel F, Klar TA, Feldmann J (2010) Properties and applications of colloidal nonspherical noble metal nanoparticles. Adv Mater 22:1805–1825
Sun Z, Wang C, Yang J, Zhao B, Lombardi JR (2008) Nanoparticle metal-semiconductor charge transfer in ZnO/PATP/Ag assemblies by surface-enhanced Raman spectroscopy. J Phys Chem C 112:6093–6098
Velikov KP, Zegers GE, Blaaderen AV (2003) Synthesis and characterization of large colloidal silver particles. Langmuir 19:1384–1389
Wang ZL (2000) Transmission electron microscopy of shape-controlled nanocrystals and their assemblies. J Phys Chem B 104:1153–1175
Wang Y, Yan B, Chen L (2013) SERS tags: novel optical nanoprobes for bioanalysis. Chem Rev 113:1391–1428
Wen X, Xie YT, Mak MWC, Cheung KY, Li XY, Renneberg R, Yang S (2006) Dendritic nanostructures of silver: facile synthesis, structural characterizations, and sensing applications. Langmuir 22:4836–4842
Yang J, Qi L, Zhang D, Ma J, Cheng H (2004) Dextran-controlled crystallization of silver microcrystals with novel morphologies. Cryst Growth Des 4:1371–1375
Yang J, Lu L, Wang H, Shi W, Zhang H (2006) Glycyl glycine templating synthesis of single-crystal silver nanoplates. Cryst Growth Des 6:2155–2158
Yang J, Wang H, Zhang H (2008) One-pot synthesis of silver nanoplates and charge-transfer complex nanofibers. J Phys Chem C 112:13065–13069
Yang J, Dennis RC, Sardar DK (2011) Room-temperature synthesis of flowerlike Ag nanostructures consisting of single crystalline Ag nanoplates. Mater Res Bull 46:1080–1084
Acknowledgments
This work was supported by the Science Research Foundation of Northwest University (13NW121) and the Education Commission of Shaanxi Province (No. 12JS114, 2014JQ6223).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yang, J., Cao, B., Li, H. et al. Investigation of the catalysis and SERS properties of flower-like and hierarchical silver microcrystals. J Nanopart Res 16, 2651 (2014). https://doi.org/10.1007/s11051-014-2651-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-014-2651-4