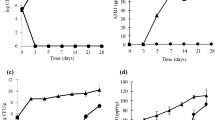
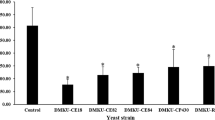
Abstract
Various species of fungi in the genus Aspergillus are the most common causative agents of invasive aspergillosis and/or producers of hepato-carcinogenic mycotoxins. Salicylaldehyde (SA), a volatile natural compound, exhibited potent antifungal and anti-mycotoxigenic activities to A. flavus and A. parasiticus. By exposure to the volatilized SA, the growth of A. parasiticus was inhibited up to 10–75% at 9.5 mM ≤ SA ≤ 16.0 mM, while complete growth inhibition was achieved at 19.0 mM ≤ SA. Similar trends were also observed with A. flavus. The aflatoxin production, i.e., aflatoxin B1 and B2 (AFB1, AFB2) for A. flavus and AFB1, AFB2, AFG1, and AFG2 for A. parasiticus, in the SA-treated (9.5 mM) fungi was reduced by ~13–45% compared with the untreated control. Using gene deletion mutants of the model yeast Saccharomyces cerevisiae, we identified the fungal antioxidation system as the molecular target of SA, where sod1Δ [cytosolic superoxide dismutase (SOD)], sod2Δ (mitochondrial SOD), and glr1Δ (glutathione reductase) mutants showed increased sensitivity to this compound. Also sensitive was the gene deletion mutant, vph2Δ, for the vacuolar ATPase assembly protein, suggesting vacuolar detoxification plays an important role for fungal tolerance to SA. In chemosensitization experiments, co-application of SA with either antimycin A or strobilurin (inhibitors of mitochondrial respiration) resulted in complete growth inhibition of Aspergillus at much lower dose treatment of either agent, alone. Therefore, SA can enhance antifungal activity of commercial antifungal agents required to achieve effective control. SA is a potent antifungal and anti-aflatoxigenic volatile that may have some practical application as a fumigant.



Similar content being viewed by others
References
Martin MD, Rex JH. Antifungal drug resistance: a focus on Candida. Clin Updates Fungal Infec. 1997;1:238–41.
Moore CB, Sayres N, Mosquera J, et al. Antifungal drug resistance in Aspergillus. J Infect. 2000;41:203–20.
Cowen LE, Kohn LM, Anderson JB. Divergence in fitness and evolution of drug resistance in experimental populations of Candida albicans. J Bacteriol. 2001;183:2971–8.
Ghannoum MA, Rice LB. Antifungal agents: mode of action, mechanisms of resistance, and correlation of these mechanisms with bacterial resistance. Clin Microbiol Rev. 1999;12:501–17.
Luo H, Morsomme P, Boutry M. The two major types of plant plasma membrane H+ -ATPases show different enzymatic properties and confer differential pH sensitivity of yeast growth. Plant Physiol. 1999;119:627–34.
Beekrum S, Govinden R, Padayachee T, et al. Naturally occurring phenols: a detoxification strategy for fumonisin B1. Food Addit Contam. 2003;20:490–3.
Kim JH, Yu J, Mahoney N, et al. Elucidation of the functional genomics of antioxidant-based inhibition of aflatoxin biosynthesis. Int J Food Microbiol. 2008;122:49–60.
Aziz NH, Farag SE, Mousa LA, et al. Comparative antibacterial and antifungal effects of some phenolic compounds. Microbios. 1998;93:43–54.
Curir P, Dolci M, Dolci P, et al. Fungitoxic phenols from carnation (Dianthus caryophyllus) effective against Fusarium oxysporum f. sp. dianthi. Phytochem Anal. 2003;14:8–12.
Kim J, Campbell B, Mahoney N, et al. Chemosensitization prevents tolerance of Aspergillus fumigatus to antimycotic drugs. Biochem Biophys Res Commun. 2008;372:266–71.
Kim JH, Mahoney N, Chan KL, et al. Chemosensitization of fungal pathogens to antimicrobial agents using benzo analogs. FEMS Microbiol Lett. 2008;281:64–72.
Smits GJ, Brul S. Stress tolerance in fungi—to kill a spoilage yeast. Curr Opin Biotechnol. 2005;16:225–30.
Jaeger T, Flohe L. The thiol-based redox networks of pathogens: unexploited targets in the search for new drugs. Biofactors. 2006;27:109–20.
Guillen F, Evans CS. Anisaldehyde and veratraldehyde acting as redox cycling agents for H2O2 production by Pleurotus eryngii. Appl Environ Microbiol. 1994;60:2811–7.
Shvedova AA, Kommineni C, Jeffries BA, et al. Redox cycling of phenol induces oxidative stress in human epidermal keratinocytes. J Invest Dermatol. 2000;114:354–64.
Jacob C. A scent of therapy: pharmacological implications of natural products containing redox-active sulfur atoms. Nat Prod Rep. 2006;23:851–63.
Schwartz MA, Madhani HD. Principles of MAP kinase signaling specificity in Saccharomyces cerevisiae. Annu Rev Genet. 2004;38:725–48.
Levin DE. Cell wall integrity signaling in Saccharomyces cerevisiae. Microbiol Mol Biol Rev. 2005;69:262–91.
Toone WM, Jones N. Stress-activated signalling pathways in yeast. Genes Cells. 1998;3:485–98.
Hamilton AJ, Holdom MD. Antioxidant systems in the pathogenic fungi of man and their role in virulence. Med Mycol. 1999;37:375–89.
Clemons KV, Miller TK, Selitrennikoff CP, et al. fos-1, a putative histidine kinase as a virulence factor systemic aspergillosis. Med Mycol. 2002;40:259–62.
Campbell BC, Molyneux RJ, Schatzki TF. Current research on reducing pre- and post-harvest aflatoxin contamination of U. S. almond, pistachio and walnut. J Toxicology-Toxin Rev. 2003;22:225–66.
Food and Drug Administration (FDA). Compliance policy guides manual. Washington, DC: U.S. FDA; 1996. Sec. 555.400, 268; Sec. 570.500, 299.
European Commission (EC). Commission regulation (EU) No. 165/2010 of 26 February 2010 amending regulation (EC) No. 1881/2006 setting maximum levels for certain contaminants in foodstuffs as regards aflatoxins. Off J Eur Union. 2010; L50:8–12.
Gross J, Schumacher K, Schmidtberg H, et al. Protected by fumigants: beetle perfumes in antimicrobial defense. J Chem Ecol. 2008;34:179–88.
Official Methods of Analysis of AOAC INTERNATIONAL. 18th ed. Gaithersburg, MD: AOAC INTERNATIONAL; 2005. Official method 971.22.
Vincent JM. Distortion of fungal hyphae in the presence of certain inhibitors. Nature. 1947;159:850.
Sekiyama Y, Mizukami Y, Takada A, et al. Effect of mustard extract vapour on fungi and spore-forming bacteria. J Antibact Antifung Agent. 1994;24:171–8.
Winzeler EA, Shoemaker DD, Astromoff A, et al. Functional characterization of the S. cerevisiae genome by gene deletion and parallel analysis. Science. 1999;285:901–6.
Tucker CL, Fields S. Quantitative genome-wide analysis of yeast deletion strain sensitivities to oxidative and chemical stress. Comp Funct Genomics. 2004;5:216–24.
Kim JH, Campbell BC, Yu J, et al. Examination of fungal stress response genes using Saccharomyces cerevisiae as a model system: targeting genes affecting aflatoxin biosynthesis by Aspergillus flavus Link. Appl Microbiol Biotechnol. 2005;67:807–15.
Rep M, Proft M, Remize F, et al. The Saccharomyces cerevisiae Sko1p transcription factor mediates HOG pathway-dependent osmotic regulation of a set of genes encoding enzymes implicated in protection from oxidative damage. Mol Microbiol. 2001;40:1067–83.
Ammar H, Michaelis G, Lisowsky T. A screen of yeast respiratory mutants for sensitivity against the mycotoxin citrinin identifies the vacuolar ATPase as an essential factor for the toxicity mechanism. Curr Genet. 2000;37:277–84.
Dietz KJ, Tavakoli N, Kluge C, et al. Significance of the V-type ATPase for the adaptation to stressful growth conditions and its regulation on the molecular and biochemical level. J Exp Bot. 2001;52:1969–80.
Hamilton CA, Taylor GJ, Good AG. Vacuolar H(+)-ATPase, but not mitochondrial F(1)F(0)-ATPase, is required for NaCl tolerance in Saccharomyces cerevisiae. FEMS Microbiol Lett. 2002;208:227–32.
MacDiarmid CW, Milanick MA, Eide DJ. Biochemical properties of vacuolar zinc transport systems of Saccharomyces cerevisiae. J Biol Chem. 2002;277:39187–94.
Nelson N. The vacuolar H(+)-ATPase: one of the most fundamental ion pumps in nature. J Exp Biol. 1992;172:19–27.
Wood PM, Hollomon DW. A critical evaluation of the role of alternative oxidase in the performance of strobilurin and related fungicides acting at the Qo site of complex III. Pest Manag Sci. 2003;59:499–511.
Takimoto H, Machida K, Ueki M, et al. UK-2A, B, C and D, novel antifungal antibiotics from Streptomyces sp. 517–02. IV. Comparative studies of UK-2A with antimycin A3 on cytotoxic activity and reactive oxygen species generation in LLC-PK1 cells. J Antibiotics. 1999;52:480–4.
Acknowledgments
This research was conducted under USDA-ARS CRIS Project 5325-42000-035-00D. We thank the Almond Board of California for partially funding this research (Agreement No. 58-5325-9-156).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, J.H., Campbell, B.C., Mahoney, N. et al. Chemosensitization of Aflatoxigenic Fungi to Antimycin A and Strobilurin Using Salicylaldehyde, a Volatile Natural Compound Targeting Cellular Antioxidation System. Mycopathologia 171, 291–298 (2011). https://doi.org/10.1007/s11046-010-9356-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11046-010-9356-8