Abstract
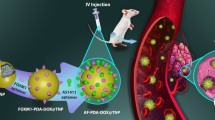
We synthesized c(RGDyK)-coupled superparamagnetic iron oxide nanoparticles for specific delivery of large amount of doxorubicin to α v β 3 integrin rich tumor cells (RDSP) by using a novel triplex hands coupling reagent tris-succinimidyl aminotriacetate and evaluated their structure, drug release, target cell uptake, and cytotoxic effects. Besides the high-trapping efficient for magnetic targeting, RDSP also has integrin α v β 3 targeting property. Moreover, RDSP shows a high-drug load ratio and can carry a large amount of doxorubicin to the target tumor cells. Compare with those of doxorubicin coupled DSP without peptide c(RGDyK) modification, RDSP shows an increase uptake by target tumor cells and stronger tumor cell cytotoxicity. This investigate provides an idiographic way for targeted delivery therapeutic agents to tumors with high efficiency and low-carrier toxicity.








Similar content being viewed by others
References
Alexiou C, Arnold W, Klein RJ, Parak FG, Hulin P, Bergemann C, Erhardt W, Wagenpfeil S, Lubbe AS (2000) Locoregional cancer treatment with magnetic drug targeting. Cancer Res 60:6641–6648
Allen TM, Cullis PR (2004) Drug delivery systems: entering the mainstream. Science 303:1818–1822
Andhariya N, Chudasama B, Mehta RV, Upadhyay RV (2011) Biodegradable thermoresponsive polymeric magnetic nanoparticles: a new drug delivery platform for doxorubicin. J Nanopart Res 13:1677–1688
Arruebo M, Fernández-Pacheco R, Ibarra MR, Santamaría J (2007) Magnetic nanoparticles for drug delivery. Nano Today 2:22–32
Bliss C (1935) The calculation of the dose-mortality curve. Ann Appl Biol 22:134–167
Brulé S, Levy M, Wilhelm C, Letourneur D, Gazeau F, Ménager C, Visage CL (2011) Doxorubicin release triggered by alginate embedded magnetic nanoheaters: a combined therapy. Adv Mater 23:787–790
Du WJ, Xu ZQ, Nystrom AM, Zhang K, Leonard JR, Wooley KL (2008) 19F- and fluorescently labeled micelles as nanoscopic assemblies for chemotherapeutic delivery. Bioconjugate Chem 19:2492–2498
Ferrara N, Kerbel RS (2005) Angiogenesis as a therapeutic target. Nature 438:967–974
Gaihre B, Khilb MS, Lee DR, Kimb HY (2009) Gelatin-coated magnetic iron oxide nanoparticles as carrier system: drug loading and in vitro drug release study. Int J Pharm 365:180–189
Gamel E, Nadia EA, Wortmann L, Arroubb K, Mathur S (2011) SiO2@Fe2O3 core-shell nanoparticles for covalent immobilization and release of sparfloxacin drug. Chem Commun 47:10076–10078
Guo L, Huang J, Zhang X, Li Y, Zheng LM (2008) Bacterial magnetic nanoparticle as a drug carrier. J Mater Chem 18:5993–5997
Guo S, Li D, Zhang L, Li J, Wang E (2009) Monodisperse mesoporous superparamagnetic single-crystal magnetite nanoparticles for drug delivery. Biomaterials 30:1881–1889
Hood JD, Cheresh DA (2002) Role of integrins in cell invasion and migration. Nat Rev Cancer 2:91–100
Hu Y, Xie J, Tong YW, Wang CH (2007) Effect of PEG conformation and particle size on the cellular uptake efficiency of nanoparticles with the HepG2 cells. J Controlled Release 118:7–17
Huang SH, Juang RS (2011) Biochemical and biomedical applications of multifunctional magnetic nanoparticles: a review. J Nanopart Res 13:4411–4430
Jin H, Varner J (2004) Integrins: roles in cancer development and as treatment targets. Br J Cancer 90:561–565
Kang YS, Risbud S, Rabolt JF, Stroeve P (1996) Synthesis and characterization of nanometer-size Fe3O4 and gamma-Fe2O3 particles. Chem Mater 8:2209–2211
Lien YH, Wu TM, Wu JH (2011) Cytotoxicity and drug release behavior of PNIPAM grafted on silica-coated iron oxide nanoparticles. J Nanopart Res 13:5065–5075
Liu X, Ma Z, Xing J, Liu H (2004) Preparation and characterization of amino-silane modified superparamagnetic silica nanospheres. J Magn Magn Mater 270:1–2
Mary M (1997) Applications of magnetic particles in immunoassays In: Hafeli U, Schutt W, Zborowski M (eds) Scientific and Clinical Applications of Magnetic Carriers, Plenum, New York, p 303
Miao QH, Li SP, Han SY, Wang Z, Wu Y, Nie GJ (2012) Construction of hydroxypropyl-b-cyclodextrin copolymer nanoparticles and targeting delivery of paclitaxel. J Nanopart Res 14:1043–1057
Mikhaylov G, Mikac U, Magaeva AA, Itin VI, Naiden EP, Psakhye I, Babes L, Reinhecke T, Peter C, Zeiser R, Bogyo M, Turk V, Psakhye SG, Turk B, Vasiljeva O (2011) Ferri-liposomes as an MRI-visible drug-delivery system for targeting tumours and their microenvironment. Nat Nanotechnol 6:594–602
Minotti G, Menna P, Salvatorelli E, Cairo G, Gianni L (2004) Anthracyclines: molecular advances and pharmacologic developments in antitumor activity and cardiotoxicity. Pharmacol Rev 56:185–229
Nasongkla N, Bey E, Ren J, Ai H, Khemtong C, Guthi JS, Chin SF, Sherry AD, Boothman DA, Gao J (2006) Multifunctional polymeric micelles as cancer-targeted, MRI-ultrasensitive drug delivery systems. Nano Lett 6:2427–2430
Pollakis G, Goormaghtigh E, Ruysschaert JM (1983) Role of the quinine structure in the mitochondrial damage induced by antitumor anthracyclines. FEBS Lett 155:267–272
Santos DP, Ruiz MA, Gallardo V, Zanoni MVB, Arias JL (2011) Multifunctional antitumor magnetite/chitosan-l-glutamic acid (core/shell) nanocomposites. J Nanopart Res 13:4311–4323
Temming K, Meyer DL, Zabinski R, Dijkers ECF, Poelstra K, Molema G, Kok RJ (2006) Evaluation of RGD-targeted albumin carriers for specific delivery of Auristatin E to tumor blood vessels. Bioconjugate Chem 17:1385–1394
Xie J, Chen K, Lee HY, Xu C, Hsu AR, Peng S, Chen X, Sun SH (2008) Ultrasmall c(RGDyK)-coated Fe3O4 nanoparticles and their specific targeting to integrin αvβ3-rich tumor cells. J Am Chem Soc 130:7542–7543
Xiong JP, Stehle T, Zhang R, Joachimiak A, Frech M, Goodman SL, Arnaout MA, Diefenbach B, Dunker R, Scott DL (2002) Crystal structure of the extracellular segment of integrin alpha V beta 3 in complex with an Arg–Gly–Asp ligand. Science 296:151–155
Acknowledgments
This study was financially supported by the Natural Science Foundation of China (NSFC21073091) and Natural Science Foundation of Jiangsu Province (No. BK2009009).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Guo, L., Ding, W. & Zheng, LM. Synthesis and evaluation of c(RGDyK)-coupled superparamagnetic iron oxide nanoparticles for specific delivery of large amount of doxorubicin to tumor cell. J Nanopart Res 15, 1720 (2013). https://doi.org/10.1007/s11051-013-1720-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-013-1720-4