Abstract
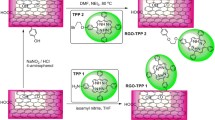
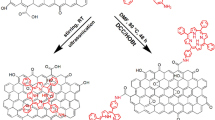
The synthesis of a porphyrin–graphene oxide hybrid (GO–TAP) was carried out by covalently functionalizing graphene oxide (GO) with 5,10,15,20 mesotetra (4-aminophenyl) porphyrin (TAP) through an amide linkage. The GO–TAP hybrid has been characterized by Fourier transform infrared spectroscopy, X-ray photoelectron spectroscopy, and UV–visible spectroscopy. The peak intensity of the Soret band of the material was suppressed compared to neat TAP. This indicates a strong interaction between the electronic energy level of TAP and GO in the GO–TAP hybrid. The functionalization of GO with TAP significantly improved its solubility and dispersion stability in organic solvents. Scanning electron micrographs reveal that the hybrid was found to be similar to the unmodified GO but slightly more wrinkled. Transmission electron micrographs also demonstrate that GO sheet in the hybrid is more wrinkled with some dark spot due to functionalization. Atomic force microscopy results also reveal that the TAP functionalization increases the thickness of GO sheet to 2.0–3.0 nm from 1.2 to 1.8 nm. We observed improved nonlinear optical and optical limiting properties for the hybrid compared to both graphene oxide and porphyrin. GO–TAP shows fluorescence quenching compared with porphyrin, indicating excellent electron and/or energy transfer to GO from TAP. Thermogravimetric analysis confirms that the GO–TAP hybrid has outstanding thermal stability.






Similar content being viewed by others
References
Adler AD, Longo FR, Finarelli JD, Goldmacher J, Assour J, Korsakoff L (1967) A simplified synthesis for meso-tetraphenylporphine. J Org Chem 32:476
Akhavan O (2010) The effect of heat treatment on formation of graphene thin films from graphene oxide nanosheets. Carbon 48:509–519
Baker SE, Cai W, Lasseter TL, Weidkamp KP, Hamers RJ (2002) Covalently bonded adducts of deoxyribonucleic acid (DNA) oligonucleotides with single-wall carbon nanotubes: synthesis and hybridization. Nano Lett 2:1413–1417
Bala Murali Krishna M, Venkatramaiah N, Venkatesan R, Narayana Rao D (2012) Synthesis and structural, spectroscopic and nonlinear optical measurements of graphene oxide and its composites with metal and metal free porphyrins. J Mater Chem 22:3059–3068
Bettelheim A, White BA, Raybuck SA, Murray RW (1987) Electrochemical polymerization of amino-, pyrrole-, and hydroxy-substituted tetraphenylporphyrins. Inorg Chem 26:1009–1017
Geim AK, Novoselov KS (2007) The rise of graphene. Nat Mater 6:183–191
Geng J, Jung H-T (2010) Porphyrin functionalized graphene sheets in aqueous suspensions: from the preparation of graphene sheets to highly conductive graphene films. J Phys Chem C 114:8227–8234
Gilje S, Han S, Wang M, Wang KL, Kaner RB (2007) A chemical route to graphene for device applications. Nano Lett 7:3394–3398
Hasobe T, Fukuzumi S, Kamat PV (2006) Organized assemblies of single wall carbon nanotubes and porphyrin for photochemical solar cells: charge injection from excited porphyrin into single-walled carbon nanotubes. J Phys Chem B 110:25477–25484
Hummers WS, Offeman RE (1958) Preparation of graphitic oxide. J Am Chem Soc 80:1339
Karousis N, Sandanayaka ASD, Hasobe T, Economopoulos SP, Sarantopoulou E, Tagmatarchis N (2011) Graphene oxide with covalently linked porphyrin antennae: synthesis, characterization and photophysical properties. J Mater Chem 21:109–117
Kim DY, Ahn TK, Kwon JH, Kim D, Ikeue T, Aratani N, Osuka A, Shigeiwa M, Maeda S (2005) Large two-photon absorption (tpa) cross-section of directly linked fused diporphyrins. J Phys Chem A 109:2996–2999
Krishna MBM, Kumar VP, Venkatramaiah N, Venkatesan R, Rao DN (2011) Nonlinear optical properties of covalently linked graphene-metal porphyrin composite materials. Appl Phys Lett 98:081103–081106
Li H, Zhou B, Lin Y, Gu L, Wang W, Fernando KAS, Kumar S, Allard LF, Sun Y-P (2004) Selective interactions of porphyrins with semiconducting single-walled carbon nanotubes. J Am Chem Soc 126:1014–1015
Liu Z, Liu Q, Huang Y, Ma Y, Yin S, Zhang X, Sun W, Chen Y (2008a) Organic photovoltaic devices based on a novel acceptor material: graphene. Adv Mater 20:3924–3930
Liu ZB, Tian JG, Guo Z, Ren DM, Du F, Zheng JY, Chen YS (2008b) Enhanced optical limiting effects in porphyrin-covalently functionalized single-walled carbon nanotubes. Adv Mater 20:511–515
Liu Y, Zhou J, Zhang X, Liu Z, Wan X, Tian J, Wang T, Chen Y (2009a) Synthesis, characterization and optical limiting property of covalently oligothiophene-functionalized graphene material. Carbon 47:3113–3121
Liu ZB, Xu YF, Zhang XY, Zhang XL, Chen YS, Tian JG (2009b) Porphyrin and fullerene covalently functionalized graphene hybrid materials with large nonlinear optical properties. J Phys Chem B 113:9681–9686
Luguya R, Jaquinod L, Fronczek FR, Vicente MGaH, Smith KM (2004) Synthesis and reactions of meso-(p-nitrophenyl)porphyrins. Tetrahedron 60:2757–2763
Marder SR, Kippelen B, Jen AKY, Peyghambarian N (1997) Design and synthesis of chromophores and polymers for electro-optic and photorefractive applications. Nature 388:845–851
Meng H, Sui GX, Fang PF, Yang R (2008) Effects of acid- and diamine-modified MWNTs on the mechanical properties and crystallization behavior of polyamide 6. Polymer 49:610–620
NÃ- Mhuircheartaigh ÃinM, Giordani S, Blau WJ (2006) Linear and nonlinear optical characterization of a tetraphenylporphyrin−carbon nanotube composite system. J Phys Chem B 110:23136–23141
Park S, An J, Piner RD, Jung I, Yang D, Velamakanni A, Nguyen ST, Ruoff RS (2008) Aqueous suspension and characterization of chemically modified graphene sheets. Chem Mater 20:6592–6594
Pham VH, Pham HD, Dang TT, Hur SH, Kim EJ, Kong B-S, Kim S, Chung JS (2012) Chemical reduction of an aqueous suspension of graphene oxide by nascent hydrogen. J Mater Chem 22:10530–10536
Qu K, Xu H, Zhao C, Ren J, Qu X (2011) Amine-linker length dependent electron transfer between porphyrins and covalent amino-modified single-walled carbon nanotubes. RSC Adv 1:632–639
Ramanathan T, Fisher FT, Ruoff RS, Brinson LC (2005) Amino-functionalized carbon nanotubes for binding to polymers and biological systems. Chem Mater 17:1290–1295
Senge MO, Fazekas M, Notaras EGA, Blau WJ, Zawadzka M, Locos OB, Ni Mhuircheartaigh EM (2007) Nonlinear optical properties of porphyrins. Adv Mater 19:2737–2774
Subrahmanyam KS, Vivekchand SRC, Govindaraj A, Rao CNR (2008) A study of graphenes prepared by different methods: characterization, properties and solubilization. J Mater Chem 18:1517–1523
Venugopal G, Krishnamoorthy K, Mohan R, Kim S-J (2012) An investigation of the electrical transport properties of graphene-oxide thin films. Mater Chem Phys 132:29–33
Wojcik A, Kamat PV (2010) Reduced graphene oxide and porphyrin. An interactive affair in 2-D. ACS Nano 4:6697–6706
Wu W, Zhang S, Li Y, Li J, Liu L, Qin Y, Guo Z-X, Dai L, Ye C, Zhu D (2003) PVK-modified single-walled carbon nanotubes with effective photoinduced electron transfer. Macromolecules 36:6286–6288
Xu Y, Liu Z, Zhang X, Wang Y, Tian J, Huang Y, Ma Y, Zhang X, Chen Y (2009) A graphene hybrid material covalently functionalized with porphyrin: synthesis and optical limiting property. Adv Mater 21:1275–1279
Zhang X, Feng Y, Lv P, Shen Y, Feng W (2010) Enhanced reversible photoswitching of azobenzene-functionalized graphene oxide hybrids. Langmuir 26:18508–18511
Zhu Y, Murali S, Cai W, Li X, Suk JW, Potts JR, Ruoff RS (2010) Graphene and graphene oxide: synthesis, properties, and applications. Adv Mater 22:3906–3924
Acknowledgments
Dr. R. Yamuna and Ms. R. Devi thank the Council of Scientific and Industrial Research (CSIR, Project No: 01(2256)/08/EMR-II), New Delhi, India for their funding. Ms. D. Keerthy thanks the Department of Science and Technology (DST), India for INSPIRE fellowship.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yamuna, R., Ramakrishnan, S., Dhara, K. et al. Synthesis, characterization, and nonlinear optical properties of graphene oxide functionalized with tetra-amino porphyrin. J Nanopart Res 15, 1399 (2013). https://doi.org/10.1007/s11051-012-1399-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-012-1399-y